Q: Ideally, the van 't Hoff factor should be equal to the number of ions that make up a compound. In…
A: Van't Hoff factor : It is defined as the ratio of the concentration of particles formed when a…
Q: Explain the influence of the electronegative atoms like chlorine on radical stability.
A: Introduction to Electronegativity It is a measure of the atom's ability to attract electrons to…
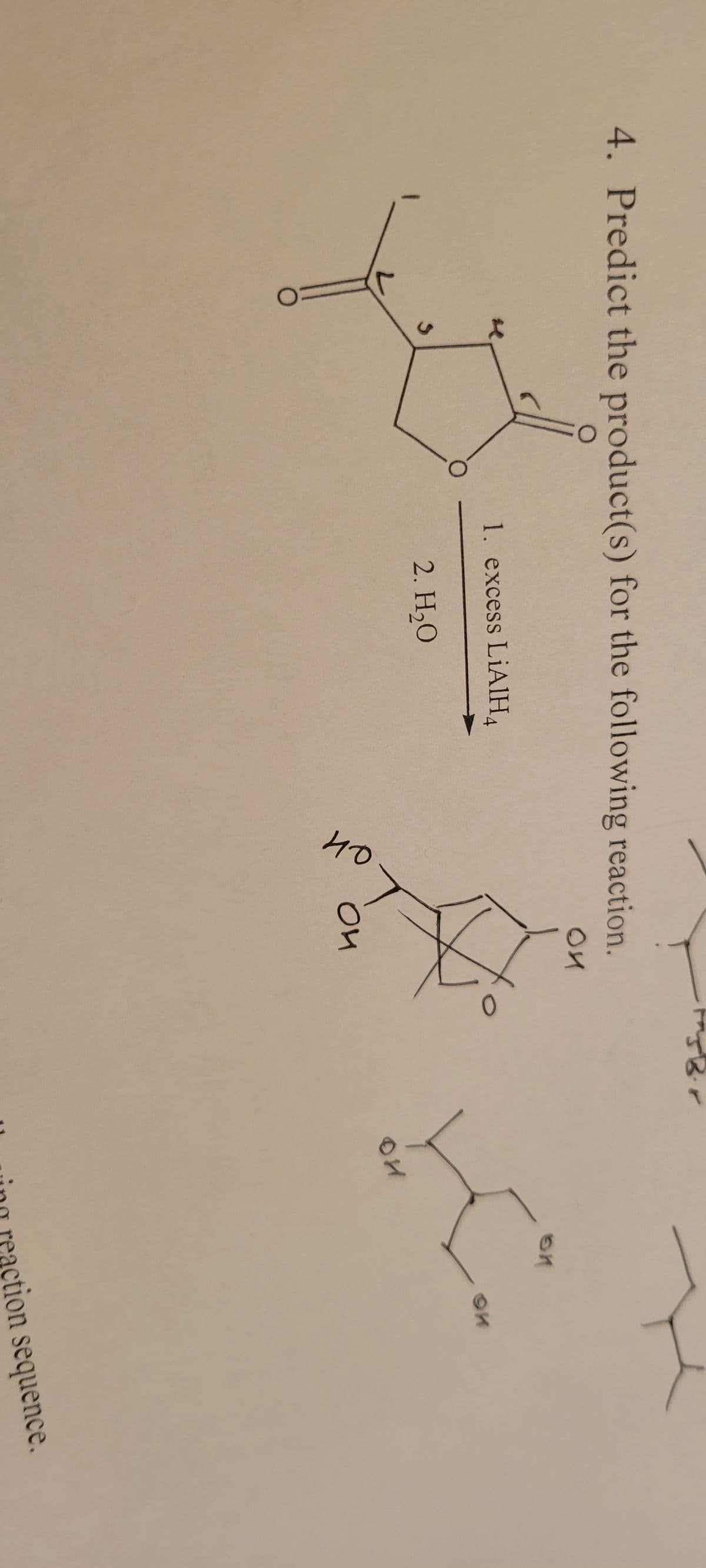
Q: 4. Write the condensed formula of the following structures. H 3. H Н I N- hydroxylamine H-Q…
A:
Q: 4. Aluminum sulfate (Al2(SO4)3) is an aluminum-based coagulant that is used during water and…
A:
Q: 1.74. What is the ratio of the populations of two energy states whose energies differ by 1000 J at…
A:
Q: Accurately and precisely record the volume reading for a liquid in a 100-ml graduated cylinder in…
A: The given cylinder is a 100 mL graduated cylinder with least count of 1 mL There are markings for…
Q: Give mechanism of this reaction ASAP
A:
Q: Two heat capacity values are given in the task. Why are two different heat capacities needed?
A: Heat capacity is the amount of heat to be supplied to a substance to increase its temperature by 1…
Q: The vapour pressures of the pure components P and Q are 700 Torr and 500 Torr, respectively. When…
A: Given: Vapor Pressure of Pure P = 700 torr Vapor Pressure of Pure q = 500 torr Two phases are in…
Q: 7) How many isomers can you construct for the molecular formula C,H,O? Use a red ball with two holes…
A: Isomers are those compounds which have the same molecular formula but different structures either…
Q: . Use secondary resonance to explain why phenol is an activator and an ortho/para director. : OH
A:
Q: ▼ Part A F10 Convert 8.8 kcal to its value in calories. Express your answer to two significant…
A: We will convert kcal into calories.
Q: Question 4 A sample of 3.15 moles of a substance was burned in a constant pressure calorimeter. The…
A: We have to calculate the enthalpy of combustion for the reaction
Q: Starting from benzene, how will you prepare the target? Show all the steps in the correct order. NH₂…
A:
Q: 24. Upon titration, a mixture of weak acid and another weak acid whose Ka values are not too…
A: Equivalence point is the volume of the titrant at which the moles of titrant added equal to the…
Q: Consider the following equilibrium: Fe(OH)3 (s) + 3H* (ag) Fe3* (ag) + 3H₂O (1) a) Given a Kap of…
A: Ksp is the solubility product constant which is determined as the equilibrium constant for a solid…
Q: 2 CH₂OH(g) + H₂(g) → C₂H, (g) + 2 H₂O(g) Predict the signs of AHrxn and ASrxn for the following…
A: The enthalpy change of the reaction measures the amount of heat energy absorbed or released by the…
Q: Exp 2 given compound set number (from Canvas): 5 Compound 1, number: 12 LOO Student name: Katherine…
A: A question based on IR spectroscopy. Two organic compounds, their structure and the respective IR…
Q: 1.72. Calculate the average grade using equation 1.32 if the scores on a 10-point quiz are 5, 5, 5,…
A: Since, Average of the given sample or data is calculated as sum of all the data divided by the…
Q: what is number of 3d electrons of the transition metal ion in [Ni(acac)2(OH2)2]
A: Transition metal complex [Ni(acac)2(OH)2]
Q: Natural gas is a naturally occurring fossil fuel that is abundant in Alberta. Based on typical…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: You have a sample of 121.5 g of C₂H6. How many moles of C₂H6 are in the sample?
A:
Q: 1.38. Calculate the Boyle temperatures for carbon dioxide, oxygen, and nitrogen using the van der…
A: Boyle temperature is that particular temperature at which real gas tends to behave like ideal gas.…
Q: Consider the following reaction: PC15 (9) PCl3(g) + Cl₂ (9) If 0.00686 moles of PC15 (g), 0.438…
A: We know, Concentration = Number of moles / Volume
Q: What is the approximate concentration of free Ag" ion at equilibrium when 1.15E-2 mol silver nitrate…
A:
Q: A student determined the concentration of total reducible iodine in the aqueous phase based on the…
A: Given: Total reducible iodine in the aqueous phase is 0.0177M. The concentration of I2 in the CH2Cl2…
Q: Given the information A+B C 2D D calculate AG at 298 K for the reaction A + B 2C ᎪG° = AH = -641.9…
A: Given : reaction and thermodynamic data
Q: Please answer the following study questions: How many electrons can be found in the following? a)…
A:
Q: calculate the heat lose (in Joules) by 115 g of water as it cools from 71.4 C to 34.4 C
A: Given data, Mass of water = 115 g Initial temperature = 71.4oC Final temperature = 34.4oC
Q: (b) oct-2-sulfane S.
A: When sulpher is present in a hydrocarbon chain, i.e when there are 2 alkyl substituents with a…
Q: draw the lewis structures for benzophenone and 4-hydroxybenzoic acid
A: Given : name of compounds
Q: Infrared spectroscopy is most useful for
A: Infrared spectroscopy is most useful for determining the molecular structure of a substance.…
Q: Which substance has the highest/lowest van't hoff factor? Explain how the charge and radii size…
A: The van't Hoff factor (i) is a measure of the number of particles that a substance dissociates into…
Q: What is the number of moles in 4.5 x 1024 atoms of oxygen? A 7.5 moles B C D 8.2 moles 9.5 moles…
A:
Q: Use the References to access important values if needed for this question. Under certain conditions,…
A: Given : mass of n2 = 25.8 g Mass of H2 = 5.6 g Tip : write reaction first
Q: A student performed three aqueous layer titrations in Part 1 and they reported the following…
A: To answer: Which determinations were the student's 'best two'? Calculate the percent difference for…
Q: 2. What quantity should be dispensed when 50 mg of amitryptyline has been prescribed two times per…
A: Dosage of medication
Q: aqueous layer OH (acid) "HCI layer" + dilute NaOH (aq) solution + NaCl (aq) (neutral) (base) +…
A: In aqueous solution, ionic compounds are mostly soluble where in organic layer organic compounds are…
Q: In a fluorescence resonance energy transfer experiment... a) Minor conformational changes can induce…
A: FRET (Förster Resonance Energy Transfer) is a phenomenon in which energy is transferred from one…
Q: * 4. CH₂ Circle the pairs of organic solids which can be separated from each other by solvent…
A: Solvent extraction: It is a method of separating compounds based on their relative solubilities in…
Q: CH3CH₂ CH3 CH3 CH₂CH3 CH₂CH3
A: IUPAC name of the molecule based on nomenclature:
Q: 7. Н H₂C CH3 А
A: A cycloaddition reaction in which a diene reacts with a dienophile in the presence of heat and forms…
Q: Given the Ksp = 1.4 x 10-13, what is the solubility of CoCO3 (i.e., what is the [Co2+]…
A:
Q: C. Write the electron configuration of the following atoms and identify what Family and period do…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Freezing point depression can be used to experimentally determine the van 't Hoff factor of a solute…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: Match the following aqueous solutions with the appropriate letter from the column on the right.…
A: with increasing the number of particles or ions, depression of freezing point occurs. Thus solution…
Q: Humidity is the percentage of partial pressure of water in air over the total possible water in the…
A: Introduction Humidity is an important concept in chemistry. It refers to the amount of water vapor…
Q: Atomic mass of a metal can be linked to its specific heat capacity in J/goC units by a simple…
A:
Q: 2. Assign the formal charges of each atom in the structures below. O || C. a. CIO HH S H C H H N-H…
A: Given
Q: Question 9 Identify the different types of protons in 1-chdoro-2-tuunane. What would be the chemical…
A: Chemical shift of following H are.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Fill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.Trimethyllead hydride, (CH3)3PbH, can react with methyl bromide (CH3Br) in a homolytic dissociation reaction. What will the lead-containing product be? (CH3)2Pb PbBr2 (CH3)3PbBr Pb metalI’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?
- What are the final products for these reactions? *mechanisms not necessaryIllustrate the Addition of X2 to an Alkene—Halogenation ?What will be the major product obtained from the reaction of Br2 with 1-butene if the reaction is carriedout ina. dichloromethane? b. water? c. ethyl alcohol? d. methyl alcohol?
- What products will be obtained from the E1 reaction of the alkyl halides in Problem 45?The nitroso group, −N=O, is one of the few nonhalogens that is an ortho- and para-directing deactivator. Explain this behaviour by drawing resonance structures of the carbocation intermediates in ortho, meta, and para electrophilic reaction onnitrosobenzene, C6H5−N=O.Given: Mass spec for 4-acetylbiphenyl. Knowing that 196 m/z is the major product, how would you assign 181 m/z and 152 m/z? (i've also attached a picture of the reaction scheme)

