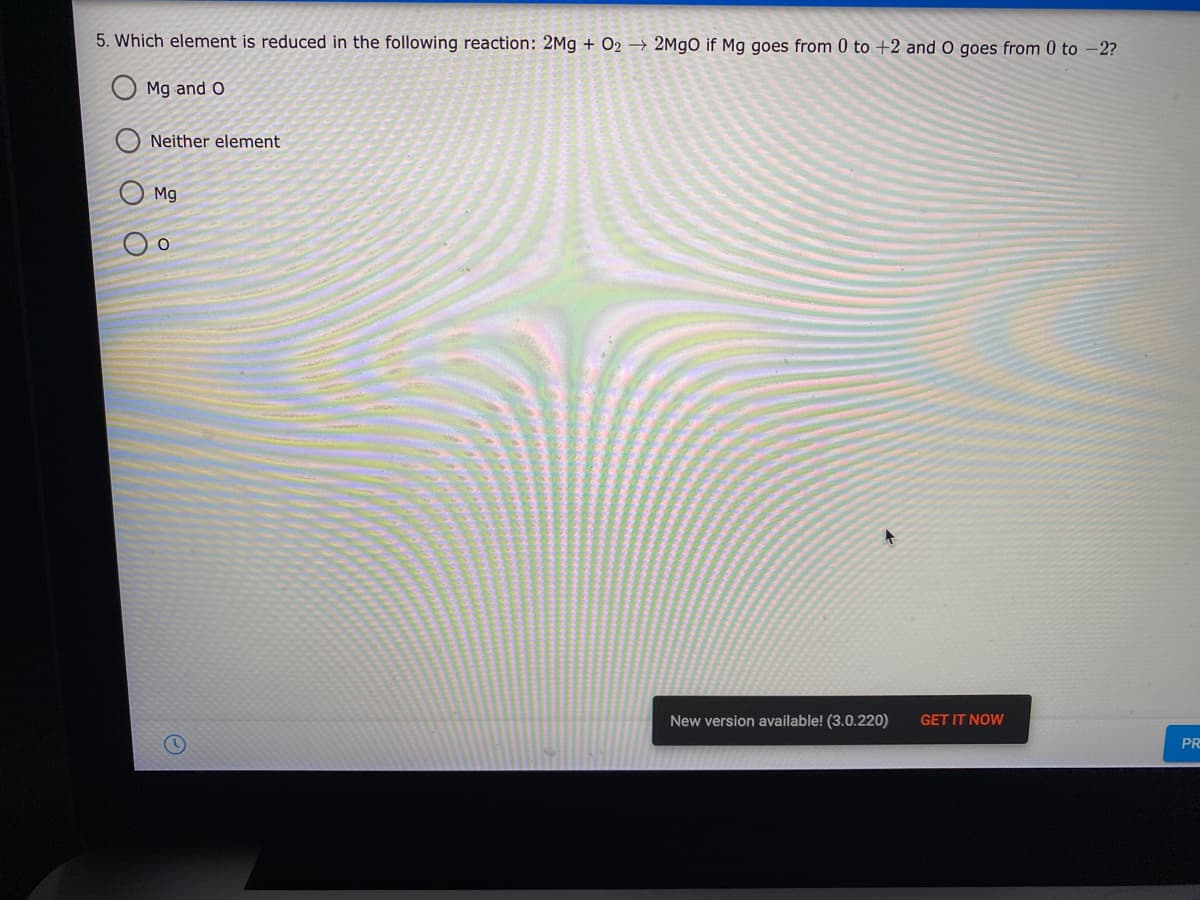
5. Which element is reduced in the following reaction: 2Mg + O2 → 2M9O if Mg goes from 0 to +2 and O goes from 0 to -2? O Mg and O Neither element O Mg
Q: Condifer the reaction below, which element is being reduced? Pb(NO3)2 (aq) + Fe(s) --> Fe(NO3)2…
A: Oxidation - increase in Oxidation state. Reduction - decrease in Oxidation state.
Q: high reatii Fest 4. From the data in Table B, rai high reactiity e tates valenee 5. For each of the…
A: The substance which oxidation state increases in a chemical reaction is called as oxidation and…
Q: Which substance is oxidized in the following redox reac:on? 2H2S + O2 ------------→ 2H2O + 2S a.…
A: Chemistry deals with varieties of matter and change of onekind of matter into the other.…
Q: For each of the following reactions: a. Determine whether the reaction is a REDOX reaction b. If…
A:
Q: For reaction below, identify the atom oxidized, the atom reduced. 3H2C2O4 + 2K2MNO4 ===> 6CO2 + 2K2O…
A:
Q: 5 In the following reaction which element got oxidized : 3HNO3(aq) + Al(s) → Al3+ + 3NO2(g) +…
A: Since you have asked multiple questions, we will solve first 3 questions for you. For remaining…
Q: Ca Na Al Mn Fe Cd Pb Cu Ag The spectator ion in the reaction shown below is and aluminum (will/will…
A: Spectator ion are those ions which are canceled out in net ionic equation, I.e which are not…
Q: Which substance is the oxidizing agent in this reaction? 2CuO + C 2Cu + CO2
A: Answer: Redox word is the combination of two words, reduction and oxidation. So, when in a chemical…
Q: 3Cl- + 2Cr(OH)33ClO- + 2Cr+ 3H2O In the above redox reaction, use oxidation numbers to identify…
A: In redox reaction oxidation and reduction takes place simultaneously,
Q: Consider the reaction below. Choose the statement that best describes this reaction. Fe203 (s) + 3…
A: Redox reaction:Redox reaction is a type of chemical reaction in which the oxidation states of atoms…
Q: Hi, Which substance is oxidized in each reaction? CH4(g) + 2O2(g) -----> CO2(g) + 2H2O(g)…
A:
Q: I'm not completely sure on how to do this question as a total
A: Oxidizing agent is a substance that can oxidize other substance and gets reduce itself. Reducing…
Q: Which substance contains the element that is reduced in the following reaction: Al + Fe2O3 --> Al2O3…
A: The reaction given is 2 Al + Fe2O3 ---------> Al2O3 + 2 Fe
Q: What element is being oxidized in the following redox reaction? C10H22O2(aq) + KMnO4(aq)…
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and…
Q: to the given reaction 4 Ag + 4 Cl- → 4 AgCl + 4 e- by ....... A. Pt cathode B. Ag cathode C. Ag…
A: In Electrochemical cell in which anode undergoes oxidation ( loss of electron) and cathode undergoes…
Q: 31. Consider the reaction, 2 Al +3 Cu²* → 2 Al* + 3 Cu. The oxidizing agent is, A. Al B. Cu?+ C.…
A:
Q: What element is reduced in the following chemical reaction? Cu + 2H2SO4 → CUSO4 + SO2 + 2H2O Он O Cu…
A:
Q: For the following equation, which atom goes though the reduction? 2 F e ( s ) + 3 C l 2 ( g ) → 2 F…
A:
Q: For the reaction below, identify the statement that is false. Mgs) + H2SO4(ag) →MgSO4(ag) + H2lg)…
A: Oxidised which loose electron Reduce which gain for electron Redox reaction in which oxidation and…
Q: 3. a loss of oxygen during the process is called oxidation process A. True B.False
A: To find: Whether the given statement is true or false
Q: Consider the decomposition of red calx of mercury (HgO) into its elements as follows: 2 Hgo --> 2 Hg…
A: Molar mass of HgO is 216.59 g/mol Molar mass of O2 is 32 g/mol
Q: reactants reactants reduced reaction oxidized 31, () + 2Fe(s) - 2Fel, (s) 0,) + 2Mg (s) - 2M O (s)…
A: An increase in the oxidation number means the oxidation and decrease in oxidation number means…
Q: Of the following metals, which ions are most easily reduced? c. aluminum 17. a. iron b. mercury d.…
A: The approach for this question is based on reactivity series or electrochemical series. The order of…
Q: 2- If a piece of iron were to rust for 4 years, how much H2O did it produce overtime? Please use: Fe…
A: when iron reacts with oxygen and water, iron oxide is fomed. This iron oxide is commonly known as…
Q: An equilibrium is established according to the following equationHg2 2+(aq) + NO3 −(aq) + 3H+(aq) ⇌…
A: Redox reaction A reaction that involves oxidation and reduction simultaneously is called redox…
Q: Consider each of the three reactions below. If the reaction is a redox reaction, select which…
A: Answer 1 : The balance chemical reaction : 4Na(s) + O2(g) ---> 2Na2O(s) Reaction is redox…
Q: Identify the substance being oxidized in the following reaction: CH4 + 202 → CO₂ + 2H₂O Select one:…
A:
Q: Find the oxidizing agent in the following balanced reaction (Please note that element X and Y are…
A: Detail description is given below
Q: Part A Permanganate reacts with methanol to produce formic acid according to the following…
A:
Q: How many electrons are transferred? PbO + CO → Pb + CO2
A:
Q: 9) How many electrons are transferred in the following reaction? (The reaction is unbalanced.) 9)…
A:
Q: Identify the oxidizing agent and draw tie-lines to determine the reducing and oxidizing agents for…
A: Oxidizing agent-an oxidizing agent is a substance that has the ability to oxidize other substances…
Q: Which element is reduced in this reaction? 2Cr(OH)3 + 3OCI + 40H¯→2CrO4-2 +3C1 + 5H20 Enter the…
A: The answer to the following question is-
Q: Identify which substance is oxidized and which is reduced in the following reaction. 2S02 (g) + O2…
A: We have to tell about oxidised and reduced species
Q: In the following reaction, [ Select ] is being oxidized and [ Select ] is being reduced. Note:…
A:
Q: Find the oxidation number for each element in the following substance. UO22+ 1. U = 2. O =
A:
Q: How many electrons are transferred in the following reaction? (The reaction is unbalanced.) Mg(s) +…
A: The given reaction is represented as follows:
Q: How many electrons are transferred in the following unbalanced reaction? I2(s) + Fe(s) → Fe²+ (aq) +…
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and…
Q: Find the oxidizing agent in the following balanced reaction (Please note that element X and Y are…
A: Detail calculation is shown below
Q: Consider each of the three reactions below. If the reaction is a redox reaction, select which…
A:
Q: In the following oxidation-reduction reaction, what is being oxidized? C4H8 + 6 O2 → 4 CO2 + 4 H20 O…
A: Oxidation is a chemical process or reaction in which electrons will be ejected from a species.…
Q: Which substance is oxidized in each reaction? a) 2Zn(s) + O2(g) = 2ZnO(g) b) CH4(g) + 2 O2(g) =…
A: The substance whose oxidation state is increasing in the reaction will be getting oxidised a) In…
Q: For each reaction below, identify the element oxidized, the element reduced, the oxidizing agent,…
A: Oxidation : The increase in oxidation state is called oxidation. Reduction : The decrease in…
Q: __________ is an oxidation reaction. Group of answer choices Table salt dissolving in water for…
A: Rusting of iron is a redox reaction. During the process of rusting, iron metal combines with the…
Q: For each reaction below, identify the element oxidized, the element reduced, the oxidizing agent,…
A: The answer is given as follows
Q: When molten lithium chloride, LiCl, is electrolyzed, lithium metal is liberated at the cathode. How…
A: Welcome to bartleby ! We have to calculate weight of lithium
Q: 1. Find the coefficient for the balanced reaction for Iron and the type of reaction Pb +…
A: Number placed in front of a formula to balance a reaction is called the coefficient for the balanced…
Q: In an electrochemical decomposition apparatus constructed with a magnesium chloride (MgCl2) cell and…
A: Mg2+ +2e -> Mg K+ + e -> K Molar mass of Mg = 24 g/mol
Q: Determine the oxidization states of each element for both the reactants and the products in the…
A: Given: Cr2O72- (aq) + 6I- (aq) + 14H+ (aq) --> 2Cr3+ (aq) + 3I2 (l) + 7H2O (l) Oxidation means…
Q: 5. For the following reaction which reactant is getting oxidized and which is getting reduced:…
A: Species for which oxidation state is increased is getting oxidised while species for which oxidation…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- 18.2 Balance the following chemical equation for the stoichiometric combustion of methane with oxygen. ____ CH4(g) + ______O2(g) ⟶ ______ CO2(g)+ ______H2O(g) What are the coefficients for O2(g) and CO2 (g)?For the following reaction: 5Ca + V2O5 --> 5CaO + 2V In one process 5 moñ of V2O5 react with 3 mol of acá. Calculate the theoretical yield of V in the unit of mol.How much heat (in kilojoules) is evolved or absorbed in the reaction of 1.65 gg of NaNa with H2OH2O?2Na(s)+2H2O(l)→2NaOH(aq)+H2(g) ΔH∘2Na(�)+2H2O(�)→2NaOH(��)+H2(�) Δ�∘ = -368.4kJkJ.
- Background-info: https://drive.google.com/file/d/1G7sPTuESIgWk9wpRdqAnFcsBmOlbBfLv/view?usp=sharing Question: A student measured qnxn for the reaction of 0.398 g of a metal, Xa(molar mass = 97.42), with HClag to be -902 J. Assuming this metal reacts in the same manner as magnesium, what is the AH°; (in kJ/mol) for this metal's cation in solution, i.e., X2* (og? Report your answer to the correct number of significant figures and only include the numerical answer (no units).Give only typing answer with explanation and conclusion to all parts CH4 + 2O2 ------- CO2 + 2H2O + 890.1 kj What is theoretical yield of CO2 in moles using 1.5 moles of O2? What is theoretical yield of H2O in grams using 1.5 moles of CH4? How much heat is released by burning 120 g of O2?When the reaction shown below is performed using: A + B --> C + D The temperature changes from 25.0 degrees Celsius at the start to 19.0 degrees Celsius at the end of the reaction. a. Is the reaction endothermic or exothermic? b. What is the value of q for the reaction if the mass of the reaction was 54.4 grams ?
- Balance the reaction for the combustion of pentane: ?C5H12+?O2→?CO2+?H2O?C5H12+?O2→?CO2+?H2O Enter the four coefficients in order, separated by commas (e.g., 1,2,3,4), where 1 indicates the absence of a coefficient.1. Balance the following chemical equation: Fe2O3 + CO ------> Fe + CO2, answers are listed in order - 1st coefficient, 2nd coeficient, etc. and so on. (1st one is in front of Fe2O3, 2nd one in front of CO, etc. and so on) Group of answer choices a) 1,3,3,2 b) 2,3,2,3 c) 1,3,2,3 d) 1,2,2,2Stoichiometric calculations. Show computations K, a pharmaceutical scientist aims to synthesize paracetamol by reacting 3.075 mg of p-aminophenol and 2.25 milliliters of acetic anhydride to produce paracetamol and acetic acid. C6H7NO + C4H6O3 à C8H9NO2 What is the limiting reactant? How many grams of paracetamol was formed? K was able to produce 1.88 grams of paracetamol. What is the percentage yield?
- Deterioration of buildings, bridges, and other structures through the rusting of iron costs millions of dollars every day. Although the actual process also requires water, a simplified equation (with rust shown as Fe2O3) is:4 Fe(s) + 3 O2(g) → 2 Fe2O3(s) ΔHrxn = −1.65 × 103 kJ (a) What is the ΔHrxn when 0.250 kg of iron rusts?Deterioration of buildings, bridges, and other structures through the rusting of iron costs millions of dollars every day. Although the actual process also requires water, a simplified equation (with rust shown as Fe2O3) is: 4 Fe(s) + 3 O2(g) → 2 Fe2O3(s) Δ Hrxn = −1.65 × 103 kJ (a) What is the ΔHrxn when 0.250 kg of iron rusts?The enthalpy change of neutralization is -55.8 kJ/mol for a strong base strong acid reaction generating water. If you have 0.5 mol of H2SO4 and 0.5 mol of NaOH, what is the balanced chemical reaction, and calculate the limiting reagent for the reaction?

