Q: What is the total number of carbon atoms on the right-hand side of this chemical equation?…
A: To answer this question we need to count the total number of atoms of carbon (i.e.- "C") on the…
Q: Be sure to answer all parts. For the titration of 40.0 mL of 0.150 M acetic acid with 0.100 M sodium…
A:
Q: A student has a sample of 0.50 M lactic acid (HC 3H5O 3). She adds 10 mL of this solution to 40 mL…
A:
Q: Felix Hoffmann is most important for having synthesized: Select one: O a. Acetic Acid O b. Urea O c.…
A: The German chemist Felix Hoffmann famously synthesized two drugs: aspirin, one of the most widely…
Q: What is the equilibrium constant at 25°C for the following reaction? Sn2+ (aq) + Fe(s) = Sn(s) +…
A:
Q: CH₂ HCI 1) Li / éter 2) Cul 3) CH3CI H₂SO4
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: A. What is/are the major product(s) of the following reaction? B. A ΟΕ OF OH Br C. D. HO Br. 1 eq…
A: Ether reacts with HBr to form one molecule of alcohol and one molecule of Alkyl bromide. Usually…
Q: How many mL of a 0.200 M sulfuric acid solution will react with 3.50 g alun 2AI + 3H₂SO4 Al2(SO4)3 +…
A: The molarity of the sulfuric acid solution is 0.200 M.The mass of aluminum reacted is 3.50 g.The…
Q: The standard reduction potential o Fe +2 is 0.41 V o Ag is +0.80 V • Overall redox reaction is Feº +…
A:
Q: Determine the higher heating value of n-butane, according to its gas phase with and without…
A: The objective of the question is to calculate the higher heating value () for .
Q: Give detailed mechanism Solution with explanation needed..don't give Handwritten answer
A: A question based on reactions of alkene. The most appropriate reagent among the given reagent is to…
Q: Which reagent(s) could most efficiently bring about the following transformation ? 1º-alderre Rhas…
A: We have given the organic reaction and we have to find out the reagent for this transformation.
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. H3N. (BOC) 20 (CH3CH2)3N
A: Given is organic reaction. This is amine protection reaction.
Q: Acidity: Complete the statement about the acidity/basicity of compounds due to aromaticity from the…
A: We will first draw all possible resonating structures that include the given anion. The more the…
Q: Draw the structure of the major organic product(s) of the reaction. CH3 N CH3 1. LIAIH4, ether 2.…
A: LiAlH4 is a strong reducing agent capable of reducing aldehydes, esters, ketones, carboxylic acid…
Q: A OH I HCI I
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: b. Provide a complete arrow pushing mechanism for each reaction. [Hint: Pay attention to the product…
A: The two given reactions are examples of substitution reactions. Substitution reactions are of two…
Q: What is the total number of oxygen atoms on the right-hand side of this chemical equation? 8 Al(s) +…
A: To count the number of atoms in the chemical equation, the coefficient and number of the subscripts…
Q: 9.- It has been observed that a pure enantiomer has an optical rotation of -0.62⁰ measured in a 1 dm…
A: Chiral molecules are capable of rotating the plane polarized light. Specific rotation is identified…
Q: What was the major product of the reaction for compound 4 in the table? Did the product distribution…
A: Elimination and substitution often compete with each other. The same reactant can give elimination…
Q: 1. In this problem we consider a heat engine functioning via a four-step thermody cycle known as the…
A:
Q: please provide mechanism for the following reaction. Will like response! HO HO Scheme 5. G 26 (94%)…
A: Given reaction is an example of the oxidation reaction.In this reaction oxidation of secondary…
Q: Complete the following reaction and write the IUPAC names of the two products. "NH₂ + H₂O + HCI heat
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: 1. For the reaction provided, the rate of disappearance of I (aq) is 2.4 x 103 mol/L s. I03 (aq) +…
A: The rate of the formation of the product or the rate of disappearance of the reactants is known as…
Q: Draw the major organic product of the reaction shown. OH HI Select Draw Templates More / |||||| G e…
A: Due to the presence of weak nucleophile the reaction proceeds via SN1 mechanism with the formation…
Q: b). -C-CH₂ 1. xs LDA 2. CH₂CH₂CH 3.H₂O
A: The reactant belongs to a class of organic compounds called ketone. The name of the compound is…
Q: Ammonia (NH3) chemically reacts with oxygen gas (O2) to produce nitric oxide (NO) and water (H2O).…
A: The objective of this question is to find out the mass of oxygen gas that is consumed when 6.58 g of…
Q: At 45.0 °C, a 14.0 L vessel is filled with 7.25 moles of Gas A and 8.45 moles of Gas B. What is the…
A: From ideal gas law equation P•V = n•R•T P = pressure of gas V = volume R = gad constant T =…
Q: Which of the following nuclides is most likely to undergo beta decay? O 185 Re 75 O 187 Re 75
A: Beta decay is a nuclear decay process in which a neutron is transformed into a proton, accompanied…
Q: Draw the product of the reaction shown below at physiological pH (pH = 7.4). Ignore inorganic…
A: During the synthesis of peptides, sometimes it is necessary to protect the amino group or carboxylic…
Q: Macmillan A compound composed of 3.3% H, 19.3% C, and 77.4% O has a molar mass of approximately 60…
A: Molecular Formula: The molecular formula of a compound provides information about the actual number…
Q: Complete the electron-pushing mechanism for the given decarboxylation reaction. Add bonds,…
A: The term decarboxylation means removal of carboxylic acid group (-COOH) group from any reactant…
Q: A 7.92 mL sample of MgCl2 aqueous solution was diluted to a volume of 27.3 mL. If the chloride ion…
A: Given,volume of original MgCl2 aqueous solution = 7.92 mLVolume of MgCl2 solution after dilution =…
Q: Which is the weakest acid? A) O OH C) CH3COOH B) D) H₂SO4 SH
A: The strength of acids is typically determined by their ability to donate protons (H⁺ ions). In…
Q: What is the major product of the following reaction? дун он H 342 H 6) T چلو d) MCPBA? epoxic HH OH…
A:
Q: How many orbitals are allowed in a subshell if the secondary quantum nur S = 2 A) 3 B) 7 C) 1 D) 9…
A: Answer:There are total 4 quantum numbers:Principle quantum numberSecondary quantum numberMagnetic…
Q: 8.- Provide the absolute configuration of the following molecule. t N CI N H H HN S Cloxacillin…
A: Nomenclature of Configuration is basically based on CIP Rule .According to it, after giving priority…
Q: If Randy Santel's after-dinner mint contains 1092 Calories of energy, it also contains: a. 4568928 J…
A: Answer:This problem is based on conversion of units where given physical quantity needs to be…
Q: Some soluble compounds are listed in the table below. Classify each compound using the checkboxes.…
A: Types of compounds:1) Ionic compounds - The compounds which are formed by the combination of ions…
Q: Which of the following does NOT match a condition change to a resulting change in reaction rate? O…
A: Answer:Here:
Q: 2 To which family of organic compounds does the following lactone belong? A a) alcohol b)…
A: lactone is a class of cyclic organic esters
Q: At a certain temperature the reaction below has Kc = 1.38. If 4.00 mol each of carbon dioxide and…
A:
Q: "rms' The root mean square velocity, expressed u is for a collection of particles and is directly…
A: The formula given is .The root mean square velocity is .The ideal gas constant is .The temperature…
Q: Benzene is burned in oxygen. Write the balanced chemical question. Calculate the change in heat of…
A: Given ,Benzene is burned in oxygen.Objective :Write the balanced chemical equation.Calculate heat of…
Q: Which best represents the major organic product(s) of the reaction shown? (A) -Br Br (B) Brz hv Br…
A: the reactant is . It reacts with bromine in the presence of sunlight to give a brominated product.
Q: A 0.050 M solution of a weak acid, HA, has pH = 2.74. Determine the value of K₂.
A:
Q: Which reagent will produce the target product in the highest yield? Br A) H₂O B) NaOH 2- HO C) CH₂OH…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Which conditions will produce the target product in the highest yield? ? OH Y racemic mixture A) 1.…
A: Oxymercuration-demercuration is a organic reaction that can be used to convert an alkene into an…
Q: 3. How would you achieve the following transformations? AICI 3 COOH
A: Given is organic reaction. The given reactant is benzene.The given product is 2-phenylpropionic…
Q: A 124.0 gram sample of an unknown substance (MM = 189.50 g/mol) is cooled from 233.0 \deg C to 147.6…
A: Mass of the unknown substance= 124.0 g = m Heat capacity of liquid = 1.62 J/g・°CHeat capacity of the…
Nitesh
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

- Draw a stepwise mechanism for the following polymerization reaction.Propylene forms only oligomer under normal free radical conditions, yet the monomer forms high-molecular-weight polymer if it is polymerized while trapped in the channels of a crystalline host substance such as urea or thiourea. Explain [G. Di Silvestro, P. Sozzani, and M. Farina, Polym. Preprints, 27(1), 92, 1986].Although chain branching in radical polymerizations can occur by intermolecular H abstraction as shown in Mechanism 30.2, chain branching can also occur by intramolecular H abstraction to form branched polyethylene that contains butyl groups as branches.a. Draw a stepwise mechanism that illustrates which H must be intramolecularly abstracted to form butyl substituents.b. Suggest a reason why the abstraction of this H is more facile than the abstraction of other H’s.
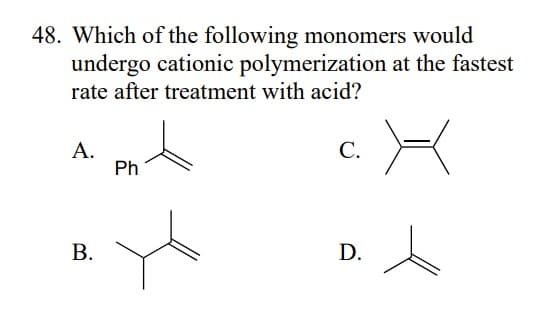
- Consider monomers A–C. (a) Rank the monomers in order of increasingreactivity in cationic polymerization. (b) Rank the monomers in order ofincreasing reactivity in anionic polymerization.Cationic polymerization of 3-phenylpropene (CH2=CHCH2Ph) affords Aas the major product rather than B. Draw a stepwise mechanism toaccount for this observation.Although chain branching in radical polymerizations can occur by intermolecular H abstraction as shown in Mechanism 28.2, chain branching can also occur by intramolecular H abstraction to form branched polyethylene that contains butyl groups as branches.a. Draw a stepwise mechanism that illustrates which H must be intramolecularly abstracted to form butyl substituents. b. Suggest a reason why the abstraction of this H is more facile than the abstraction of other H's.
- Rank the following monomers in order of increasing reactivity toward anionic polymerization (least reactive to most reactive).Poly(vinyl alcohol) is a polymer used to make fibers and adhesives. It is synthesized by hydrolysis or alcoholysis of the polymer obtained from polymerization of vinyl acetate as shown below. a. Why is poly(vinyl alcohol) not prepared by polymerizing vinyl alcohol? b. Is poly(vinyl acetate) a polyester?Rank the following monomers in order of increasing reactivity toward cationic polymerization (least reactive to most reactive).
- Draw the starting structure that would lead to this polymer under radical cond(a) Explain why poly(vinyl alcohol) cannot be prepared by the radical polymerization of vinyl alcohol (CH2=CHOH). (b) Devise a stepwise synthesis of poly(vinyl alcohol) from vinyl acetate (CH2=CHOCOCH3). (c) How can poly(vinyl alcohol) be converted to poly(vinyl butyral), a polymer used in windshield safety glass?(a) Show how poly(propylene oxide) can be synthesized via anionic ring-opening polymerization. (b) Draw the mechanism for the initiation and first two propagation steps.

