5.Some rocket engines use a mixture of hydrazine, N2HA, and hydrogen peroxide, H202, as the propellant. The reaction equation is N2H4 +2 H2020 how much N2, in moles, is formed? Be sure to use the limiting reagent. Nae) +4 H2Ot) When 0.750 mol N2H is mixed with 0.500 mol H,O2, O a. 0.250 moles O b. 0.500 moles Oc 2.0 moles O d. 1.00 moles
5.Some rocket engines use a mixture of hydrazine, N2HA, and hydrogen peroxide, H202, as the propellant. The reaction equation is N2H4 +2 H2020 how much N2, in moles, is formed? Be sure to use the limiting reagent. Nae) +4 H2Ot) When 0.750 mol N2H is mixed with 0.500 mol H,O2, O a. 0.250 moles O b. 0.500 moles Oc 2.0 moles O d. 1.00 moles
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter3: Mass Relations In Chemistry; Stoichiometry
Section: Chapter Questions
Problem 53QAP: Cyanogen gas, C2N2, has been found in the gases of outer space. It can react with fluorine to form...
Related questions
Question
Transcribed Image Text:All changes save
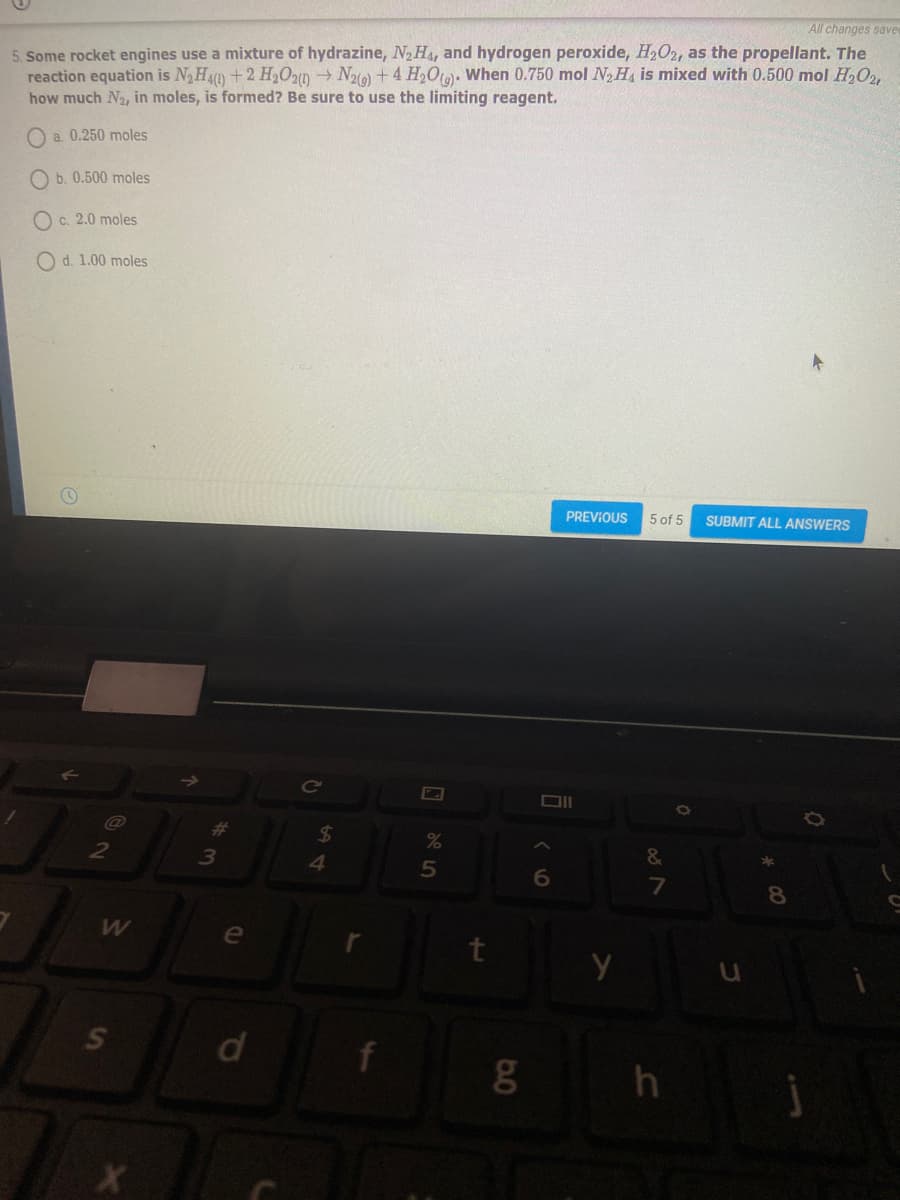
5. Some rocket engines use a mixture of hydrazine, N2H4, and hydrogen peroxide, H2O2, as the propellant. The
reaction equation is NHA) +2 H2O20 N2(0)+4 H2Ot): When 0.750 mol N, H is mixed with 0.500 mol H,O,
how much N2, in moles, is formed? Be sure to use the limiting reagent.
O a. 0.250 moles
O b. 0.500 moles
O c. 2.0 moles
O d. 1.00 moles
PREVIOUS
5 of 5
SUBMIT ALL ANSWERS
Ce
DII
24
23
4.
&
6.
8.
e
g h
d'
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co