Q: Consider the tetrasaccharide stachyose drawn below. Stachyose isfound in white jasmine, soybeans,…
A: Stachyose is a polyhydroxy carbohydrate compound. It is not present in human edible foods .
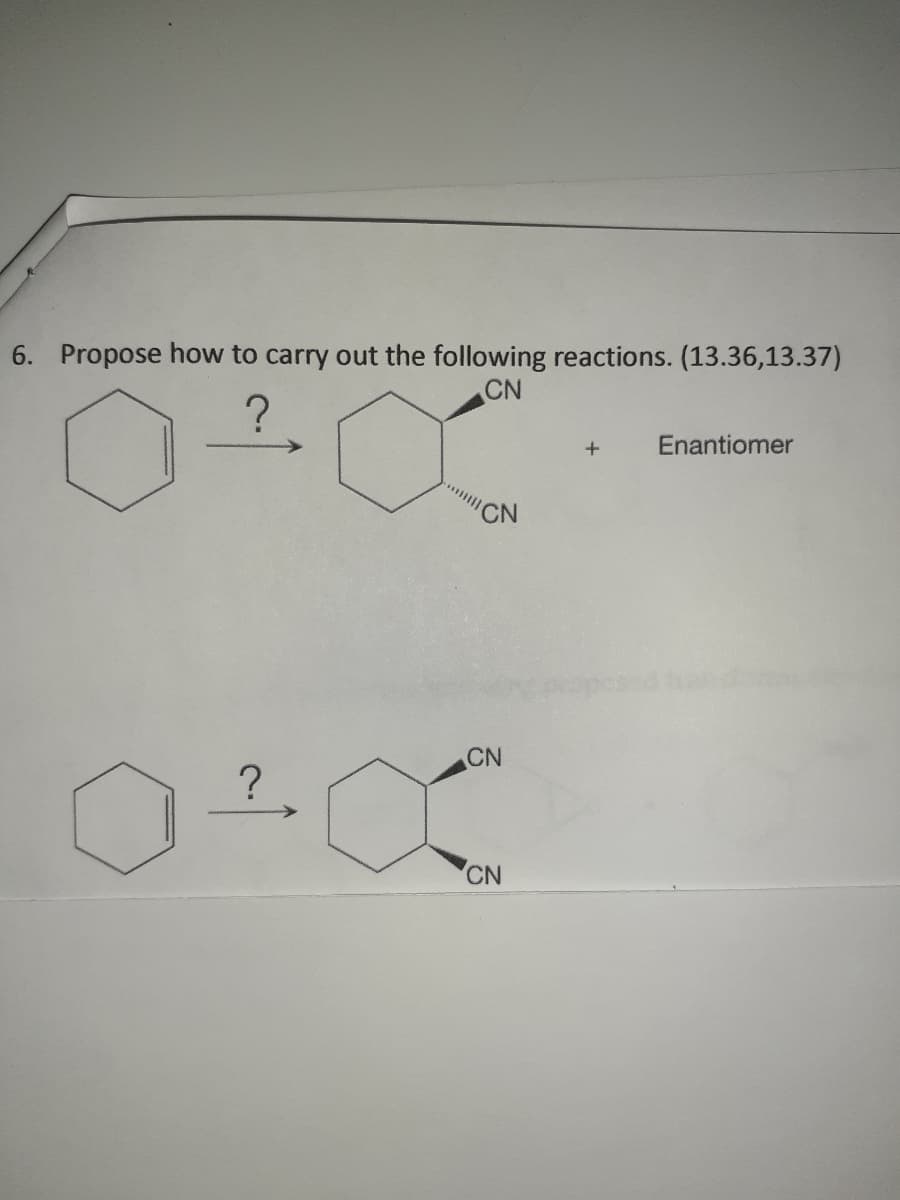
Q: What is the ee for each of the following mixtures of enantiomers A and B? 95% A and 5% B
A: The structure of chemical compound can be represented as structural formula. A structural formula…
Q: a.Label the four stereogenic centers in sorbitol as R or S. b.How are sorbitol and A related? c. How…
A: A carbon atom that has four different atoms or groups bonded to it is known as a chiral carbon and…
Q: Calculate both saponification and iodine value? Fatty acid Molecular Formula 16:0 C16H32O2 13.2 16:1…
A: The saponification value is the number of milligrams of potassium hydroxide required to neutralize…
Q: 8. Consic structure s
A: GivenH2NCH2CH2COOHLewis structure = To be determined Hybridisation = To be determined
Q: b) How many reducing and non reducing does O- a-D-glucopyranofil(l ➔2)BETA -D-fructofuranoside has?…
A: Introduction: We have to tell number of reducing and non-reducing sugars in…
Q: (A) Identify the glycosidic bond in the following disaccharide (B) Decide whether the compound is a…
A: Reducing sugar are those sugar which can reduces mild oxidising agent like Tollens reagent, Fehling…
Q: In the structure of Simvastatin as shown below, determine the configuration of the carbon atoms with…
A: Assign priority (1-4) of the stereocentre of the carbon.
Q: Trabectedin, shown in a ball-and-stick model on the cover of this text, is an anticancer drug sold…
A: Since you have posted multiple questions, and have not specified which question you want us to…
Q: I. a) Draw the structure of the products that form upon complete hydrolysis (both aq HCl and aq NaOH…
A:
Q: The following trisaccharide derivative is important to human health. The A ring is ?-deoxy? но но-…
A: In numbering of cyclic ether start numbering from that side where substituent is near.
Q: 5.69 Carvone is present in many essential oils. It is chiral, and its two enantiomers are shown…
A: carvone is a chiral molecule; its enantiomers have different physical and chemical properties.The…
Q: Nutmeg contains a simple, fully saturated triacylglycerol with a molecular weight of 722. Draw its…
A: The myristicin is a main psychoactive constituent of nutmeg. It is also a major component found in…
Q: If a mixture has an ee of 90%, what is the percentage of each enantiomer? Multiple Choice 80% : 20%…
A: Explanation: The enantiomeric excess (ee) shows how much more of one enantiomer is present in the…
Q: Draw the linear form, Haworth formula and chair conformation(if applicable) for the two anomeric…
A: We know that the pyranose form is a six membered ring having oxygen as a hetero atom while furanose…
Q: Explain the method to separate enantiomers ?
A: Chiral compounds which are mirror images of each other and are not superimposable are enantiomers.…
Q: The [α] of pure quinine, an antimalarial drug, is −165. a.Calculate the ee of a solution with the…
A: a). The specific rotation [α] of pure quinine is -165. [α] value for the solution is -50 The…
Q: (a) Draw the more stable chair form of fucose, an essential monosaccharide needed in the diet and a…
A: The Fisher projection is drawn by converting the stable staggered conformation into less stable…
Q: 3.30 Are the formulas within each set identical, enantiomers, or diastereomers? но н and (b) and H…
A:
Q: Spongothymidine is an N-glycoside isolated from Tectitethya crypta, a shallow-water Caribbean…
A: SOLUTION: Step 1: The monosaccharide present in spongothymidine is D- arabinose (β-D-arabino…
Q: B. Consider the structure of the amino acid D-serine: COOH H. -NH₂ CH₂OH BIAL 1. 2. Draw the Fischer…
A: As per our guideline we are supposed to solve one question at a time. please resubmit the remaining…
Q: 10.51 Draw all stereoisomers formed in each reaction. b. Cl2 H20
A:
Q: Monosaccharide A is a diastereomer of d-lyxose. Treatment of A with nitric acid forms an optically…
A: First, the D-lyxose is shown as below by using Fischer projection. For given compound the structure…
Q: Captopril is a drug used to treat high blood pressure and congestiveheart failure.Designate each…
A: Chiral center: A carbon atom attached with 4 different atoms or groups To find R and S…
Q: an be separatd using GC why can nesc two compounds separae in Ġ C and Ot by a fractional…
A: Seperating Cis and trans isomers by fractional distillation is not possible because they are having…
Q: Threohine is a naturally occurring amino acid used in the pro ction of proteins in the body. Draw…
A: We have to draw the Enantiomer of given threonine molecule
Q: QUESTION 31 If a mixture has an ee of 90%, what is the percentage of each enantiomer? O 65% : 35%…
A:
Q: 3.3 Draw line structures of (a) an enantiomer and (b) a diastereomer of Compound P. NH2 HO HO…
A: Enantiomers are non-superimposable mirror images isomer whereas diastereoisomers are not mirror…
Q: Draw all possible constitutional isomers of a triacylglycerol formed from one mole each of palmitic,…
A: Triacylglycerols (triglycerides) are the lipids formed by the reaction of glycerol and fatty acids.
Q: Captopril is a drug used to treat high blood pressure and congestiveheart failure.Draw the…
A: Given,
Q: Refer to the following disaccharide to answer the questions. OH OH H H OH OH Identify the type of…
A:
Q: 1. D-idose at C4 ( alpha anomer ) 2. L- galactose at C5 ( beta anomer ) 3. D-altrose at C4 ( alpha…
A:
Q: d) Draw the structure of the expected product when monosaccharide C undergo mutarotation upon…
A:
Q: What is the percent enantiomeric excess (ee) of a mixture that has 86% of one enantiomer and 14% of…
A: Given information, A mixture that has 86% of one enantiomer and 14% of the other.
Q: 7. Lactose octaacetate What can you conclude about the conformation across the glycosidic linkage?
A: Lactose is a disaccharide made up of glucose and galactose. A glycosidic bond is a bond (covalent…
Q: When heated with Benedict's reagent, one dairy product (with no additives) turns bright red; as…
A: Benedict's test is used to test for the presence of monosaccharides and reducing disaccharides…
Q: 8. What is the relationship between these two molecules? vs A.Enantiomers B. Identical C.…
A: The molecules with the same molecular formula but different structures are known as isomers. If the…
Q: Indicate the correct correlation between the foOllowing pairs of molecules. Write tne appropriate…
A:
Q: a. Draw a three-dimensional structure for the following steroid. b. What is the structure of the…
A: a. In 3D structure, each color represent a specific atom and each line represents each bond present…
Q: 2. 1.056g of a compound X is dissolved in 2.5g water (K 1.86°C/molal) and the solution has a free…
A: Given: Mass of solute X = 1.056 gMass of solvent (water) = 2.5 gKf = 1.86°C/molalTf (final) =…
Q: The [α] of pure quinine, an antimalarial drug, is −165.Calculate the ee of a solution with the…
A: The formula for enantiomer excess of a solution is calculated as follows: ee=αmixtureαPure…
Q: What is the ee for each of the following mixtures of enantiomers A and B? 85% A and 15% B
A: The enantiomeric excess explains the excess of one enantiomer over another in a given mixture. Thus,…
Q: Raffinose is a trisaccharide (found in sugar beets) containing D-galactose ,D-glucose ,and…
A: Given information: Raffinose is a trisaccharide and made of three units such as galactose, glucose…
Q: Monosaccharide Structure and Stereochemistry For D-arabinose: Draw a constitutional isomer that…
A: Two compounds are considered as constitutional isomers if they have same molecular formula but…
Q: Specity elationsh ionsnip between these two molecules CI Cl, diastereomers, O enantiomers,
A: Applying concept of R,S configuration of organic molecules.
Q: (b) The optical rotation of a- and B-anomers of D-glucose are [a]p = +112.2° and [a]p = +18.7°, %3D…
A:
Q: (c) Draw a three-dimensional line structure of the Fischer projection (a) using the template (c)…
A: The Given structures are of monosaccharides. They can be structured in Normal carbohydrate chain…
Step by step
Solved in 2 steps with 1 images

- Chlorhexidine gluconate is available in different products in concentrations of 4% w/vand 0.12% w/v. H ow many milliliters of the more dilute product may be prepared fromeach fluidounce of the more concentrated product?A sunscreen contains 6% oxybenzone and 3% avobenzone bymass. If a bottle contains 40. g of sunscreen, how many kilogramsof oxybenzone and avobenzone are needed to manufacture1 ton of sunscreen? How many bottles of sunscreen can beproduced for 1 ton of sunscreen?The pKb of codeine is 5.8. Calculate the percentage of drug in unionized form in (a) plasma at pH 7.4 and (b) pH 6.
- C6H8O6(aq) + C12H7O2NCL2(aq) ----> C6H6O6(aq) + C12H9O2NCL2(aq) - C12H7O2NCL2 is abbreviated as DCIP - Molar Lass of C6H8O6(s) is 176.14 g/mol - Daily recommended intake (DRI) for adults: 90 mg of Vitamin C Askor & Bich found that it required 23.14 mlof 1.32 x 10^-3 M DCIP soliution to reach the endpoint when titrating a 7.50ml aliquot of pear juice. How many mg of ascorbic acid, C6H8O6 are present in the juice aliquot? _5.38_____(can you please show how to calculate this answer) How many ml of this juice would Bich have to drink in order to meet the daily recommended intake (DRI)?__126__ (During exercise when the body lacks an adequate supply of oxygen to support energy production, the pyruvate that is produced from the breakdown of glucose is converted into lactate. High lactate levels can lead to acidity in the muscle cells as some of the lactate hydrolyzes to lactic acid. The dissociation of lactic acid to lactate is shown in the reaction. Lactic acid has a p?apKa of 3.86.3.86. A solution containing a mixture of lactic acid and lactate was found to have a pH of 3.24.3.24. Calculate the ratio of the lactate concentration to the lactic acid concentration in this solution.An unkown solvent has a formula of C4H8O and molecular weight of 72.11 g/mol. Determine the ff: a.) index of hydrohgen deficiency b.) all possible functional group present base on IHD
- What is the (a) molecular weight of glutathione (C10H17N3O6S)? If 30 grams of glutathione is dissolved in a solution, what is the: (b) number of moles of Glutathione, (c) millimoles of N, (d) number of O atoms, and (e) grams of H.Eq 7In extraction of caffeine from tea, CaCO3 (pKa of HCO3- is 10.3) is used to convert tannin to glucose and calcium salt of gallic acid. Explain why glucose is not in its salt form but gallic acid is. Assuming pKa of -OH of glucose is 12, and pKa of -OH of gallic acid is 9.5 and pKa of -COOH of gallic acid is 4.5.

