Q: jlp.3
A: The objective of this question is to calculate the rate constant at a different temperature using…
Q: The tertiary structure of a protein is a complex arrangement formed as the polypeptide chain folds…
A: Types of bond in tertiary structureExplanation:The interactions stabilizing the tertiary structure…
Q: C2H4(g) + O2(g) → CO2(g) + H₂O(g) what is the coefficient of O2 in the balanced equation?
A: The objective of this question is to find the coefficient of in the given equation
Q: At a certain temperature, the K, for the decomposition of H₂S is 0.755. H₂S(g) H₂(g) + S(g)…
A: Given Kp for the below dissociation reaction is 0.875.H2S(g) H2(g) + S(g) ; Kp = 0.755
Q: Draw and Identify the following structures at physiological pH NH₂ 1 HO 3 IZ H₂C CH₂ 2 H CH, CH3 O:…
A: To identify the structures of molecules at physiological pH (around pH 7.4) we will first identify…
Q: Consider the following unbalanced reaction: H₂O(g) ⇒ 0₂(g) + H₂(g) 2nd attempt Complete the K,…
A: Write the balanced reaction for the given equilibrium reaction.The partial pressure of the products…
Q: A mixture is two or more substances mixed together that are not chemically bonded. Which of the…
A: The objective of the question is to identify which of the given substances are mixtures. A mixture…
Q: GenAlex Medical, a leading manufacturer of medical laboratory equipment, is designing a new…
A:
Q: A reaction vessel contained solid C and CO2 gas at a pressure of 0.482atm at a certain temperature.…
A: Answer:For any reaction, value of equilibrium constant KP is equal to the ratio of partial pressure…
Q: Do not give handwriting solution.
A: A is 2-phenyl ethanol and B is 1-bromo2-phenylethaneExplanation:The given compound is 2-phenyl…
Q: Please write out this mechanism the formation of fructose 1,6-bisphosphate from dihydroxyacetone…
A: The objective of the question is to describe the mechanism of the formation of fructose…
Q: Identify the atom which has the highest effective nuclear charge (Zeff) for its valence electrons.…
A: In general,down the group Zeff remains constant.but from left to right in a period Zeff Increases.
Q: Br 1. 2. CH3(CH₂)3Li 3. P: 3
A: Reaction of phosphorous ylides with aldehydes and ketones forms substituted alkenes. This reaction…
Q: Consider the following equilibrium. A + 2B C + 4D If the change in concentration for C is 0.260 M,…
A: Given data,reactionthe change in concentration of C = 0.260 Mwe have to calculate the change in…
Q: What is the half-life in minutes of the reaction if k is 27.5616 /min and the initial amount of the…
A:
Q: OH Click and drag to start drawing a structure.
A: we have to draw the major product
Q: he provided starting and procct structures, draw the curved electron-pushing arrows for the…
A: Sulfonation of benzene is the aromatic substitution reaction in which the introduction of -SO3H…
Q: When silver nitrate reacts with barium chloride, silver chloride and barium nitrate are formed. How…
A: The mass of silver nitrate is 10.2 g.The mass of barium chloride is 15.0 g.To find the mass of…
Q: - Suppose a small sample of pure X is held at -236.°C and 2.0 atm. What will be the state of the…
A: The phase diagram shows the temperature and pressure at which different phases of a substance are…
Q: The conventional standard Gibbs energy of ammonia at 25 °C is -16.5 kJ/mol. Calculate the value of…
A: To calculate the molar Gibbs energy at different pressures, we can use the equation:ΔG = ΔG° + RT…
Q: Draw an equivalent resonance structure that minimizes charge. Include all lone pairs in your…
A: AnsweredExplanation:Step 1: All resonance structures with curved arrow notation is shown below Step…
Q: A chemist dissolves 625. mg of pure barium hydroxide in enough water to make up 160. mL of solution.…
A: The mass of barium hydroxide is 625 mg which is .The volume of the solution is 160. mL which is .To…
Q: . 15.25 In the mass spectrum of the product of the following reaction, the base peak appears at m/z…
A: The objective of the question is to find the structure of the product of the given reaction that…
Q: An organic chemist attempted to produce an ether using the Williamson ether synthesis. What was the…
A: In Williamson ether synthesis, a sodium alkodixe reacts with alkyl halide to produce ether. In the…
Q: The reaction between nitrogen dioxide and carbon monoxide is NO₂(g) + CO(g) → NO(g) + CO₂(g) The…
A:
Q: نے B. C. ttt سکند
A: To encounter longest chain,(i) one has to numbering the longest chain possible.(ii) Number the…
Q: Draw the skeletal structure for the product that forms when the alcohol 3-methyl-2-butanol is…
A: Given,The reactant of the molecule is:3-methyl-2-butanol
Q: Identify the expected major product of the following Diels-Alder reaction. + En I || ||| IV V + + En…
A: REaction involved diene and dienophile. This is (4+2) cyclo addition reaction. Dienophile contains…
Q: which one is the correct name for the following? 1-fluoro-2,5-diemethylcyclohexane…
A: The objective of the question is to identify the correct IUPAC name for a given compound. The IUPAC…
Q: What is the relationship between the two chair conformations below? F
A: Conformational isomers, also known as conformers or rotational isomers, are a type of stereoisomer.…
Q: You have a 2 g/mL solution of erythromycin. What volume must be added to a 500 mL infusion to…
A: The objective of the question is to find out the volume of erythromycin solution that needs to be…
Q: Click the "draw structure" button to launch the drawing utility. Draw the diene and dienophile that…
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: I and II are: OH Br O identical. constitutional isomers. diastereomers. enantiomers. HO O not…
A: The question is asking us to determine the relationship between two molecules, I and II, which are…
Q: . Provide a base to prepare the following major products for each of the following E2 reactions.…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Which is the major isolated organic product of the reaction shown? A) C) Br Br HBr -42 °C B) D) Br á…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Neuramidic acid (HNad, pKa=8.0) is dissolved in an aqueous solution, and the pH is 6.0. What is the…
A: Henderson–Hasselbalch equation relates the pH, pKa, and ratio of an acid and base.pH=pKa - log…
Q: What is the major product of the following reaction? а (d) a Ob с CH3 НА CH3OH CH2-OH 0- CH3 CH3 (b)…
A: Under acidic conditions, cyclic oxide open in an SN1 like reaction with the nucleophile attacking…
Q: Draw the lewis structure for GaO3
A: Lewis Structure:The Lewis structure is also known as the electron dot structure.It is a way of…
Q: Draw the skeletal structure of the disulfide formed when the thiol shown is oxidized. SH
A: The oxidation of thiol groups to disulfide bonds involves the removel of hydrogen atoms from the…
Q: If 100mL of 0.481M NaSO4 are added to 100.0mL of 0.819M Pb(NO3)2, how many grams of PbSO4 can be…
A: The objective of the question is to calculate the amount of PbSO4 that can be produced when 100 mL…
Q: ONCEPT MAPPING Marvin started a concept map to demonstrate the properties of water. He lost the map…
A: Given is a mind map for water molecule.To fill all the blanks by understanding the properties of…
Q: Rank the elements or compounds in the table below in decreasing order of their boiling points. That…
A: The objective of the question is to rank the boiling point in the order of decreasing order for the…
Q: The heat of fusion AH, of methanol (CH,OH) is 3.16 kJ/mol. Calculate the change in entropy AS when…
A: The objective of this question is to calculate the change in entropy (ΔS) when methanol melts. The…
Q: What is/are the product(s) of the following acid-base reaction mechanism?
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic…
Q: Using the data in the table, calculate the rate constant of this reaction. k= A+B C+D Trial [A] (M)…
A: Given,The reaction:A + B C + D
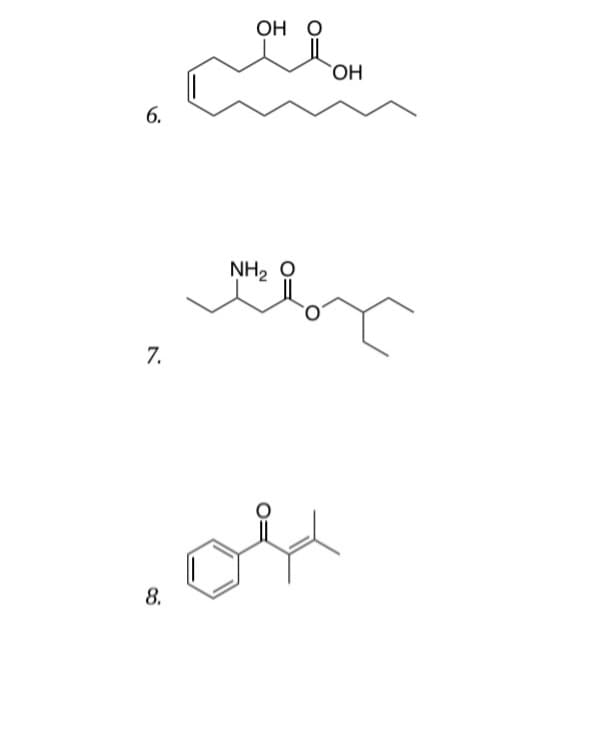
Q: Give correct detailed Solution with explanation needed with structures...don't give Handwritten…
A: Structure 1: The functional groups are as follows:AlcoholAldehydeAlkeneCarboxylic acidsStructure 2:…
Q: Aldehydes and ketones differ only by location of the carbonyl group and can be isomers of each…
A:
Q: ОН
A: The basic principle is; the bulky group should always come at equatorial position.The higher the…
Q: The progress of a reaction is tracked by following the concentration of one reactant as it is…
A: To find the average rate of the reaction between the time mentioned in the question.Concept Average…
Q: -Br NaCN, CH3CN
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Step by step
Solved in 5 steps with 4 images

- Answer 1,2,3,4,5. Take note of the directionCalculate Delta Hof for PbO (s) using the data below. PbO (s) + CO (g) ® Pb (s) + CO2 (g) DH° = –131.4 kJ DHf°: CO2(g) = –393.5 kJ/mol; CO(g) = –110.5 kJ/mol –151.6 kJ/mol –283.0 kJ/mol +283.0 kJ/mol –372.6 kJ/mol +252.1 kJ/molMy question is for the part circled in pencil, how do we get -12,970.4 and then -25,940.8 j/mole. Are we normalizing twice? If so, why?
- For question 1,2,3,4 What will the shift be? How do you know? Options: shift left, shift right, or no shiftCalculate the ΔG°rxn using the following information.4 HNO3(g) + 5 N2H4(l) → 7 N2(g) + 12 H2O(l) ΔG°rxn = ?ΔH°f (kJ/mol)HNO3 = -133.9N2H4 = 50.6N2 = 0.0H2O = -285.8S°(J/mol∙K)HNO3 = 266.9N2H4 = 121.2N2 = 191.6H2O = 70.0Don't understand 4 coming out with a number slightly off

