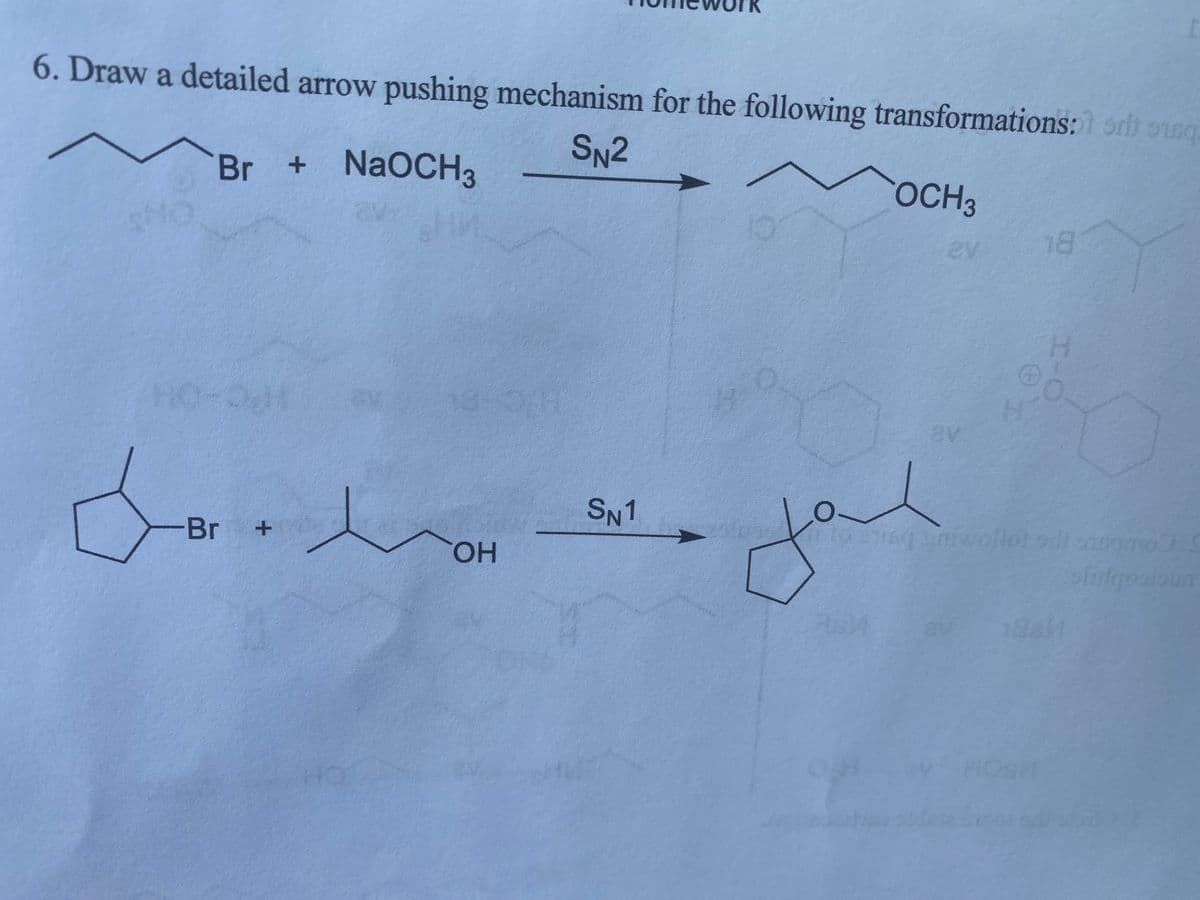
Q: The cyclohexyl bromide shown below does not undergo E2 elimination. Draw both cha conformations for…
A: Welcome to bartleby !
Q: Which given option is correct for the following transformation? MCPBA H2SO4 1 2 CH3OH OH OCH3 LOCH3…
A: The reaction given is,
Q: 2. Which one of the following compounds is optically inactive? A B E
A:
Q: Using Cahn-Ingold-Prelog rules, rank these substituents from highest priority to lowest priority. II…
A:
Q: Dtermine the Product from the rxn 1. ΗΝΟ,/Η,SO4 2 Br2/FeBr3 Product 3. H2/Pd/C 4. Cl2/FeBr3 5.…
A: Here we have to choose correct option about product of above reaction-
Q: 10. Write priorities on these groups ( 1= highest, 4 = lowest) and assign the stereocenter as R or…
A: Here we are required to assign R /S configuration to stereocentre.
Q: -CF3 (7) Please select the major product in the nitration to CF3 CF3 NO2 ÇF3 ÇF3 NO2 A. В. C. `NO2…
A: Given organic compound is (Trifluoromethyl)benzene. It is a deactivating substituent. Deactivating…
Q: When determining R AND S rank these groups from highest to lowest priority
A: Priorities are given according to the CIP rules. 1. Higher the atomic number greater will be its…
Q: Which group in each pair is assigned the higher priority in R,S nomenclature? a. – CD3, – CH3 b. –…
A: INTRODUCTION: The arrangement of atoms or groups (LIgands) in space around a chiral centre that…
Q: PQ-6. What is the rate determining step of this CH2CH3 NACN CN reaction? H3C OTs acetone H3C CH2CH3…
A: Rate determining step is the slowest step of reaction .
Q: Rank each set of substituents using the Cahn-Ingold-Prelog sequence rules by numbering the highest…
A: According to Cahn Ingold prelog sequence substituents with higher atomic number has highest…
Q: 2. H₂C CH₂ مله CH3 + H₂C + AICI3 → (f) (major product only) CH₂ + Cl₂ (in uv light) → (g) (major…
A:
Q: 2.2 Draw all the important resonance contributors for the arenium ions formed from each of these…
A:
Q: Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing…
A: In the electrophilic addition reaction of dienes, the proton is added to form the allylic resonance…
Q: Determine the missing product. а. НСI + H 0 > Нзо+ + b. F- + H2О > HF + с. НSO4 + Н-О — HSO4 + d.…
A: In each of the given reactions, hydrogen ions (H+) are substituted from one species to another.…
Q: Draw all stereoisomers formed in the following two-step reaction sequence.
A: mCPBA is meta-chloroperoxybenzoic acid is widely used as an oxidant in organic chemistry. mCPBA…
Q: Draw a Newman projection for the conformation adopted by 2-bromo-2,4,4-trimethylpentane in a…
A:
Q: Which group in each pair is assigned the higher priority in R,S nomenclature? a.−CD3, −CH3…
A: The rules that must be followed while prioritizing the substituent group for R-S configurations are-…
Q: Which of the given compound is most reactive towards E2 elimination? CI H3C, ICH3 H3C CI H3C CI H3C…
A:
Q: Rank the set of substituents below in order of priority according to the Cahn-Ingold-Prelog sequence…
A: Cahn-Ingold-Prelog sequence rules are followed to find the priority of groups attached to chiral C…
Q: Rank the following groups in order of decreasing priority according to the Cahn-Ingold-Prelog…
A: Cahn-Ingold-Prelog system is a sequencing rule used in organic chemistry to assign priorities to the…
Q: 6. Draw the structure for compound A and for compound B in the following synthetic sequence. 1) Mg.…
A: In this question we have to tell the product of the reaction.
Q: To which compound is the addition of HBr more highly regioselective (without any shift) and what…
A:
Q: The degree of unsaturation of the formula C10H7BRN203 is Select one: 4. 6. 8. 7.
A: The compound given is C10H7BrN2O3.
Q: Circle one or more: E2 SN1 E1 SN2 PHSH, 25°C Mechanism: Circle one or more: SN1 SN2 E1 E2 iPrONa,…
A: There are number of functional group associated with organic compounds which impart specific…
Q: Acid-catalyzed bromination of pentan-2-one (CH3COCH2CH2CH3) forms two products: BrCH2COCH2CH2CH3…
A: In the presence of an acid, the tautomerization of ketone takes place in two ways as shown in the…
Q: Which of the following groups would have the HIGHEST priority using the Chan-Ingold-Prelog System?…
A:
Q: Arrange the following compounds in their ease of undergoing an SN2 reaction: a) CH3CH2CH2-CI b)…
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: To which compound is the addition of HBr more highly regioselective?
A:
Q: Which is more stable nh2ch2ch2+ or ch3ch2ch2+?
A: stability of carbocation : The electron releasing group stabilize the carbocation .
Q: Be sure to answer all parts. In the following pair of compounds one is chiral and the other is…
A:
Q: How useful is the (R) or (S) designation for predicting the sign of an optical rotation? Can you…
A: Here, un this question, we have to discuss wheather the R/S -nomenclature and the sign of optical…
Q: In RPLC, Compound A is more polar than compound B. In which column will compound A be more retained…
A: To answer: In RPLC, Compound A is more polar than compound B. In which column will compound A be…
Q: Q6) (use a Newman or Saw Horse projection) the stereochemical requirements for the E2 elimination…
A:
Q: What alkyl halide will be obtained in greatest yield? Ignore stereoisomers.
A:
Q: Which alkyl halide from the following pair is (i) Chiral and (ii) undergoes SN1 reaction faster?(a)…
A: SN1 is the type of nucleophilic substitution reaction which proceeds via formation of carbocation.…
Q: 1. NaOCH3 2. Br- Br 3. NaOCH3 4. LIAIHĄ 5. Н.О 6. , H3O* (cat.)
A: Dimethyl malonate reacts with base.
Q: Assign R and S to the asymmetric carbon product below (in numerical order). 5 6 3 Br 2.
A: According to the question, we need to determine the configuration of symmetric carbon atoms in the…
Q: 1. Draw the reaction mechanism for the hydroboration step of the reaction and explain why the…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing…
A:
Q: which newman projection displays the proper antiperiplanar orientation for an E2 elimination of the…
A: In an E2 eliminations, the new π bond is formed by overlap of the C-H σ bond with the C-X σ*…
Q: Br CH3 Br2 CH3CHCHCH2ČHCH3 Br OH 1. Hg(OAc)2, H20 2. NABH4 CH3CH2CH2CHCH3 HCI CH3CHCHCH2CH2CH2CH3…
A: Organic reaction mechanisms. As per our guideline we have to answer first three questions only.
Q: Which reagent could be used to distinguish between CH3C(CH3)2CH2CHO and CH3COCH2CH2CH2CH3 O NaOl (12…
A:
Q: The first step in assigning configuration is to assign the priorities to the groups attached to the…
A: Please find your solution below : The priority is assigned on the basis of atomic and molecular…
Q: Arrange the following groups in the order of increasing priority (lowest priority to highest…
A: Polar covalent bond:The covalent bond is formed by the sharing of electrons between the atoms. If…
Q: Chemistry Given the three-dimensional structure of the compound below determine the product of an E2…
A: Stereochemistry is branch of chemistry in which we deal with three dimensional arrangement of atoms…
Q: 11) which molecule below has S-configuration? A B D
A: We'll answer the first question since the exact one wasn't specified. Please submit a new question…
Q: Rank the following in order of increasing reactivity toward electrophilic substitution (start with…
A: Electrophilic substitution will occur in benzene rings whose electron density is increased by the…
Q: Use retrosynthetic analysis to determine the three starting materials of the shown compound. All…
A: The correct answer about retro synthesis is given below
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 6 images

- The CH3 group is considered a .... Ortho/Para Directing Activator Ortho/Para Directing Deactivator Meta Directing DeactivatorIdentify compounds A and B, two synthetic intermediates in the 1979 synthesis of the plant growth hormone gibberellic acid by Corey and Smith. Gibberellic acid induces cell division and elongation, thus making plants tall and leaves large.This is Wittig Rxn: Whatever mechanism you choose to draw is fine since you can leave the base as B: Draw the arrow pushing mechanism using the compounds below – Constant: 4-nitrobenzyl benzaldehydebase 1: triethylaminebase 2: NaOHbase 3: K2CO3ylide: Acetonyltriphenylphosphonium chloride
- Draw the products formed when attached dihalide is treated with excess NaNH2.The cyano (-CN) group is considered a .... Ortho/Para Directing Activator Ortho/Para Directing Deactivator Meta Directing DeactivatorDraw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a synthesis of the steroid cortisone by R. B. Woodward and co-workers at Harvard University in 1951.
- Provide the reagents necessary to achieve each transformation. Draw the intermediate structure(s).(R)-Carvone, the major component of the oil of spearmint, undergoesacid-catalyzed isomerization to carvacrol, a major component of the oilof thyme. Draw a stepwise mechanism and explain why thisisomerization occurs.Draw the Mechanism of Ether Cleavage in Strong Acid—(CH3)3COCH3 + HI → (CH3)3CI + CH3I + H2O
- The treatment of isoprene [CH2=C(CH3)CH=CH2] with one equivalent ofmCPBA forms A as the major product. A gives a molecular ion at 84 in itsmass spectrum, and peaks at 2850–3150 cm−1 in its IR spectrum. The 1HNMR spectrum of A is given below. What is the structure of A?The reaction below is homogenic homolysis heterolysis heterogenicIllustrate the Dehydration of a 1° ROH—An E2 Mechanism ?

