Q: (ii) What is the significance of Kp? Hence, arrange the three molecules in terms of decreasing…
A: Given:
Q: What is the coefficient, b, in the reaction below, if Kc = 1.39e-5 and Kp = 2.25e-10 at 207.96°C?…
A: Given, 2A(s) + bB(g) ↔ 2C(g) Kc = 1.39 x 10-5 Kp = 2.25 x 10-10 Temperature = 207.96 °C To…
Q: Describe the formulation of the Eyring equation; in what sense is it superior to the coll ision…
A: Transition state theory: In transition state theory it is supposed that an activated complex is in…
Q: 6. Indicate the reaction chemistry and draw the most probable reactant for the reaction below: 1. BH…
A: In this question, we will indicated the reaction chemistry and draw the structure of the starting…
Q: What type of reacton is C(s) + CO2(g) ⇌ 2CO(g) a. reversible equilibrium b. catalytic…
A: There are two types of equilibrium - 1) homogeneous equilibrium 2) heterogeneous equilibrium…
Q: Show the complete electron transfer reaction for the following chemical reaction: C6H12O6+O2=CO2+H2O
A: Balancing of reaction – A chemical reaction involves reactants and products.According to law of…
Q: (a) When the metallic element sodium combines with thenonmetallic element bromine, Br21l2, what is…
A: Below are the answers to all the parts.
Q: What type of bond cleavage takes place in/what type of intermediate is produced in the following…
A: Concept introduction: Chemical reactions are the result of bond breaking and bond making. There are…
Q: Write down the mechanism of producing OH in the remote clean environment.
A: Answer - The hydroxyl (OH) radical is the key oxidant in the global atmosphere as it controls the…
Q: Molecular collisions may or may not lead to a chemical reaction between Cl and NaCl. True or false?
A: The given statement is, Molecular collisions may or may not lead to a chemical reaction between Cl…
Q: Which of the following is an example of a catalyst? dNTP's used as building blocks for a new DNA…
A: Catalyst which affects the rate of reaction:
Q: (g) 1) OsO4 2) NaHSO3
A: Alkenes can be converted to diols by treating with OsO4 and KMnO4 . Both reagents gives cis-diols.…
Q: Determine the type of reaction. Br `NH2 + 2 NH3 + NH,Br O A. Hydrolysis O B. condensation O C.…
A:
Q: Given the following balanced equation, determine the rate of reaction with respect to [SO2]. 2SO2(g)…
A: The rate of a given reaction can be defined as the speed at which the conversion of reactants into…
Q: Oxygen gas speeds up the rate at which food is spoiled. To improve the shelf-life of foods, food…
A: The reason why the given method is not preferred to enhance the shelf-life of all foods has to be…
Q: To determine the rate of the following reaction what physical property could be measured? H2 + I2 ->…
A: Answer - Rate of reaction - The reaction rate or rate of reaction is the speed at which a chemical…
Q: What are the intermediates of the following reaction?
A: The reaction of 1-methyl-cyclopentene with diborane followed by hydrogen peroxide and sodium…
Q: If the concentration at 3 hours is 50mg/l and k = 0.1hr-1. What is co?
A: Answer:- this question is answered by using the simple concept of chemical kinetics which involves…
Q: Predict the product of the following reaction: Na OH, H₂O 2
A: Note : Hydroxide removes from secondary carbon. But ketones pka value is around 20. Water pka value…
Q: Br Bu,NF/ THF (iii) L: C4H3O
A:
Q: EtO, Eto NABH, N. ELOH NH 6 5a Ph Ph
A: The above reaction proceeds through a two step mechanism. At first step the bolu hydride group…
Q: HBr, (t-BuO)2
A: Major product of following reaction
Q: Give the major product(s) of the following reaction. 1) KMNO4, OH", heat ? 2) Hзо A OH В но
A:
Q: 21. The reaction scheme for the preparation of Z starting from benzene is as follows: C,H;CI/AICI,…
A: The main reaction of benzene is an aromatic electrophilic substitution reaction. The first step of…
Q: 2a. Consider the reaction: H₂O(1) H*(aq) + OH(aq), which is known at the autoionization of water.…
A: To determine the ∆H0 and ∆S0, we'll plot the ln Kw against 1/T.
Q: H2O HO. H,SO4 OH
A: Lactone in presence of acid undergoes hydrolysis results in the formation of carboxylic acid and…
Q: Which of the following parameters would be different for a reaction carried out in the presence of a…
A: The addition of a catalyst increases the rate of the reaction by providing an alternate lower energy…
Q: Which of the following disturbances can increase the production of ammonia in the Haber process? N2…
A: The reaction of the formation of ammonia is exothermic.
Q: What is the Kc expression for this reaction? 2N6CIA(g) NbCla(g) + NbCls(g)
A:
Q: A proposed mechanism for ozone destruction in the late spring over northern latitudes in the lower…
A:
Q: Which of the following statements is TRUE about catalysts- a. They lower the Ea for at least one…
A: In the presence of a catalyst, the reaction proceeds by the new path, and the activation energy of…
Q: What is the coefficient, b, in the reaction below, if Kc = 8.96e-3 and Kp = 4.92e-8 at 417.53°C?…
A:
Q: What is the mechanism of the phosphate buffer action? Write down the equations of these reactions.
A:
Q: Select the products formed under the following one-step reaction: Br2 ? Br Br Br, Br Br, Br Br Br…
A:
Q: Ir-CI HO H-CI + Ph = + 1 atm CO Ph
A: We have to give the mechanism for the reaction below. Include the oxidation state of the metal as…
Q: Pd(PPH3)4 CI + -B(OH)2 КОН
A: To know deep about this reaction please read suzuki cross coupling and then try to solve. I have…
Q: 3. The decomposition of hydrogen peroxide in dilute sodium hydroxide solution is : 2 H2O2(ag) 2…
A:
Q: Which of the following compounds will liberate nitrogen gas when reacted with nitrous acid at room…
A: The given reaction is a test for amines.
Q: Complete the reactions by predicting the product/s: MgO + H20 – Mgo + H2 b. Mg(OH)2 c. no reaction…
A:
Q: H20, H*
A: Given Given : Reaction of alkene with H2O To find : Product
Q: Predict the products and what is the reaction mechanism?
A: The reaction mechanism for the following reaction is SN2 mechanism.
Q: Which of the following compounds will liberate nitrogen gas when reacted with nitrous acid at room…
A: Nitrous acid is a kind of inorganic acid which can produce NO+ by the following reaction-- HNO2 →H+…
Q: Answer the following questions regarding reactions involving ions in aqueous solution. i. Consider…
A:
Q: -COOH (7a)_ + 2 NaOH _(7b)_ NazCOs. +. H2O
A: An acid is a chemical substance that can furnish the hydrogen ions in an aqueous solution and can be…
Q: Which of the following is the intermediate for the following reaction? A) I B) II C) III D) IV
A: When an alkene reacts with a water molecule in the presence of acid (H+), the reaction is called…
Q: H20 H;0*
A:
Q: Predict the products of the following reaction. I. NaOH, 70Y 2. 10 NO HO IV NO.
A: NOTE : according to the policies, we can only answer one question at one time. you have to post…
Step by step
Solved in 2 steps with 2 images

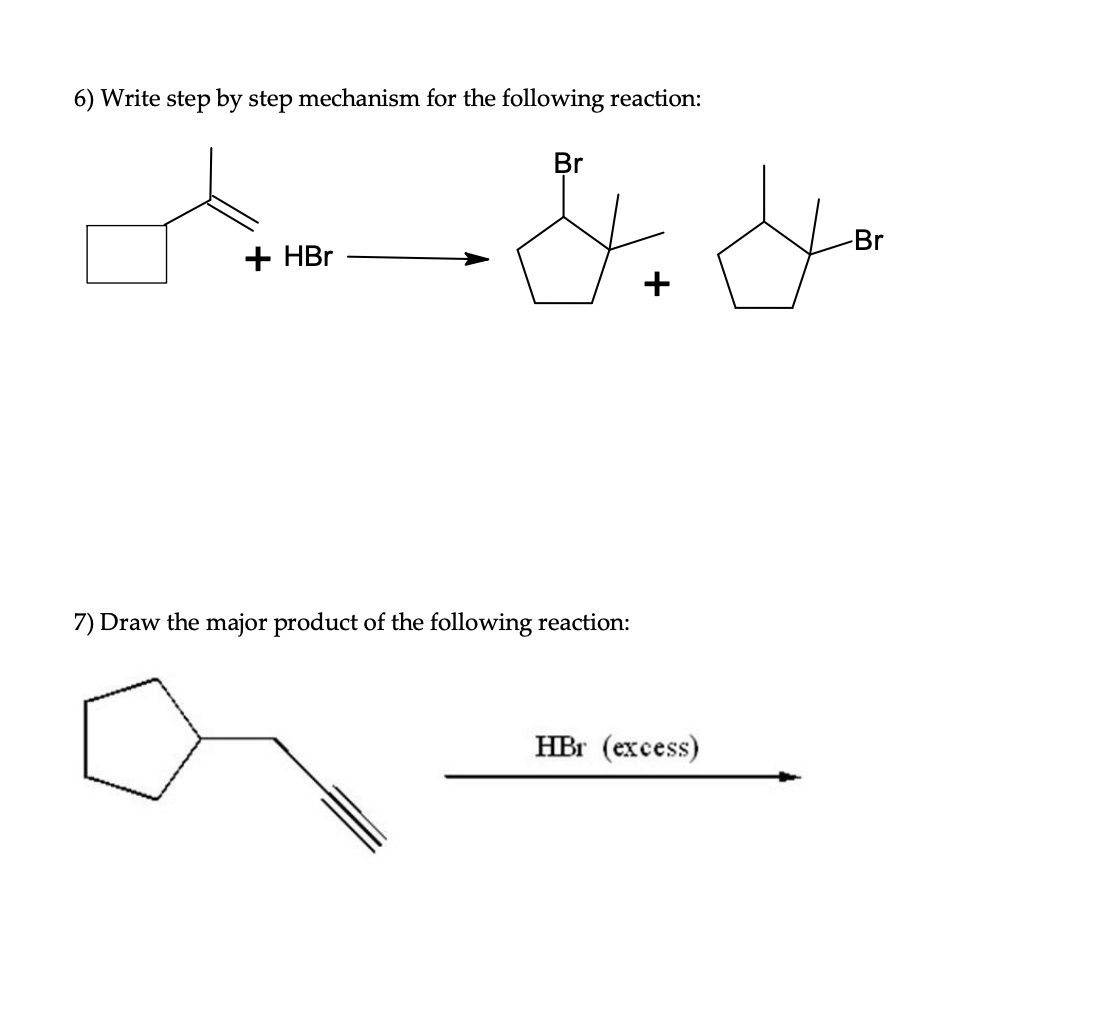
- The following reaction occurs at room temperature in acidic solution, first formingintermediate A, which then rearranges into final product B .i. Show the mechanism for the formation of Intermediate A. )ii. Show the mechanism for the rearrangement of A to B. Explain why the reaction does NOT occur readily in basic solution using your mechanism from 2.a5..8. What is the expected major for the following reaction ?3. Prepare each compound from cyclopentanol. More than one step may be needed.llustrate the mechanism of the reaction. Include the intermediate product and by-product formed in the reaction. (see attached photo)
- In each reaction box, place the best reagent and conditions from the list below. (A reagent may be used more than once.) Hint: step 1 is conducted at –78 °C.6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, PdI. Propose a mechanism reaction for the reaction below. 2. Identify the major and the minor products.
- Consider the reactions shown below. For each reaction state if it would favor S N 1 or S N 2pathway and give the major product of the reaction.What is the product of the reaction below when and b are the steps needed for the reaction to occur. Reacting with a first followed by b.Identify the reagents needed to come up with the given compounds. Pls Separate the reagents with a comma and a space. (ex. HNO3, H2SO4). For a multi-step reaction, separate the reagents with a semi-colon. (ex. HNO3, H2SO4; CH3Cl, AlCl3) There should be 9 reagents
- What are the products for the following ozonolysis reaction? A) I and II B) II and III C) I and III D) I and IVHydrazine reacts with 2,4-pentanedione to yield 3,5-dimethylpyrazole. Including protonations and deprotonations, the reaction takes 12 steps. Write out the mechanism on a sheet of paper and then draw the structure of the product of step 6.Propose a reaction mechanism for the reaction below 2 H2 (g) + 2 NO (g) --> N2 (g) + 2 H2O (g)

