5- Vacuum fractional distillation of crude oil, when performed, allows the distillation of the most volatile parts without raising the temperature to the point where pyrolysis reactions occur. * Error O correct
5- Vacuum fractional distillation of crude oil, when performed, allows the distillation of the most volatile parts without raising the temperature to the point where pyrolysis reactions occur. * Error O correct
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter33: Automated Methods Of Analysis
Section: Chapter Questions
Problem 33.8QAP
Related questions
Question
Ans.
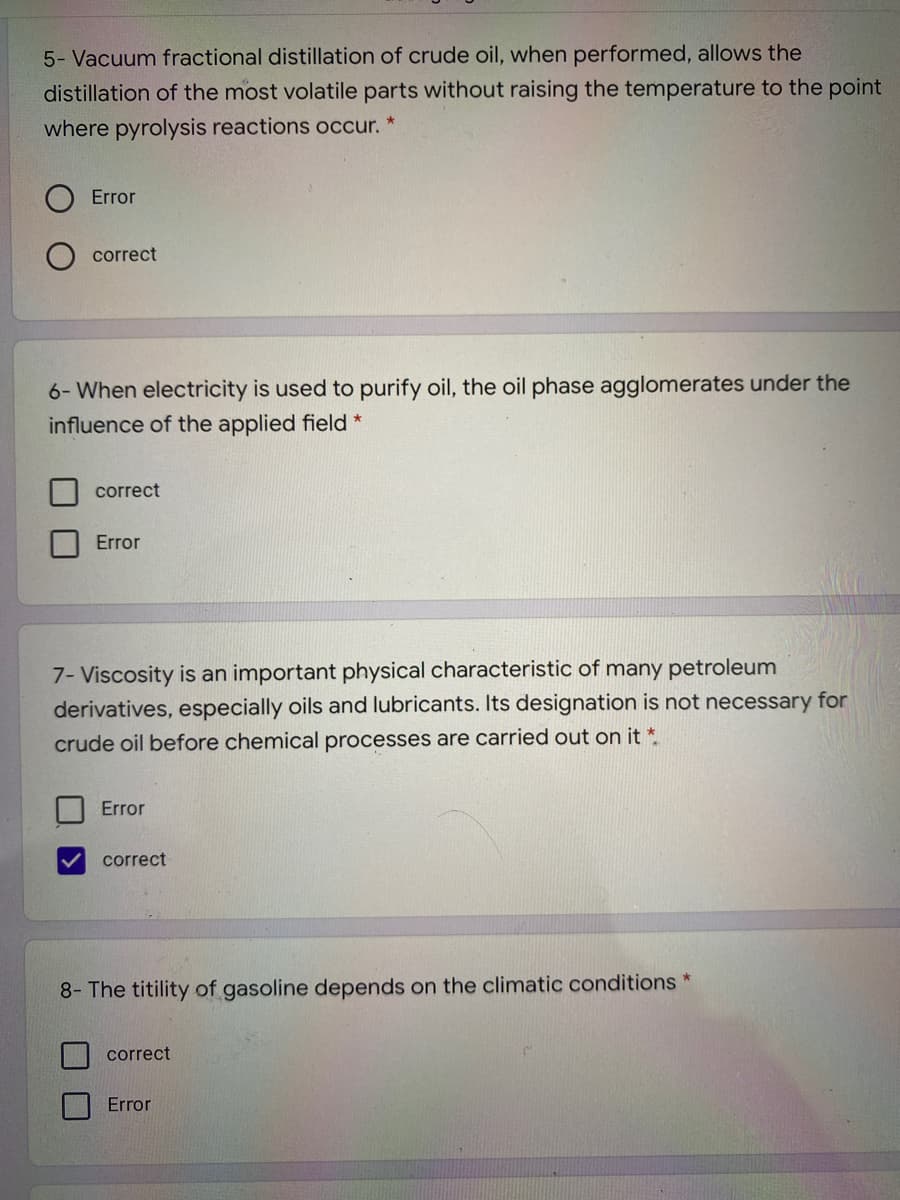
Transcribed Image Text:5- Vacuum fractional distillation of crude oil, when performed, allows the
distillation of the most volatile parts without raising the temperature to the point
where pyrolysis reactions occur. *
Error
correct
6- When electricity is used to purify oil, the oil phase agglomerates under the
influence of the applied field *
correct
Error
7- Viscosity is an important physical characteristic of many petroleum
derivatives, especially oils and lubricants. Its designation is not necessary for
crude oil before chemical processes are carried out on it *.
Error
correct
8- The titility of gasoline depends on the climatic conditions *
correct
Error
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
