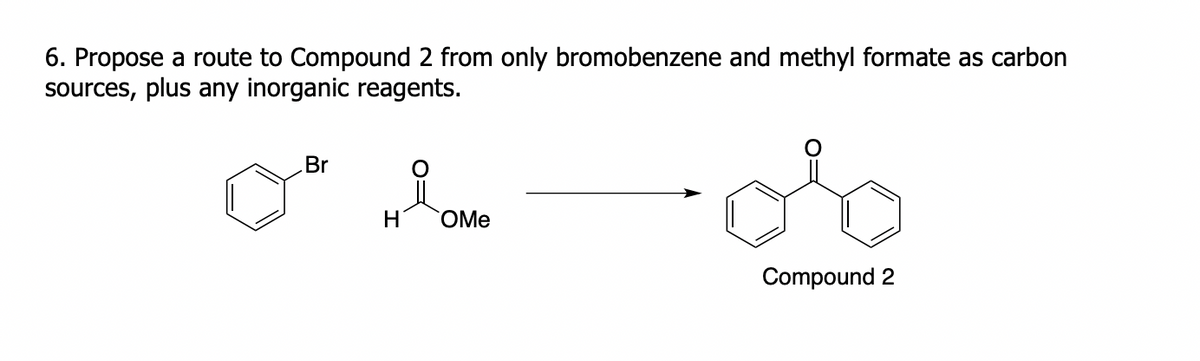
6. Propose a route to Compound 2 from only bromobenzene and methyl formate as carbon sources, plus any inorganic reagents. of Br H OMe Compound 2
Q: Wine goes bad soon after opening because the ethanol (CH,CH,OH) in it reacts with oxygen gas (0,) fr...
A:
Q: In a flask containing the reaction, PCI3(g) + Cl2(g) = PCI5(g), equilibrium concentrations were foun...
A: Recall the reaction, PCl3(g) +Cl2(g) ⇔PCl5(g) Kc=4.6*104 then,
Q: What volume of 0.5 M NaOH is required to react completely with 25.0 mL of 1.0 M H2CO3? Assume the r...
A: Given, Note: 1000 mL = 1 L Molarity of NaOH solution = 0.5 M = 0.5 mol/L Volume of NaOH solution = ?...
Q: This is the chemical formula for methyl tert-butyl ether (the clean-fuel gasoline additive MTBE): CH...
A: The chemical formula for methyl tert-butyl ether :- CH3OC(CH3)3 Moles of C in methyl tert-butyl et...
Q: Calculate the distribution ratio (organic/aqueous ) of HA.
A: Distribution ratio is defined as the ratio of solute distributed between two solvents. ...
Q: The decomposition of nitrogen dioxide gas (NO2) to nitrogen monoxide (NO) and oxygen(O2) gases is a ...
A: Here decomposition of NO2 to NO and O2 gases is a second order chemical reaction. The initial concen...
Q: 1.0 x 10-11 moles of HCl are added to 1.OL of H20. What is the pH? О 3.0 O 10.0 O 11.0 О 1.0 O 7.0
A: Moles of HCl = 1.0 × 10-11 moles Volume of water = 1.0 Liter
Q: Phosphorus trichloride and chlorine gas react to form phosphorus pentachloride: PCh (g) + Cl, (R) O ...
A: Using ICE table PCl3 Cl2 PCl5 Initial 1.0 M 2.0 M 0 Change x x x Equilibrium 1.0 -x 2...
Q: If K, for HCN is 6.2 x 10-10, what is K, for CN ? O 6.2 x 104 O 6.2 x 10-10 O none of these O 6.2 x ...
A:
Q: Draw the starting alkyne that would lead to this major product under these conditions. Select to Dra...
A: Alkynes undergoes oxymercuration to form ketone
Q: (b) What volume of 0.10 moldm³ sulphuric acid would be required to neutralise a mixture of 1.06 g of...
A:
Q: Why does the presence of solute in a solution lower its freezing point?
A: Freezing point is the temperature at which a liquid freezes and the solid and liquid phases are in e...
Q: 3. What is the molar concentration of a solution that is 28.15% HNO, (w/w) (MM= 63.01 g/mol)? The sp...
A:
Q: Pure benzoic acid melts at 122 °C and its heat of fusion is 147.34 J g. If the corrected fractions m...
A: When there is an impurity present in a compound, it will decrease the melting point of that compound...
Q: What is the name of the non-reducing monosaccharide found in the disaccharide shown here? но H. H. Н...
A: Nonreducing disaccharides can be defined as the disaccharides having glycosidic bonds between their ...
Q: is question is based on the five compounds (A-E) given CH;-CH=CH-CH3 ) CH;-C(CI)(CH;)-CH2-CH3 ) CHCI...
A: Esters have fruity smell or aroma due to their volatile nature caused by their chemical composition.
Q: Calculate AH° and AE for the following reactions at 25 °C. Use standard enthalpies of formation from...
A:
Q: 2. Calculate the solvation free energy AG of Mg" using the Born theory. Given that the ionic radius ...
A: When a cation is dissolved in a particular solvent (mostly polar solvent), the cation gets surrounde...
Q: ections: Balance the chemical equation below and complete its mole ratio on the table below. _C3H8 C...
A: C3H8 + 5O2 ----> 3CO2 + 4H2O
Q: 4) The decomposition of phosphorus pentachloride (PCIS) into phosphorus trichloride (PCI:) and chlor...
A:
Q: O The conjugate base of a strong acid will significantly raise the pH of water O For a polyprotic ac...
A: The correct statement is :
Q: A mixture of helium and neon gas is expanded from a volume of 83.0 L to a volume of 87.0 L, while th...
A: Initial volume (V1)= 83.0 L Final volume (V2) = 87.0 L Pressure (P) = 64.0 atm
Q: Aqueous sulfuric acid (H, SO,) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium s...
A:
Q: Charles's Law 200 00 025 ノ-00 1-25 -50 Volume (liters) B Write an equatiun f the line Shoun in the a...
A: Charles law states that at constant pressure, volume is directly proportional to temperature. Let us...
Q: Consider the following equilibrium with K = 2.3 x 10-6: N2 (3) + 3H2 3) + 2NH3 (§) 0.1 moles of N2, ...
A:
Q: Only one of the following changes will shift the equilibrium of this reaction left, favoring reactan...
A:
Q: What is the calculated value of the cell potential at 298K for an electrochemical cell with the foll...
A:
Q: (ii) Calculate the enthalpy change for the dissolution of (NH4)2SO4(s) using the enthalpy of formati...
A:
Q: Match the following enthalpy changes with the reactions below: enthalpy change of combustion, enthal...
A: These reactions are called-
Q: quid octane (CH3(CH,) CH;) reacts with gaseous oxygen gas (02) to produce gaseous carbon dioxide (CO...
A:
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium b...
A:
Q: 1) HCI (1 eq.) F) Br CI 2) HBr (1 eq.) Major Minor Br G) EIOH Diene Dienophile CO,Me H) Нeat CO,Me A...
A:
Q: Explain why the following reaction is endothermic based on your knowledge of both bond enthalpy and ...
A:
Q: SUMMA ASS: Phosphorous trichloride and phosphorous pentachloride equilibrate in the presence of mole...
A: Given , Reaction : PCl3(g) + Cl2(g) → PCl5(g). The equilibrium concentration : PPCl3=0.202 atmPCl2=...
Q: When the entropy change of the surroundings is negative, the enthalpy change for system O will be ne...
A: A multiple choice question based on chemical thermodynamics that is to be accomplished.
Q: The decomposition of phosphene (COCI,) gas into carbon monoxide (CO) and chlorine (Cl2) gases is ver...
A: Equilibrium constant ( Kp ) is the ratio of partial pressures of products to partial pressures of re...
Q: 2- Calculate the final pressure of a sample of gas that is changed at constant temperature to 14.3 L...
A:
Q: Consider the following balanced chemical equation: H2O2 (aq) + 3 1 (aq) + 2 H* (aq) →I; (aq) + 2 H20...
A:
Q: Use MO theory to explain the following: O2P TT 2p 2р, 2pх 2ру 2р, 2рх 2ру 2р, 2рx 2ру 2р, 2рх 2ру T ...
A: Ans-i. The valence electron of fluorine molecule is reside in 2p energy orbital, but in difluorine m...
Q: T:E3 → E2
A: In this question we have to prove if the Ker T where T:E3 → E2 by T((x1,x2,x3)) = (x1 + x2, x2 - x3)...
Q: The dye has a percent composition of 75.95% C, 17.72% N, and 6.33% H by mass with a molar mass of ab...
A: % of C = 75.95 % of N = 17.72 % of H = 6.33 Molar mass of dye = 240 g/mol The question requires to...
Q: Calculate the difference, in kilojoules, between AE and AH for the following exothermic reaction at ...
A:
Q: Nitrogen (N,) gas and hydrogen (H,) gas react to form ammonia (NH,) gas. Suppose you have 5.0 mol of...
A:
Q: UV - spectra of two Bomers of cinmamic auld g dentify the is amer 2016|| The absorb at 272 nm & 268 ...
A: This Problem Is Based upon UV Spectroscopy, and it can be solved by the concept of steric hindrance ...
Q: Why do you feel thirsty after playing or working outside? Sometimes, you suffer from cramps or heada...
A:
Q: Constants 12 C he lowng S rencti nd coreponding equilibenam constants Roaction AK-1) Reaction B(K-9....
A:
Q: is question is based on the five compounds (A-E) given below. CH;-CH=CH, CH;-CHBR-CH2-CH; cyclopropa...
A: Organic chemistry classification: As per our guideline we have to answer first question only.
Q: 1. A 55.0 ml sample of 0.102 M potassium sulfate is mixed with excess lead (II) acetate to produce p...
A: Potassium sulfate reacts with lead(II) acetate to form potassium acetate and lead(II) sulfate. The e...
Q: Calculate the mass of glucose (C,H,20,) that contains a billion (1.00 x 10') hydrogen atoms. Be sure...
A: No. of H atoms = 1.00×109 atoms mass of glucose = ? molar mass of glucose = 180 g mol-1 #of H in glu...
Q: If we are expecting about 48 g of citric acid per L of lemon juice, how many moles of citric acid sh...
A: Given that we expect 48 g of citric acid per L of lemon juice. We have to determine the moles of cit...
Step by step
Solved in 2 steps with 1 images

- Which compounds will give positive iodoform tests?(a) 1-phenylethanol (b) pentan-2-oneWhich reagents would you use to perform reaction B?a) PBr3.b) p-toluenesulfonyl chloride.c) SOCl2.d) NaOCl, NaOHwhy is 12 M HCl added to the reaction mixture after the Williamson Ether Synthesis reaction was complete. (choose all that apply)
- What is reaction 1 and 2 called? Choices: A. Williamson ether synthesis, B. Reduction with Grignard reagent, C. Acidic ether cleavage, D. FC Alkylation What are the reagents for reaction 1 and 2? Choices: A. methanol in acidic medium, B. sodium hydride and bromomethane, C. methyl bromide and aluminum bromide, D. methanoyl bromide1-Complete the reaction in the first picture. 2-Show the synthesis of ethyl propyl ether by using suitable reagents via Williamson ether synthesis?.Show the 2 reactions including starting materials and reagents of this compound
- 1. Based on the theoretical result, what is the order of reactivity of primary, secondary, and tertiary alkyl halides with sodium iodide in acetone? Explain. 2. Compare SN1 (Sodium Iodide in Acetone Reagent) and SN2 (Silver Nitrate Test) Reactions of Alkyl HalidesIf the structure below is subjected to retrosynthetic analysis, which reagent is least useful?Fill in the missing reagents below.
- For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).Reaction with Alcohols (esterification)a. Mix 1 mL of glacial acetic acid, 1 mL of ethyl alcohol and 3 drops ofconcentrated sulfuric acid (CAUTION!). Cautiously note the odor.Result: ___________________________________________________________________To what compound is the odor due? ________________________________________Equation: _________________________________________________________________b. To about 0.1 gm of salicylic acid in a test tube, add 1 mL of methyl alcoholand 3 drops of concentrated sulfuric acid (CAUTION!). Heat the mixture gentlyfor 1 – 2 minutes, then pour the mixture into about 20 mL of cold watercontained in a beaker. Observe the odor of the product formed. _____________What compound produces this odor? ______________________________________Equation: ______________________________________________________________1.Show the Elimination reaction of 2-bromopentane. 2.What contributed to formation of minor and major product?

