Q: -CH3 HBr Br CH3
A: -> Conjugated Diene has nucleophilic character hence, it can give electrophilic addition…
Q: What type of holes do the blue spheres occupy in the ionic solid depicted below? Give evidence to…
A: When Coordination number surrounding a particular void is 4 that means 4 atoms surround that void,…
Q: 2. If the bromination of 4 mmol of 4-methylbenzoic acid with 4 mmol of N-bromosuccinimide resulted…
A:
Q: A voltaic cell is constructed from a standard Ni2+ Ni half cell (Eºred = -0.250V) and a standard…
A: To write a spontaneous cell reaction , first we will write anode and cathode half cell equations.…
Q: 11. Give the electron configurations (full & noble gas), and full orbital diagrams for: Germanium -…
A:
Q: [Cr³+] = 1.17 M and [Zn²+] = 0.00556 M, is -26.6 kJ: The free energy change for the following…
A:
Q: 15.6 cm? A) 2.5 .): What is the speed (f/number) of a lens with a diameter of 5.2 B) 3.0 C) 4.6 6 D)…
A: Answer :- Given - Focal length of lens = 15.6 cm Diameter of lens = 5.2 cm Determine the speed of…
Q: Radioactive decay I.Occurs in unstable isotopes II. is not spontaneous III. releases energy
A: Answer of the given question is shown below:
Q: frentel secotchy seo, Sodium is a softer than magnesium based on the Sea Electron Model. Justify…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 30 70 60 50 40 30 20 10 0 0 1 2 3 4 Time (Days) 6 7 8 9 How long is the half-life of the isotope…
A:
Q: a) b) A) (S)-3,4,4-trimethylpentanal B) (R)-3,4,4-trimethylpentanal C) (R)-…
A:
Q: (c) Draw the different products expected for the reaction below. NBS Light
A: NBS is N-bromosuccinimide it gives allylic bromination with alkene by free radical reaction…
Q: c) Use the VSEPR model to indicate the molecular geometry of the following ion and molecule. i) IC…
A: We have been given two molecules or ions and we have been asked their molecular geometry. In other…
Q: A) B) I OH OH 34 1. H-N 2. LIAIH4 3. H3O+ KCN LOH or CN
A:
Q: Give the structure of the final organic product (after the two reaction sequence): I C III Select…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Calculate the pH of a solution that has a hydroxide ion concentration, [OH-], of 8.52 x 10-6 M. pH =
A:
Q: A voltaic cell is constructed in which the following cell reaction occurs. The half-cell…
A: Please repost the second question. As per the guidelines, I am answering the first question.…
Q: 2. Let's look at the electron-ionization Rel. Intensity 100 80 60 40 20 0.0 10 mass spectra of…
A:
Q: The pH of a basic solution is 12.29. What is [OH⁻]?
A:
Q: A sample of nitrogen gas occupies 10.0 liters at 25 Celsius and 98.7 kPa. What would be the volume…
A: According to combined gas law, the the ratio of product of pressure and volume with that of…
Q: 6. Starting from the alkyne shown below, show all reagents and intermediates necessary to carry out…
A: The reaction is completed in two steps. In the first step alkylation of the alkyne takes place and…
Q: Consider the following concentration-time data for the decomposition reaction AB→A+B. Time (min) 0…
A:
Q: The chemical formulae of some acids are listed in the first column of the table below, and in the…
A:
Q: For a particular reaction, AH Calculate AG for this reaction at 298 K. AG= Incorrect - -14.20 kJ and…
A:
Q: 1. Aspirin is hydrolyzed in the human body by an enzyme known as an esterase. Write the overall…
A: The enzymatic hydrolysis of aspirin (2- acetoxybenzoic acid) gives a mixture of 2- hydroxybenzoic…
Q: For the following question, consider the following balanced equation. Mg3 N₂ (s) + 6 H₂O(l) → 3…
A:
Q: A voltaic cell is constructed from a standard Cl₂|CI¯ half cell (Eºred = 1.360V) and a standard Mg2+…
A: Less the reduction potential, less reduced the cell is . More the reduction potential, more…
Q: The normal boiling point of a certain liquid X is 136.50 °C, but when 20. g of iron(III) nitrate…
A: Firta we need to calculate elevation in boiling point and van't Hoff factor. Then we would calculate…
Q: Consider the following mechanism for the decomposition of ozone: O(g) → O₂(g) + O(g) O(g) + 0(g) →…
A:
Q: Consider the following balanced chemical reaction: N2 (g) + 3 H2 (g) → 2 NH3 (g) What is the…
A: Given: Mass of N2 = 9.36 g Mass of H2 = 4.92 g Known: Molar mass of N2 = 28 g/mol Molar mass of H2 =…
Q: ): A compound had a molar absorptivity of 2.93 x 10' L.cm mol¹. What concentration of the compound…
A: It is an application of Lambert Beer law Here we are required to find the concentration of the…
Q: b. C. d c f. 1. Give the major products of the following reactions, inxlude stereochemistry, H₂C H…
A:
Q: (i) (ii) 2. Write the complete stepwise mechanism for this reaction. Show all electron flow with…
A:
Q: Be sure to answer all parts. Draw Lewis structures for the following molecules. Be sure to minimize…
A: Lewis Structure is a representation of the valence shell electrons in a molecule. It is used to show…
Q: Identify each of the following substances as an electrolyte or a nonelectrolyte and predict which…
A: A substance is an electrolyte when it dissociate into ions when dissolved in water and these ions…
Q: НО NH₂ НО OCH3 хххх B НО A CN с НО D CH3
A: Hemiacetal carbon contain a OH ,OR,H ,and a Alkyl group attached with it
Q: OK O Phenol
A:
Q: barium hydroxide solution is prepared by dissolving 1.44 g of Ba(OH)₂ in water to make 52.2 mL of…
A: Mass of Ba(OH)2 = 1.44 g
Q: A 1.0 L sample of water has a BOD of 2.10 mg L-1. How much organic material (with an average CH₂O…
A:
Q: The enthalpy of vaporization for methanol is 35.2 kJ/mol. Methanol has a vapor pressure of 1 atm at…
A: It is given that, ∆vapH° of methanol = 35.2 kJ/mol = 35.2 × 103 J/mol Vapour pressure P1 = 1 atm…
Q: Which of these triglycerides would you expect to find in higher amounts in oil? In fats? Justify…
A: Given Triglycerides are A and B
Q: A 5.00-L cylinder initially contains 0.200 mol Br2 and 2.00 mol F2 at 275 K. Assuming 100% yield of…
A: The unbalanced chemical equation is Br2 + F2 → BrF5.
Q: 1. Identify the type or types of intermolecular forces (London, Dipole dipole, H-bond) present in…
A: We have to tell the intermolecular forces (London, Dipole-dipole, H-bond) present in each substance.…
Q: Identify the spectator ion(s) in the following reaction. L(OH)2(s) + 2T*(aq) + 2OH(aq) --> 2T…
A: In this question, we will identify the spectator ion(s) for the given reaction equation. spectator…
Q: This is a Dry Lab since we cannot meet on Campus. After calibrating a pH meter you would have…
A: Given, Kw=1.0×10-14
Q: You prepared an exact saturated solution of K₂Cr₂O7 at 70°C in 1 kg of water. You cool the water to…
A:
Q: Chromium(VI) is a widely used form of chromium that is also highly toxic (see the movie Erin…
A: To determine the concentration of Cr(VI) present in the unknown sample given that when a 20.00 mL…
Q: A compound has the chemical formula C5H11Cl. The M+ peak should be at m/z = m/z= and an M+2 peak at…
A: The molecular weight of C5H11Cl = (5xC) + (11xH) + (1xCl) = (5x12)+(11x1)+(1x35.5)= 106.5 g.
Q: Draw structures for a) (2E,4Z) - hepta-2,4-diene b) (S) - 2-bromobutane
A: R configuration - rotate clockwise S configuration - rotate anticlockwise E= trans isomer z= cis…
Q: 4. Calculate the enthalpy change upon converting 2.00 mol of ice at -20 °C to steam at 130 °C under…
A:
Step by step
Solved in 2 steps with 2 images

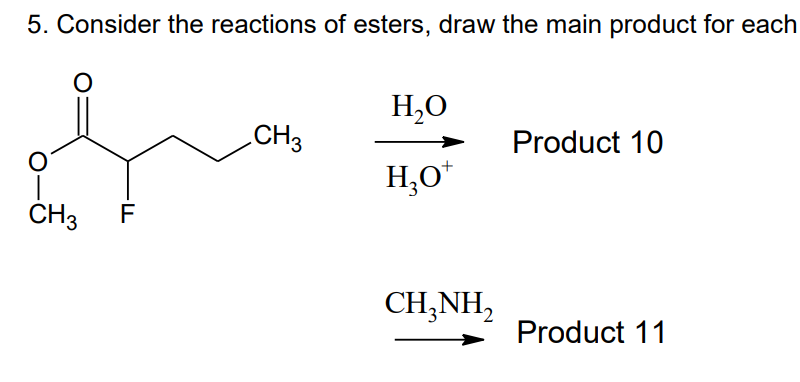
- 2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 3Which of these reagent(s) will not react with HOCH2CH2CH2COOH? A) NaCN in ethanol B) C2H5OH in the presence of an acid catalyst. C) (CH3CO)2O D) Concentrated H2SO4Observe the REACTION in the diagram below to answer questions 1. What is the type of hybridization of the carboxyl carbon in the mechanism? a. sp1.5b. sp3c. sp2d. sp 2. What is the IUPAC name of the product? a. Pentanoyl chlorideb. Pentanone chloridec. Pentanoic acid chlorided. Pentanyl chloride 3. The illustrated reaction mechanism is considered __________. a. Concertedb. Continuousc. Step-wised. Unfavorable 4. Which atom in the reaction mechanism is the nucleophile? a. Chloride anionb. Hydroxyl groupc. Methyl groupd. Carboxyl carbon 5. What type of reaction is illustrated in the reaction mechanism? a. Electrophilic substitution reactionb. Nucleophilic acyl substitution reactionc. Electrophilic acyl substitution reactiond. Nucleophilic addition reaction 6. What is the main functional group of the organic molecule? a. Alcohol functional groupb. Carboxyl functional groupc. Carbonyl functional groupd. Aldehyde functional group 7. Aside from the organic molecule, what product is…
- I recovered 3 mL of water, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.dedcue the major organic product from the following reations. be ablw to draw hydration halogenation hydrohalogenation, halohydrin formation, oxymercuratiom demercuration qnd hydroboration oxidation.Draw themajor product of this reaction. Ignore inorganic byproducts. PPC, CH2Cl2
- Draw the structure of the predominant form of CF3CH2OH (pK a = 12.4) at pH = 6.What are the major differences in the conservationof energy by methanogens that only use H2 1 CO2versus those that can use methyl compounds?(please answer all questions) 1) Predict the products from reaction of 1-hexyne with thesereagents: a) One equivalent HBr b) One equivalent Cl2 c) H2 , Lindar Catalyst d) NaNH2 in NH3 , then CH3Br
- Draw the structure of the major product of 3-hexyne + 1 equiv HBr e. 2-pentyne + acidic KMnO4the product is given predict from what alkene reactant be made from, write the complete reaction on a sheet of paper A.oxidation of 2-methyl-3-pentanol B.aldol condensation of CH3CH(Br)CHO A.Draw the major product of this reaction. Ignore inorganic byproducts. PCC, CH2Cl2



