Q: it is the correct order of stability of the following radicals (more stable > less stable)?
A: Carbon forms four bonds to complete its octet. In the formation of free radical,carbon has formed…
Q: Rank the carbocation compounds below from most stable to least stable.
A:
Q: Draw curved arrows to show the movement of the electrons as one resonance contributor is converted…
A: Resonance contributors are the ones which are formed by moving a single electron resulting in…
Q: Rank the following from most to least stable: OH ОН OH C A,
A: Applying concept of stability of alkene with the help of hyperconjugation and stereochemistry of…
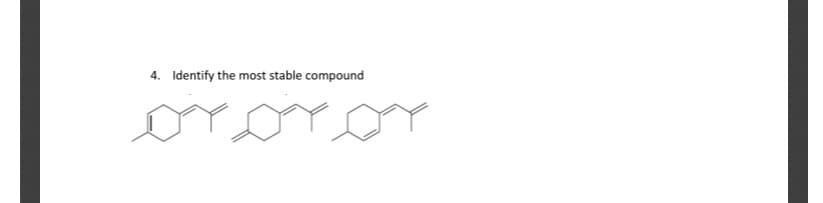
Q: Rank the alkenes shown below based on stability, should be indicated as (most stable (left) to least…
A: The correct order of the stability of alkene can be done on the basis of substitution. Higher the…
Q: Ql- Arrange the following carbonium ion as more stable. Why? Me Me Me A B
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Complete energy diagram for the two-step conversion of A–B + C:−→ A:− + B–C
A: SOLUTION: Step 1: The overall reaction given is as shown below: A-B + C: -→ A: - +B-C A possible…
Q: んnん or
A: NOTE : Since you've asked multiple questions,we'll solve the first question for you.If you want any…
Q: Calculate the heat of reaction deltaH for the following reaction: 2HCl(g)+Br2(g)——>2HBr(g)+Cl2(g)…
A: We have to calculate ., heat of reaction ∆H for the following reaction: 2HCl(g)+Br2…
Q: Calculate the heat of reaction deltaH for the following reaction: 2HCl(g)+Br2(g)——> 2HBr2(g)+ Cl2(g)…
A: Given : We have to calculate delta H.
Q: Which compound is more Stable? why?
A: Given, The structures are; Aromatic compounds are more stable than anti-aromatic and non-aromatic…
Q: Place from high stability to lowest stability for 2,4 dimethyl pentane
A: The stability of any compound is inversely proportional to the steric repulsion between the groups…
Q: Which of the following bonds absorbs higher energy? O--H or C--H? Why?
A: Given that, Bonds, O----H & C----H Which of the following bonds absorbs higher energy?
Q: Rank the following in order of stability, from most stable -1 to least stable - 4
A:
Q: What is the correct order of stability of the following radicals (more stable > less stable)?
A:
Q: lentify the 3 major contributors to the resonance hybrids A B C E OZI ZI ZI ZI OZI OZI
A:
Q: Name the following:
A: Given,
Q: CEN
A: Select the principle carbon chain Numbering Naming (prefix + word root + suffix)
Q: In an associative substitution reaction а. AS# > 0 b. AS# 0
A: Entropy is the measure of randomness or molecular disorder in a system. It is numerically equal to…
Q: Rank the structures shown from most to least stable.
A: The given structure can be ranked from most stable to least stable
Q: 5. Explain why A is much more stable than B? B NH
A: To explain why compound (A) is more stable than the compound (B)
Q: 7. In each pair, circle the compound that is more stable.
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Draw one significant resonance contributor and show the electron movement between the contributors…
A: Applying concept of formation of resonating structure.
Q: 3. In each pair, circle the species that is more stable. OH2 OH2
A: In the first pair, the second structure is more stable. The stability of the carbonation depends on…
Q: Question 8 Rank the following carbocations from most stable to least stable: II II O III >I> II OI>…
A: Given that, Three carbocations have been given,we have to find out order of stability?
Q: Which compound is the most stable?
A: When there is more charge separation in between opposite charges, stability of resonating structures…
Q: Between the following radicals; which one is more stable? I or II PHHC(*)CH2ET (1) PHCH2C(*)HEt (1I)…
A: Given free radicals are PhHC(•)CH2Et (I) PhCH2C(•)HEt (II) The free radical with more…
Q: Draw all three stereoisomers of the compound shown below. Label each structure vou draw according to…
A: Draw all three stereoisomers of the compound shown below. Label each structure you draw according to…
Q: (a) Br2 FeBr3 (b) NaCN CI (c) HO H,SO, 180 °C Major product
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub- parts for…
Q: Which properly shows resonance of an allylic system? B C H. O: D
A: Resonating structures are the different structures which are obtained by the movement of pi of…
Q: Draw the given carbocation in orbital phase. CH2 CH2 (b) (a) (d) (c) H.
A: Correct answer is (b). Benzyl carbocation is resonance stabilized. Correct resonance structure is…
Q: Label each C–C double bond as E or Z.
A: The E isomer has the two higher priority groups on the opposite sides. The Z isomer has the two…
Q: 4. In each pair, circle the anion that is more stable. CI
A: An ion bearing a positive charge is called anion. Cations and anions are present in ionic compounds.…
Q: heat OH
A: In the above reaction, reactant in presence of H3O+, dehydration and rearrangement takes place.
Q: If you were to hydrogenate molecule A shown below, how many molecules of H2 would you expect to be…
A: The hydrogenation reaction is the type of addition reaction in which the unsaturated doubles are…
Q: Draw curved arrows to show the movement of the electrons as one resonance contributor is converted…
A: The resonance structures arise when the Lewis structure of the molecule can be represented in more…
Q: O b. Which compound is most likely to be stable? B2 B₂H6 BCN BO3
A: All compounds have a 'B' atom as a central atom. Electronic configuration of B = 1s22s22p1. If B…
Q: Arrange the following compounds in order from the most stable to the least stable.
A: The stability order of the given compound from most stable to least stable can be arranged as,
Q: Rank the following compounds from least to most stable. Least stable Most stable
A: The stability of alkenes can be given by resonance or hyperconjugation. Hyperconjugation and…
Q: 4) Assign E /Z designation to each double bond where applicable. a) b) N, NH Br c)
A: Stereoisomers are the molecules having same molecular formula but differ only on the spatial…
Q: Which one is the most stable?
A: The more substituents the alkenes have, the more stable they are. Thus, a tetra substituted alkene…
Q: Which of the following resonance structures is the greatest contributor to the resonance hybrid? O:
A:
Q: Rank the indicated bonds in order of increasing bond dissociationenergy.
A: Order of increasing bond dissociation energy will be
Q: Draw the following: 1. The chair structures of the most stable molecule below. (Choose the most…
A: This question belong to stereochemistry organic chemistry. We will decide the stability of 2…
Q: Rank the compounds in order of increasing stability (i.e. least stable to most stable). 02 < 02 <…
A: Bond order is calculated from MO diagram from the given equation = e in bonding orbital _ e in…
Q: Which is more stable?
A:
Q: :o: CH2 CH2
A:
Q: 2. Arrange the structures below in terms of most stable to least stable. Ans a. A B b. Y Ans
A: The stability of carbocations can be explained by Inductive effect and Hyper conjunction . The…
Q: Which molecule has the best leaving groue A.CHISH B CH3OCH3 C.CH3 NHCH3 D.CH30H E.CH3 CH2 SCH3 CH,…
A: More is the acidity of leaving group , better is leaving group .
Can i get help with this problem please
Step by step
Solved in 2 steps with 2 images

- How many of the following elements to form compounds with an extended Octect Pb Kr Si BExplain the differences in bond dissociation energies and bond lengths on addition and removalof an electron from NO.pls help ASAP! “which term best describes the relationship between the following two molecules?”

