Q: 3. What is the pH of a 1.15 M solution of methanoic acid? K = 1.8 x 104
A:
Q: 11. Which of these molecules are meso isomers? H Cl Cl I (A) I only (C) III only H/Cl CI II H H CI…
A: A meso compound is a stereoisomer with two or more chiral centers but no optical activity due to…
Q: Why SnI4 formed under AcOH/ Ac2O condition and SnI2 formed under 2M HCl and nitrogen flow? Please be…
A: The formation of SnI4 or SnI2 depends on the reaction conditions and the oxidation state of tin…
Q: what is the mole fraction of 4.31 m KBr (aq)?
A:
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A:
Q: A sample of unknown identity was analyzed by thin-layer chromatography (TLC) using acetic acid/ethyl…
A: Find the unknown compound based of Rf value from the data.
Q: 3. Circle the compound that matches the spectrum provided. Note that the peak at 1.5 ppm exchanges…
A: NMR spectroscopy is an analytical technique for structure elucidation of a compound. 1H NMR is used…
Q: Calculate the solubility at 25 °C of PbCO3 in pure water and in a 0.0120M Pb(NO3), solution. You'll…
A: Molar solubility of a substance is defined as the number of moles of compound that dissolve to give…
Q: SQ-5. Chlorine occurs naturally as a mixture of two isotopes with atomic masses of 34.97 amu and…
A:
Q: A Lewis structure is a two-dimensional representation of a molecule that does not necessarily show…
A: Bond pairs lone pairs shape of the molecule 10linear20linear22bent30trigonal planar31trigonal…
Q: Which of the following could enter into H-bonding interactions with water? (Select all that apply.)…
A:
Q: Question Calculate the relative amounts of acetic acid and acetate ion present at the following…
A: Answer:- This question is answered by using the simple concept of titration to weak acid against the…
Q: What type of isomer (C5H10) is pentene ? Solve correctly please asap,with detailed explaination.
A: Isomers are those which have the same molecular formula but different structural formula. They are…
Q: The following table gives the volume of nitrogen (reduced to 0°C and 1 bar) adsorbed per gran of…
A: The given table of volume of nitrogen (reduced to 0oC and 1 bar) absorbed per gram of active carbon…
Q: what is the definiton of diffusion? a) gas molecules mix unequally. b) average distance between…
A: Diffusion is the movement of molecules in fluid from areas of high concentration to areas of low…
Q: -3 The densities of pure water and ethanol are 997 and 789 kg. m respectively. The partial molar…
A: To calculate the change in volume relative to the pure components when preparing a solution with a…
Q: Consider the following equilibrium: Hind + H₂O H3O+¹ + Ind-¹ At the transition point for an…
A: At transition point, solution changes colour. At transition point, [HIn] = [In-] HIn = indicator ,…
Q: Ammonia is synthesized from nitrogen gas and hydrogen gas at 400 degrees Celsius and 350 kPa. If…
A: Temperature (T) = 400°CPressure (P) = 350kPaVolume of N2 = 30.5 L-> Hydrogen is in excess.
Q: Ⓒ Macmillan Learning Identify the molecular shape of each of the molecules. Molecule SICI SF *+…
A: The arrangement of atoms in a molecule in three dimensions is known as molecular geometry, commonly…
Q: 1. Is the molecule shown below chiral or achiral? ОН ОН
A: A question based on introduction to organic chemistry. An organic structure is given whose chirality…
Q: 3d) Circle all molecules from the set below that can act as H-bond donors. i) ww H N iv) H NEC-H 4a)…
A: Two questions based on H-bonding and molecules. First question is to be answered based on the…
Q: 18000 10000 14000- 12000 10000 8 8000. 6000- 4000- 2000- 0 0 0.5 Component Acetone 0.82 Isoborneol…
A: Retention time is defined as the time that a solute spends in a column or the time spent in the…
Q: 8. For the reaction 2H₂O(1) + 2e → H₂(g) + 2OH(aq), calculate the volume of "dry" hydrogen gas…
A:
Q: PROBLEM 1-9 Draw the important resonance forms of the following cations and anions: (a) (b) (c) (e)…
A:
Q: Which reaction has the largest positive entropy change per mole of product formed? (A)S(s) + 3F2(g)…
A: We have find out the answer.Note~ Since you have posted multiple questions, we will provide the…
Q: These are actually diastereomers .
A: The given compounds are shown below.We have to assign the relationship between these two compounds.
Q: A 3D representation of a cyclohexane (C6H12) molecule, a cyclic compound used in the manufacture of…
A:
Q: 2. What is the oxidation number of N in HNO3? a. +6 b. -6 C. -5 d. +5
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Using pKa values, determine whether or not the following reaction will proceed to completion and…
A: The tendency of proton donation can be considered as acidic strength.Lower the pKa, stronger the…
Q: Question 15 Which of the following compounds has the lowest boiling point? 2-chloro-2-methyl propane…
A:
Q: A chemist dissolves 575. mg of pure hydrochloric acid in enough water to make up 380. mL of…
A:
Q: Draw a structural formula for butanoic acid.
A: To draw the structural formula -Determine the number of carbon atomsIdentify the functional group…
Q: Part C. The constitution of ectocarpene a volatile, sperm-cell-attracting material released by the…
A: Condensed formula of ectocarpene Structural formula of ectocarpene ?
Q: ered swer According to MO theory, overlap of two p atomic orbitals produces two л MOs, two л* MOs,…
A:
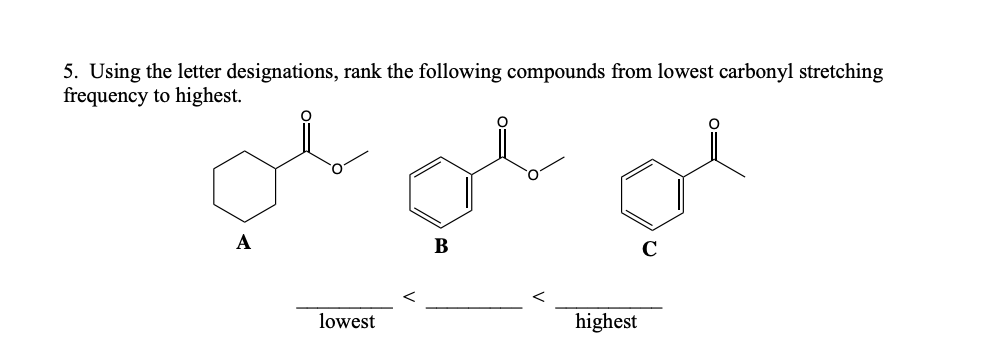
Q: 5. Using the letter designations, rank the following compounds from lowest carbonyl stretching…
A: Given some carbonyl compounds.
Q: 5 H3C. these two methyl groups are not part of the main C chain and can be consider functional…
A: A functional group is a substituent responsible for distinctive chemical properties and chemical…
Q: ion 16 of 22 > O Macmillan Learning Organic Chemistry Maxwell presented by Macmillan Learning Select…
A: The question is based on the concept of stereochemistry. we need to identify the relation between…
Q: Show that for a first-order reaction R → P, the concentration of product can be represented as a…
A: To Show that for a first-order reaction RPthe concentration of product can be represented as a…
Q: What is the volume of 10.0 g of argon gas at 157 degree celsius and 2.50 kPa pressure with the…
A: Mass of Ar = 10gTemperature = 157°C = 430.15 KCompressibility factor = 0.975Pressure = 2.50 kPaMolar…
Q: One gram of activated charcoal has a surface area of 1000 m². If complete surface coverage is…
A:
Q: Suppose a 0.36 M aqueous solution of oxalic acid (H₂C₂O₂) is prepared. Calculate the equilibrium…
A: 0.36 M Oxalic acid, H2C2O4(aq) is a diprotic acid with pKa values of pKa1 = 1.25Ka1 = antilog…
Q: Methyl acetate is hydrolyzed in approximately 1M HCI at 25°C. Aliquots of equal volume are removed…
A:
Q: Complete the following reaction by writing the name of the product. ch3coh + H2 ?
A: The reaction of two compound give a product which is chemically and physically different from the…
Q: Classify the mechanism as a substitution, elimination, or addition reaction. -Y CI The mechanism is:…
A: elimination reactionElimination reaction is a type of reaction that is mainly used to transform…
Q: What is the pH of a 0.340 mol/L solution of sodium fluoride? (K₁ HF = 7.2 x 10-4) pH=8.34
A:
Q: K MX
A:
Q: If you were trying to prepare a buffer with ammonia, NH3, what would be the substance that you would…
A: 1. Buffer solution are those whose pH value doesn't chage appricableby addition of small of acid…
Q: Question 9 Consider the ranked polarity of all fictitious compounds with different functional groups…
A: Thin-layer chromatography has two phases a stationary phase and a mobile phase. The silica gel…
Q: 2. a. Considering the five compounds shown below, which one matches the given ¹H NMR spectrum?…
A: Since you have asked a question with multiple subparts, we will answer only first three subparts. In…
Q: 12B.1 (a) Write down the expression for the partition func- tion of a molecule that has three energy…
A: (a) Write down the expression for the partition function of a molecule that has three energy levels…
NEED HELP NOT GRADED PRACTICE
Step by step
Solved in 3 steps

- Please explain it. For each stereoisomer, draw the interconversion of chairs and identify the most stable. Give handwritten answer. ASAPExplai all steps to Determine the Directing Effects of a Particular Substituent ?Can someone please label the H1, H2 etc into its respective regions (ie. aliphatic and aromatic regions)? Thank you!
- Hello, thank you this was very helpful. However, i still dont completly understnad how to determin and explain why the isomer of B is the major product. Could you explain this in a differnt way?Would it be A or D? I’m confused.Chemistry draw appropriate curved arrows and transition state! thank you!!
- Bromide Bhas normal activity (for a secondary bromide) towards SN1 substitution, but A has much higher reactivity and Chas much lower reactivity.What is the correct witig product?Draw each unique stereoisomer of this week's product 2,3-do-vroom-3-phenylpropanoic acid in the cases of syn-or anti-addition, and ??????? the reference melting point for each.

