6. Determine whether each reaction occurs by an SN2, SNI, EI, and/or E2 reaction. Provide a detailed mechanism with curved arrows and the major product(s). NaSH DMF OMs NaOH H20, heat LDA Br DMF NaOH H2O, heat OH HBr OH H, H20 heat
6. Determine whether each reaction occurs by an SN2, SNI, EI, and/or E2 reaction. Provide a detailed mechanism with curved arrows and the major product(s). NaSH DMF OMs NaOH H20, heat LDA Br DMF NaOH H2O, heat OH HBr OH H, H20 heat
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter8: Addition Via Carbocation
Section: Chapter Questions
Problem 16E: A student proposes the following reaction mechanism for the reaction in Model 6. Which step inthis...
Related questions
Question
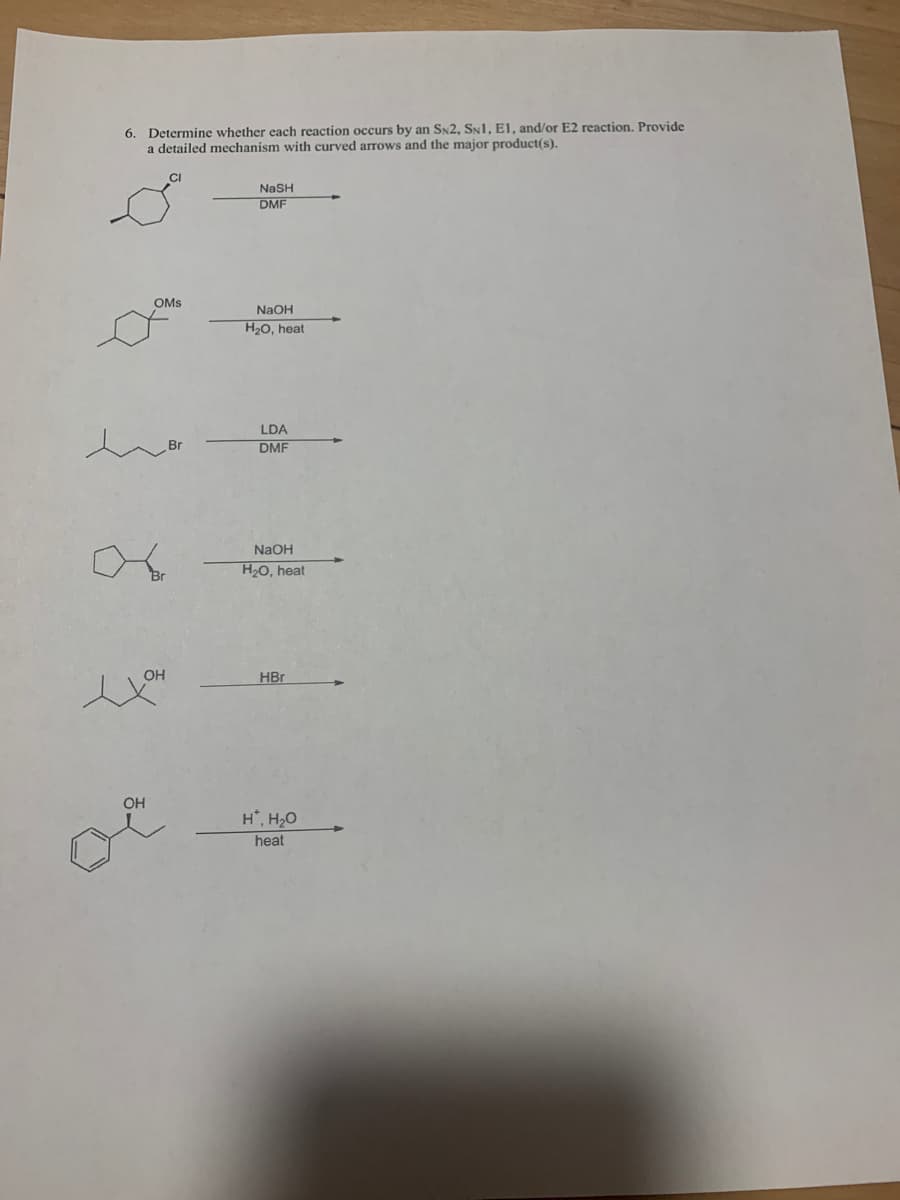
Transcribed Image Text:6. Determine whether each reaction occurs by an SN2, SN1, EI, and/or E2 reaction. Provide
a detailed mechanism with curved arrows and the major product(s).
NASH
DMF
OMS
NaOH
H20, heat
LDA
Br
DMF
NAOH
H20, heat
OH
HBr
OH
H*, H20
heat
Expert Solution
Step 1
The higher the temperature, the more likely elimination occurs. The lower the temperature, the more likely substitution occurs
Step 2
1)
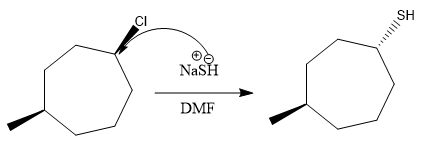
Here, SH is a nucleophile and this is a substitution reaction.
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning