5.3 buffer -pH 3.3 buffer pH 4.3 buffer Asp Thr Glu Ser Ala His Met lle Тyr Gly Phe Lys Leu NH, Val, Arg Pro 40 80 120 160 200 240 280 320 330 370 410 450 490 550 590 630 Effluent (mL) Absorbance |||| | |
Q: 220 mL of 0.03 M NaOH were added to 200 mL of 0.03 M benzoic acid (pKa = 4.2). What is the pH of the…
A: When NaOH is added to benzoic acid, sodium benzoate will form as per the chemical reaction:…
Q: From their Kb values, which of the following bases would be best for preparing a buffer of pH 9.00:…
A: Basic buffer is a solution of weak base and its salt. Salt is formed by reaction of weak base and…
Q: In the laboratory, a general chemistry student measured the pH of a 0.546 M aqueous solution of…
A: Since you have posted multiple questions which are not sub-parts, we are entitled to answer the…
Q: Part A A beaker with 1.40x10? mL of an acetic acid buffer with a pH of 5.000 is sitting on a…
A:
Q: If you were to overshoot tje endpoint in the titration of your unknown acid, would youncalculated…
A:
Q: What will be the total buffer concentration? What is the pH of a solution contains acetic acid d…
A: The solution which is made by the mixing of a weak acid and its conjugate base or vice verse is know…
Q: Why are buffer solutions of increasingly higher pH used to elute the column that generates the…
A: The reason is as given below:
Q: How many grams of Na2CO3 (FM 105.99) should be mixed with 1.70 g of NaHCO3 (FM 84.01) to produce 100…
A: Buffer is a solution that resists the change in pH when small amount of strong acid or base is added…
Q: Select Weak Acid as the analyte in the titration interactive. Determine the pK, of this weak acid.…
A: A question based on equilibrium concept, which is to be accomplished.
Q: Data The following tables include calculated as well as observed and recorded data for Part 1 and…
A:
Q: . Commonly used standards for the direct determination of the normality of an acid. 2. It is used…
A: Given: 1. Commonly used standards for the direct determination of the normality of an acid. 2. It…
Q: Ka = 1.8x10^-5 for ionization of HC2H3O2(aq). Determine the [HC2H3O2]/[C2H3O2-] ratio that will…
A: M
Q: The addition of an acidic solution to a buffer solution with a pH of 8.3 would most likely result in…
A:
Q: Can you show me how to do this
A: Given information: Volume of NH4Cl solution (V1) =25 mL Volume of NH3 solution (V2) =50 mL…
Q: A chemistry student is studying the effect of buffers. During his experiment, he collected the…
A: The Correct Options are A and B.
Q: Calculate the initial [H,O*] in mol/L in the weak acid sample using the initial pH measurement. Show…
A: Initial [H3O+] can be calculated using initial pH of solution of weak acid.
Q: What is the pH of a buffer solution containing 0.30 M HCOOH(aq) and 0.051 M NaHCOO(aq)? The acid…
A: The equilibrium constant for a reaction is expressed in terms of the ratio of the concentration of…
Q: How to compute the maximum and least buffer capacity of H3PO4 at various pH using the trend learned…
A:
Q: ne Diubility of which salt could be decrease g by the additio O NaCI O PbCl2 О КОН LICI O KNO3
A: The solubility of which salt could be decreased by adding KCl :
Q: You are asked to prepare 500. mL 0.300 M acetate buffer at pH 5.10 using only pure acetic acid…
A:
Q: A Review | Constants Period You may want to reference (Pages 314 -318) Section 10.10 while…
A: pH of buffer(aAcidic buffer) can be calculated by using Henderson equationpH=pKa+log(salt)(acid)
Q: Please answer it ALL ASAP and CORRECTLY for an upvote. PROVIDE YOUR FULL SOLUTION. You prepared a…
A: Mixing of 68ml 0.17M ammonia and 42 ml 0.13 M ammonium chloride.
Q: Concentration of standard NaOH (base) .200 M Sample V of acid V of base [HP] (X) Solubility (-X)…
A: Equilibrium is a condition at which rate of forward as well as backward reaction is same. Common ion…
Q: definition of bufter solution i - buffer capacity?
A:
Q: In Protein Precipitation, two liters of 5mM buffer solution with pH 5.2 is needed in the isolation…
A: Buffers are solution that resists the change in pH when small amount of strong acid or base is added…
Q: Determine the relationship between the observed/apparent value (EX) VERSUS that of the true value…
A: To determine relationship between observed/apparent value (EX) vs. ET by writing <, > or = on…
Q: in a container with HCl solution with volume 400 ml and pH = 1, add the salt of PbCl2. Calculate how…
A: Solubility can be calculated using the equation for solubility product.
Q: page 5) can you give me the example for initial HA: 1.00M in table?
A: The ionization reaction of acetic acid is given below,
Q: if wc dissolve an acid dissolve an a cid weak HzA in wate, t he solution volume will be loomb, and…
A: 1- First determine the concentration of H+ ion : We know that, pH = -log([H+]) -pH = log([H+])…
Q: Title: Potentiometric Acid Titration ritulo:Tabla de ph volumen NaOH (ml) [ph Titulación NaOH 20…
A: A potentiometric curve is the plot representing the change in the pH of solution on addition of a…
Q: Can someone explain part B for me I don't understand why [HX]>[X-].
A: Given information: The pKa for weak acid = 4.5 The pH of the buffer solution = 4.3
Q: How many mL of 0.2 M sodium acetate and 0.2 M acetic acid are needed to prepare 1 liter of 0.1 M…
A: Here we are required to find the volume of sodium acetate and acetic acid needed to prepare 1L of…
Q: The Ka expression for the H, PO,/HPO;- buffer is given Ionic strength (µ, M) 0.005 0.010 0.050 0.10…
A:
Q: Determine the [H3O+] of the eight buffer solutions made by mixing 1.10 M acetic acid and 0.900 M…
A: [Acetic Acid] = 1.10 M [Sodium acetate] = 0.9 M We have to determine the concentration of…
Q: Which indicator(s) is/are suitable for this type of titration curve? pH Volume of acid added pH…
A: We need to look at the graph carefully and check which indicator acts the best.
Q: Calculate the Ph of the ffg. a. 500 mL of pure water b. 10.0 mL of hydrochloric acid added to pure…
A:
Q: Ammonia Concentration = 0.17 M Ammonium Chloride Volume = 42 mL | Ammonium Chloride Concentration =…
A: Volume of Ammonia = 68 mL Volume of Ammonium Chloride = 42 mL Total volume of buffer = Volume of…
Q: In the potentiometric titration of phosphoric acid, pH readings at the first and second…
A: pH1 = 2.12 pH2 = 7.21 pH = pka pH = -log[H+] pka = -log ka. Ka1 = ? Ka2 = ?
Q: Show how to prepare a 100-mL acetic acid-acetate buffer (0.1 M, pH 4.0) using stock solutions 1.0-M…
A: A buffer solution is solution that contains both weak acid and its conjugate base or weak base and…
Q: Buffering Capacity Which group is water, which group is phosphate salt, and which group is optimen…
A: The pH of water increases sharply because the concentration of hydroxyl ions increases suddenly on…
Q: A1.0-L buffer solution is 0.125 mol L in HNO, and 0.190 mol L- in NANO,. For nitrous acik K, = 5.6 x…
A:
Q: Conc H3PO4 (85.00% w/w, sp.gr. 1.70 MW: 98g/mol) NaH2PO4 • H2O (MW: 138g/mol) Na2HPO4 • 7H2O (MW:…
A: To prepare the required buffer solutions, we will be using the Henderson-Hasselbach equitation which…
Q: Example: - How many grams ammonium chloride and how many millilitres 3.0M sodium hydroxide should be…
A: A buffer is a solution that can accept protons (by conjugate base) and donate protons (by acid) at…
Q: Select Weak Acid as the analyte in the titration interactive. Analyte, 25.0 ml. Weak Acid, 1.0...…
A:
Q: . So, we need 5.00 mmol of base (titrant) to be delivered to the analyte. Since we know the molarity…
A: The equivalence point in titration is the point where titrant neutralizes the analyte completely. In…
Q: A buffer solution is 0.397 M in H2S and 0.382 M in KHS. If Kaj for H2S is 1.0 × 10-7. what is the pH…
A:
Q: A dilute (~0.1 M) buffer solution contains imidazolium ion/imidazole (see below) and has a pH of…
A:
Q: A student pharmacist prepared a buffer solution by mixing 68 ml of 0.17 M ammonia and 42 mL of 0.12…
A:
Q: How to distinguish between an Acidic Buffer and Acidic Solution as well as Basic Buffer and Basic…
A: An acidic solution will have a pH anywhere from 0-6 and upon addition of a small amount of strong…
Q: Jisoo was tasked to ensure that an enzyme responsible for keeping her species from extinctior…
A: Given the required pH of the buffer solution = 10.00 [H+] = 10-pH = 10-10.00 = 1.00*10-10
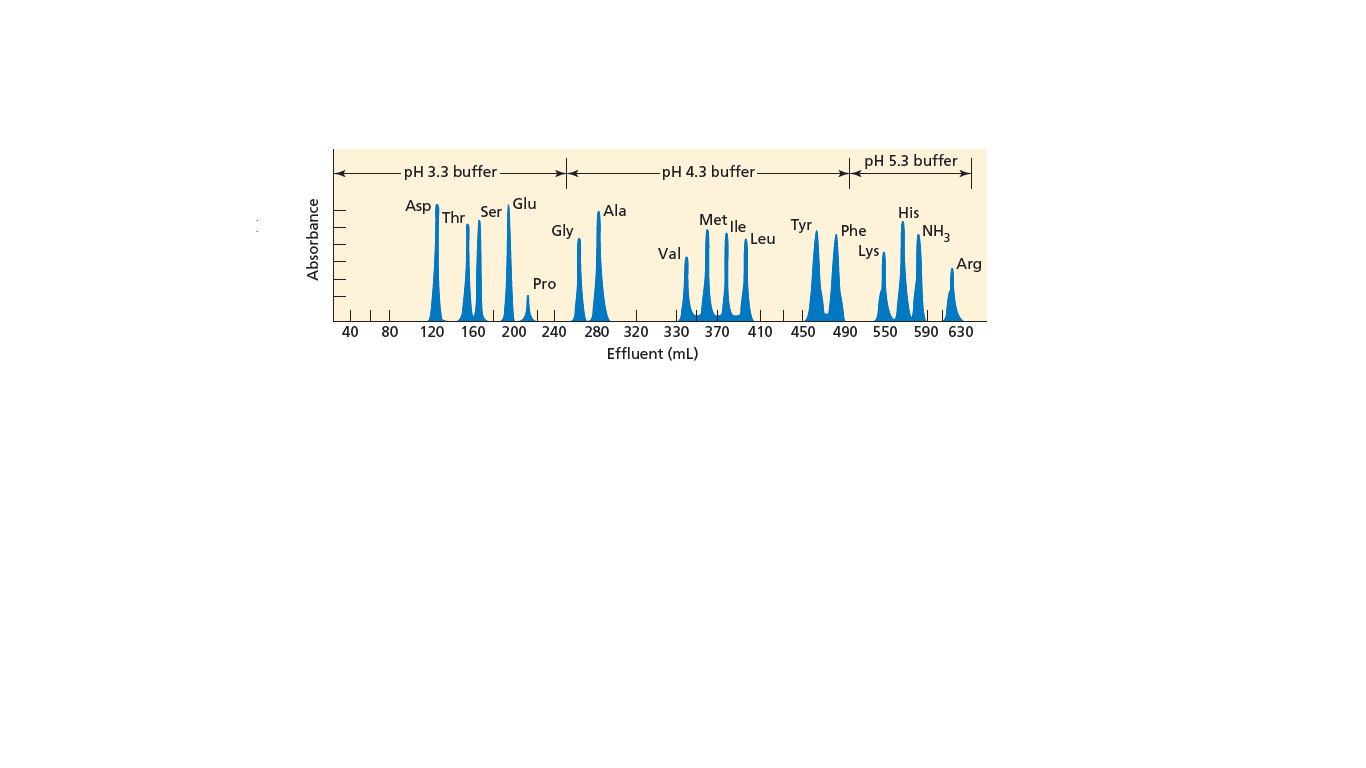
Why are buffer solutions of increasingly higher pH used to elute the column that generates the chromatogram shown in Figure
Step by step
Solved in 2 steps

- If you were to overshoot tje endpoint in the titration of your unknown acid, would youncalculated molar mass be higher, lower or equal to the actual molar massA buffer solution contains formic acid (0.75M) and an equal molar amount the sodium salt of its conjugate base. The Ka of formic acid is 0.00018. At what approximate pH does the solution buffer?A 250 cm3 volumetric flask contains exactly 200,0 cm3 of a 0,025 mol.dm3sulphuric acid solution. Thereafter ten (10) sodium hydroxide pellets, eachof mass 0,1 g are dropped into the flask. After the pellets have dissolvedcompletely, the flask is topped to the 250 cm3 mark with water and thecontents are thoroughly homogenised. Determine the pH of the resultingsolution.
- A buffer system works to keep pH within a narrow range. Buffer systemsare critical in all living cells/tissues. If pH were to measurably shift in a cell,what category of biological molecules would be most impacted, andwhat term refers to the resulting loss of their functional shapes? A buffer is a substance that, when it ionizes in water, creates a buffersystem. The substance and one of its ions represent a weak acid and aweak base. These form a pH-controlled equilibrium. Based on thedefinitions of acid and base, if [H+] increases, which would combine withthe H+, preventing a pH shift? If [H+] decreases, whichwould release H+ to counter a pH change? Check the one characteristic below that is important in a buffer system. maintains a neutral pH prevents pH from dropping prevents pH from rising minimizes change in plets do Calculations System I * 0.010 M HCl * 0.0010 NaOH System II * 0.010 M ( HA ) * 0.0010 M ( B ) System IV * Buffera). What are acids, bases and buffers?b) What are the different types of buffers? Describe the mechanism howbuffers resist pH change in a system.c) Elaborate all chemical reactions in the standardization of NaOH withKHP?d) Why do you need to use KHP in the standardization of NaOH?e) Why is phenolphthalein used as an indicator in this experiment?f) Compare and contrast: endpoint vs equivalence point.g) Discuss the role of acids, bases, buffers and pH in water qualitymonitoring and post-harvest fisheries.
- if the buffering range of benzoic acid is 3 to 5 what is the approximate pka3 5 4 nkt enough informatParameter's Starting HCl conc. (M) 1.000 mL of HCl added 25.00 mL of NaOH to endpoint 21.50 Starting NaOH conc. (M) 1.000 mL of Base Added pH Observed pH Calculated mmol of HCl mmol NaOH mmol of Excess Total Volume M(H+) M(OH-) pOH 18.00 1.30 19.00 1.50 20.00 1.75 20.10 1.80 20.20 1.89 20.30 1.95 20.40 2.01 20.50 2.05 20.60 2.07 20.70 2.09 20.80 2.11 20.90 2.20 21.00 2.27 21.10 2.54 21.20 2.76 21.30 3.30 21.40 5.30 21.50 7.00 21.60 8.40 21.70 10.12 21.80 11.40 21.90 12.10 22.00 12.45 23.00 13.25 24.00 13.54 25.00 13.71 26.00 13.85 27.00 13.93 please help calculate for the rest!Sources of Error Determine the relationship between the observed/apparent value (EX) VERSUS that of the true value (ET) for the quantity being sought by writing either <, >, or = on the space provided TOPIC: Standardization of Titrant Question 7 Distilled water was not equilibrated to room temperature before the preparation of NaOH titrant. EX __ ET TOPIC: Determination of Molar Concentration of each component (Double Indicator Titration) Question 8 No blank correction Ex ___ ET Question 9 Bubbles trapped in the tip of burette: EX ___ ET Question 10 Measuring the sample volume using a volumetric pipet while looking downwards at the meniscus: EX ___ ET
- Please answer the following questions as correct as possible. Thank you. What is the effect of total concentration of the buffer components on its buffering capacity? What is the effect of the ratio of conjugate base to acid on buffering capacity?(2). For the buffer system, what will be the pH when (a) 15.0 ml of 0.500 M HCl and (b) when 15.0 ml of 0.750 M NaOH is added to the buffer? Given: • 20.0 ml of 3.00 M sodium bicarbonate = conjugate base in molarity [? −] • 50.0 ML OF 2.00 M carbonic acid = weak acid in molarity [??] • 4.30 × 10−7 ?? of carbonic = ??? (acid-dissociation constant) • Base: (a) 15.0 ml of 0.500 M HCl (b) 15.0 ml of 0.750 M NaOH1. What is an alkaline acid? In general how we prepare an alkaline buffer? Explain with example and describe in “BULLET POINTS” with mechanism that how an alkaline Buffer can maintain its pH even after adding slight amount of acid or alkali from outside?



