4. What reagents are required to complete the following reaction? as CI- 5. What is the expected product of the following reaction? KMnO4 OOH, hot 6. Predict the product(s) of the following reaction. tant OH 1.03 2. CH3SCH3 OH
4. What reagents are required to complete the following reaction? as CI- 5. What is the expected product of the following reaction? KMnO4 OOH, hot 6. Predict the product(s) of the following reaction. tant OH 1.03 2. CH3SCH3 OH
Chapter19: Aldehydes And Ketones: Nucleophilic Addition Reactions
Section19.9: Nucleophilic Addition Of Hydrazine: The Wolff–kishner Reaction
Problem 13P
Related questions
Question
please answer all for thumbs up
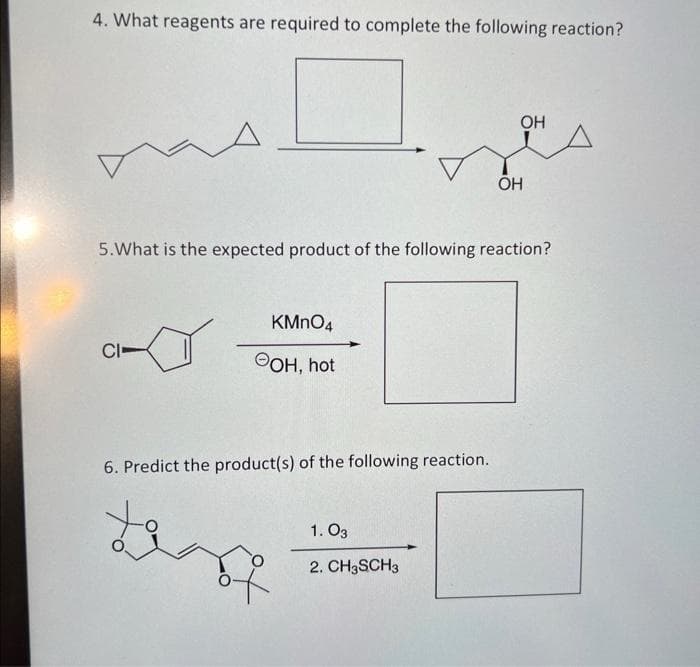
Transcribed Image Text:4. What reagents are required to complete the following reaction?
A
CI-
5. What is the expected product of the following reaction?
KMnO4
OH, hot
6. Predict the product(s) of the following reaction.
OH
1.03
2. CH3SCH3
OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning