4. What is the theoretical yield of ammonia in grams? 5. What is the percent yield if the reaction actually produced a 0.924 grams of ammonia?
4. What is the theoretical yield of ammonia in grams? 5. What is the percent yield if the reaction actually produced a 0.924 grams of ammonia?
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 33E: Chlorine gas was first prepared in 1774 by C. W. Scheele by oxidizing sodium chloride with...
Related questions
Question
A chemist combines 3.12g of magnesium nitride with 1.770g of water which react to produce magnesium oxide and ammonia. Answer the following about this reaction.
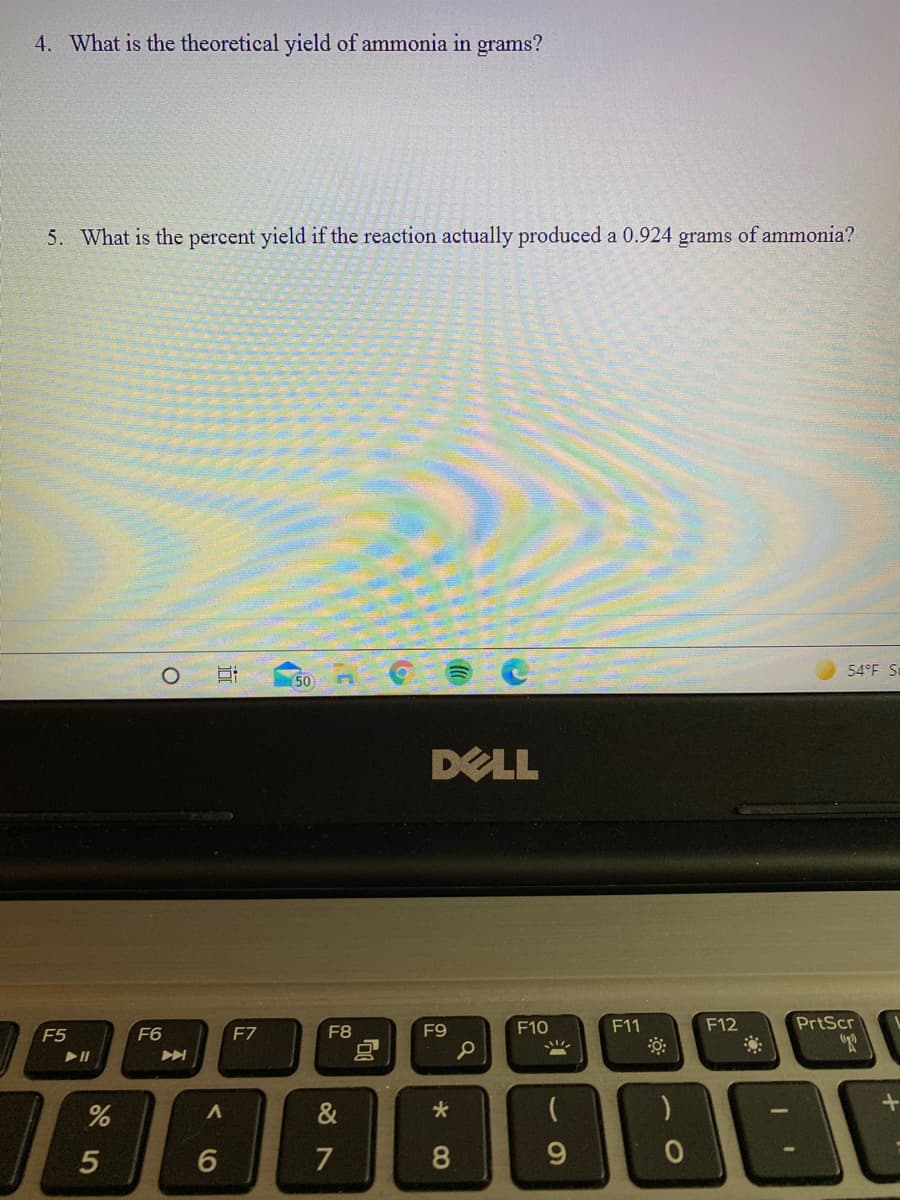
Transcribed Image Text:4. What is the theoretical yield of ammonia in grams?
5. What is the percent yield if the reaction actually produced a 0.924 grams of ammonia?
54°F S.
(50
DELL
F8
F9
F10
F11
F12
PrtScr
F5
F6
F7
7
9.
5

Transcribed Image Text:A chemist combines 3.12 grams of magnesium nitride with 1.770 grams of water which react to
produce magnesium oxide and ammonia.
1. Write the balanced molecular equation.
2. What is the formula weight of the magnesium nitride?
3. Which substance is the limiting reagent?
54°
50
DELL
F5
F6
F7
F8
F9
F10
F11
F12
PrtScr
&
5
7
9.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning