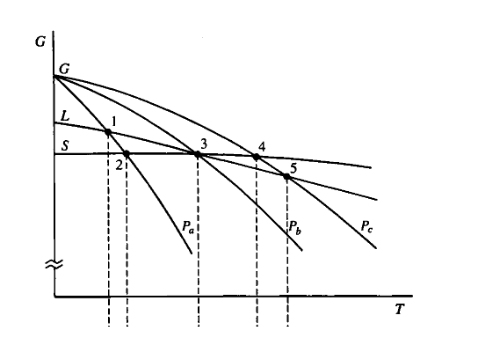
(4) The following plot corresponds schematically to the behavior of the chemical potential for the three different phases of a substance as function of temperature for three different pressures P, < Pp < Pc. Use this information to sketch the P-T phase diagram of this substance. Identify the melting, sublimation and triple points on the diagram.
(4) The following plot corresponds schematically to the behavior of the chemical potential for the three different phases of a substance as function of temperature for three different pressures P, < Pp < Pc. Use this information to sketch the P-T phase diagram of this substance. Identify the melting, sublimation and triple points on the diagram.
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter6: Equilibria In Single-component Systems
Section: Chapter Questions
Problem 6.50E
Related questions
Question
100%

Transcribed Image Text:(4) The following plot corresponds schematically to the behavior of the chemical
potential for the three different phases of a substance as function of temperature for
three different pressures Pa < Pp < Pc. Use this information to sketch the P-T phase
diagram of this substance. Identify the melting, sublimation and triple points on the
diagram.
Transcribed Image Text:Pc
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,