Q: Answer the questions based on the resonance structures of S2O32– (thiosulfate) below. See attatched…
A: Thiosulfate ion, S2O32- contains two sulfur atoms and three oxygen atoms with -2 charge. Six…
Q: For the following hydrocarbons, which one is the correct set of formal charges? 11. 111 -N- 1.…
A:
Q: Nitrogen oxides are pollutants and common by-products of power plants and automobiles. NO, can react…
A: NO2 + O2 ==> ?
Q: The compound above is an example of a(n)
A:
Q: A 1.51 How are the molecules or ions in each pair related? Classify them as resonance structures,…
A: Isomers have the same number of elements in the compounds, but with different structures, Resonance…
Q: CH3S+ pg In this structure, what is formal charge on S? Please click on jpg file to see image 0. O…
A: The formal charge on the atom in a molecule can be calculated as follows Formal charge = Valence…
Q: Which bond is the most polar? OH-N ОН-С он-о он-а OH-F
A: Polarity is defined as the separation of positive and negative charge between two element that…
Q: Consider compounds A-D, which contain both a heteroatom and a double bond. N. H. A B a. For which…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: 3. Draw the best complete Lewis structure for each of the following molecules or ions. Make sure to…
A: we have to draw the Lewis structures of the given molecules
Q: 23. Write another resonance structure for ethyl acetate. Include formal charges. CH ÖCH,CH,
A: Resonance is the phenomenon in which the lone pairs and/or the double bonds delocalize within the…
Q: How are the molecules or ions in each pair related? Classify them as resonance structures, isomers,…
A: a.
Q: ed för this question. The formal charge is the "charge" an element would have in a molecule or ion…
A:
Q: In this structure, what is formal charge on S? O-S=O.jpg Please click on jpg file to see image O +1…
A:
Q: Draw the Lewis structure of the missing reactant. Make sure to include lone pairs and non-zero…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: How are the molecules or ions in each pair related? Classify them as resonance structures, isomers,…
A:
Q: ) POCI; # valence electrons: Each atom w/ Octet: minimized Formal Charge: -) SO3 # valence…
A: Formal charge is calculated as :- Number of Valence electrons - 1/2 (number of bonded electrons) -…
Q: Label each non-hydrogen atom in the structure below with its formal charge
A: Formal charge: The formal charge (FC) is the charge assigned to an atom in a molecule, assuming that…
Q: Braw the Lewis structure of the missing reactant. Make sure to include lone pairs and non-zero…
A: In this question, we will draw the Lewis structure of the missing reactant. HNO3 is strong acid and…
Q: 2H-N-H > 3 H-H + N N You can find a table of bond energies by using the Data button on the ALEKS…
A: Enthalpy of the reaction = Total bond energy of products - total bond energy of reactants
Q: estion 23 What is the formal charge of C in the molecule below? O a.-1 O b. +1 O c. 0 O d. +2 A…
A: Carbon monoxide molecule contain triple bond between carbon and oxygen atoms
Q: 2. Determine the formal charge on each atom of the molecule below, which belongs to a class of…
A: Formal charge: The formal charge (FC) is the charge assigned to an atom in a molecule, assuming that…
Q: Which compound contains a nitrogen atom with a formal positive charge?.. 'N' II III A) I В) п C) III…
A: By using this formula we can calculate the formal charge on the atom . Valence electrons in N = 5
Q: „CH3
A: Resonance structures are sets of Lewis structures that describe the delocalization of electrons in…
Q: Draw the Lewis structure of SF,. Include all the lone pairs. 2° Select Draw Rings More F
A: Lewis structures can be defined as the structures that show the bonding between atoms of a molecule…
Q: Draw the Lewis structure of the missing reactant. Make sure to include lone pairs and non-zero…
A: Bronsted acid : Proton (H+) donar is called Bronsted acid Bronsted base : Proton (H+) acceptor…
Q: O Write the resonance structure for each of the following compounds. Which resonance structure…
A:
Q: Draw a resonance structure that shifts a pi bond to a new position. Don't increase the number of…
A: To solve this problem we have to draw the resonating structure of the given compound .
Q: Consider compounds A-D, which contain both a heteroatom and a double bond. H. A D a. For which…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: Draw the Lewis structure of the missing reactant. Make sure to include lone pairs and non-zero…
A: Given : Reaction To find : Lewis dot structure Solution : Symbols of the elements are the first…
Q: How many a bonds are present in the following molecule? F F Select one: O a. three O b. four O c.…
A: in double bond one bond is pie bond and another bond is sigma bond.
Q: Which of the following molecules or ions are likely to be free radicals: N2O, OCl-, and ClO2? a)…
A: The molecule which has unpaired electron can be free radicals.we will see the structure one by one…
Q: Draw all possible resonance structures for NPO in your work. Then, assign formal charge to each atom…
A: We have to draw all the possible resonance structure for NPO and also assign the most stable…
Q: Which statement is true regarding the pairs of molecules in a and b, below? а) and b) and H3C CH3…
A:
Q: For parts a and b calculate 1. The formal charge for all the non-hydrogen atoms in the molecules for…
A: Formal charge= Valance electron - unbounded electron - 1/2 bonded electron
Q: Carbon Thioxide (OCS) d) determine the partial charges of the CO bond e) determine the direction…
A:
Q: Assign a formal charge to each atom of the above. OCI = 2', 0- 1 O C 0,0= 0 O CI - 2", O = 1 O CI-…
A:
Q: 47. How many resonance structures does HSeO3- have? What is the formal charge of the Se atom in…
A: HSeO3- has 2 resonance structures. The resonance structures are given as:
Q: QUESTION 5: Draw a resonance structure with two formal charges and explain why that resonance…
A: The molecule given is,
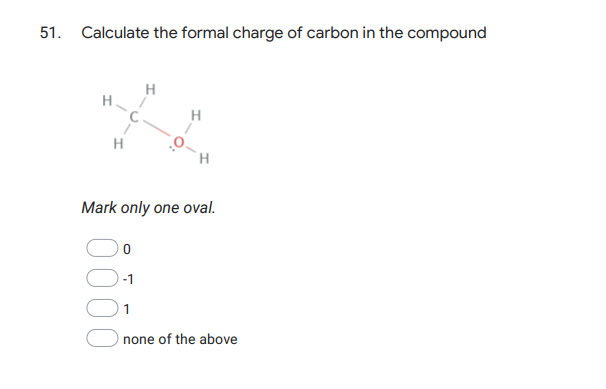
Q: 50. Calculate the formal charge of oxygen in the compound H H H Mark only one oval. -1 1 none of the…
A: Formal charge The formal charge over an atom of a polyatomic molecule or ion is the difference…
Q: 23. Write another resonance structure for ethyl acetate. Include formal charges. CH, ÖCH,CH,
A:
Q: 1. Consider the following skeletal/condensed structure: H2N. a. Add in all the Carbons, Hydrogens,…
A: Concept introduction: Expanded formula is the formula in which all atoms are shown in the molecular…
Q: Respond to Questions on Lewis structure and molecular geometry using VSEPR model. For each of the…
A: In Lewis structure of a molecule, all the valence electrons are denoted by dots. According to…
Q: NH,+ Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone…
A:
Q: Considering the structure on the far left as the original resonance structure, which of the…
A: The original structure and the resonance structures given are,
Q: Draw all possible resonance structures for NPO in your work. Then, assign formal charge to each atom…
A: The compound given is NPO.
Q: Write all resonance structures for NCO− ion. Use formal charges to predict the most stable one (i.e.…
A:
Q: ough we usually don't show lone pairs on skeletal structures, sometimes it's necessary, for example,…
A: Lewis structure is a method of of representing bonding pattern in a molecule. it represent how…
Q: In which of the structures shown below does each atom have a formal charge of zero? :F: o=N-0 =N-0-…
A: Option A is correct answer Structure B has zero formal charge.
Step by step
Solved in 2 steps

- Draw the missing lone-pair electrons and assign the missing formal charge on N or O for the following H-C-O-HUse the formal charge to draw in the lone pairs on each N or O atom in the following compounds.Draw the Lewis structure (including all lone pair electrons and any formal charges) for one of the four possible isomers of C3H9N.
- Projection drawings of all isomers of C6H4Cl2. Can you convert one into another without breaking any bonds? What type of isomerism do these represent?How many π bonds are there in 1,2-propadiene (H-C=C=C-H)?draw the full lewis structures of a ketone isomer and an aldehyde isomer with the formula C6H6O . make sure lone pairs are shown ketone aldehyde
- Write the best Lewis or Kekulé structure (showing all lone pairs of electrons, formal charges, and appropriate geometry – use wedge-dash where needed) for each of the following species given as condensed formulas: a) NH2- b) PO3H3 c) H2COH+ d) NaBH3CN e) C6H5MgBr f) CH3AlCl2Draw thee other resonance structures for the following carbocation. Use electron pushing arrows in order to derive one resonance structure from another. Indicate the formal charge on each atom in your structures.The curved arrow notation introduced in Section 1.6B is a powerfulmethod used by organic chemists to show the movement of electronsnot only in resonance structures, but also in chemical reactions.Because each curved arrow shows the movement of two electrons,following the curved arrows illustrates what bonds are broken andformed in a reaction. Consider the following three-step process. (a) Addcurved arrows in Step [1] to show the movement of electrons. (b) Use thecurved arrows drawn in Step [2] to identify the structure of X. X isconverted in Step [3] to phenol and HCl.

