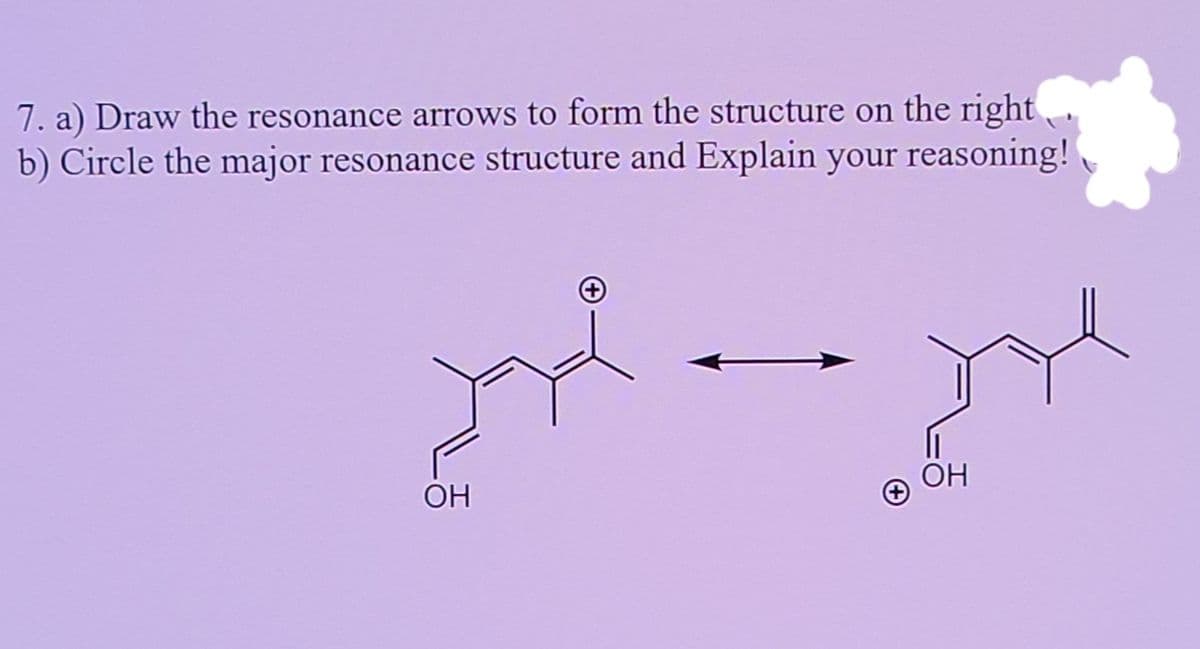
Q: Use curved arrow notation to show how the first resonance structure can be converted to the second.
A:
Q: Follow the curved arrows to draw a second resonance structure for each species. a. H-C=0: b. CH-…
A: (a) The resonance structure for given aldehyde is drawn in step two.
Q: a. Which of the following has a greater electronegativity? (circle one) Carbon / Cesium b. Explain…
A: Electronegativity is the tendency of an atom in a molecule to draw the shared pair of electrons…
Q: Follow the curved arrows to draw a second resonance structure for eachspecies.
A: Given species,
Q: Draw all reasonable resonance structures for attached compound.
A: Given,
Q: Draw two more resonance structures for each species. a. b.
A: There are different set of structures in which the position of atoms are same but position of…
Q: 9. Draw all resonance structures of the following compounds, showing electron movement with curved…
A:
Q: 3. Draw all resonance structures for the following radicals.
A: Geometry of free radicals - ' shallow pyramid' Partially filled p orbital can participate in…
Q: In Naproxen, is the lone pair of the oxygen atom delocalize? I’m other words, can the oxygen…
A: In Naproxen, is the lone pair of the oxygen atom delocalize? In other words, can the oxygen…
Q: HN ÓH
A: The two resonating structures will be formed as follows:
Q: Follow the curved arrows to draw a second resonance structure for each ion. a. CH C-CH, b. H-E-c-CH3…
A: The second resonance structure of (a) can be drawn as follows:
Q: 2) Draw two more resonance structures for the following:
A: Resonating structure
Q: why is ethane a nonpolar covalent bond? I get that it is because they have "equal" shared valence…
A: Ethane (C2H6 ) is made of Carbon(C) and Hydrogen(H).
Q: 2. Draw a resonance structure for the compound below.
A: The solution is as follows :
Q: Draw the curved arrows and the resulting resonance structure for the following structure. Include…
A:
Q: 4. Circle the following pairs of structures that do not constitute resonance structures. For proper…
A: Molecule participate in resonance by delocalization of π electrons with lone pair of electrons in…
Q: Follow the curved arrows to draw a second resonance structure for the following species. CH,-C-CH'…
A: The resonance structure given along with the movement of electrons as,
Q: Draw a second resonance form for the structure shown below. :o:
A:
Q: Use curved arrow notation to show how the rst resonance structure can be converted to the second.
A: Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure.…
Q: Identify the resonance structure that is considered the major contributor to the resonance hybrid
A:
Q: Draw four additional resonance structures for the following cation. Then draw the resonance hybrid +
A: The four resonance structures of the given cation are,
Q: This is an organic chem question. draw 2 additional resonance structures for each part a and b
A:
Q: Draw four additional resonance structures for the following cation. Then draw the resonance hybrid.
A: The resonance structure is defined as those structures which are the lewis structures having the…
Q: Create a mnemonic on how to properly distinguish the types of chemical bonds
A:
Q: Draw a second resonance form for the structure shown below.
A: Resonance is a method of representation of bonds in various molecules or ions. Resonance structure…
Q: Original structure is also included in the total number of resonance structures. Draw out bonds to…
A: Resonance structures are generally described as the set of lewis structures which consists of the…
Q: Compare columns I and II, and write A if the value in column I is greater than in II B if the value…
A: The compounds given are Column I: 1H-phenalene Column II: 1,4-dihydrophenanthrene
Q: 4. Draw three possible resonance structures of the following molecule.
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: a Draw the additional resonance structure(s) of the structure below? :o: -c-: • Include all valence…
A: Facts about Resonance: 1. Resonance structures are a set of two or more Lewis Structures that…
Q: ▾ Part A Draw the resonance structure of the following substance. Interactive 3D display mode H3C…
A: Resonance structures represents a set of Lewis structures which shows the delocalization of…
Q: Explain the Below fact ? A “better” resonance structure is one that has more bonds and fewercharges.
A: If a molecule can be represented by two or more electronic structures, each of which cannot explain…
Q: PLEASE Answer all subquestions 1) Draw resonance structures AND resonance hybrid for following…
A:
Q: 1. Draw all resonance structures for the following molecules. CF3 a. b.
A: Resonance is defined as transfer of charge from one atom to another atom
Q: Resonance structure B is a A. major (more important) resonance structure. B. minor (less important)…
A: Solution Resonance could be a technique of describing the delocalized electrons in some molecules…
Q: For this molecule draw the 2 best resonance structures in the boxes. Circle the best.
A: The lone pair of electrons of oxygen makes a double bond with the carbon atom. It eliminates the…
Q: Н.
A:
Q: Draw all reasonable resonance structures for attached compound.
A: Rsonance structure : Resonance means distribution of electron density & forms nearly same energy…
Q: Provide the two resonance structures of the molecules shown. Circle the best resonance structure out…
A: Stability of carbocations: primary < secondary < tertiary
Q: Draw all the resonance structures for periodate ion.
A:
Q: Draw the curved arrows and the resulting resonance structure for the following structure. Include…
A: we have to draw the resonance structure for the given molecule
Q: Draw all the resonance structures for the following molecule. :0:
A:
Q: Draw all possible resonance structures for NPO in your work. Then, assign formal charge to each atom…
A: The compound given is NPO.
Q: Draw two more resonance structures for attached species.
A: The given compound consists of two conjugated double bonds and a positive charge on the ring. Hence,…
Q: In the box on the right, draw the best resonance structure of the compound on the left. Draw…
A: The compound given is,
Q: Draw a second resonance form for the structure shown below. ö: ö:
A: Resonance structure are sets of Lewis structures that describe the de-localization of electrons in a…
Q: Please draw out the imporant resonance forms for the structures 1 and 2
A:
Q: Ige m gen Complete the resonance structure by drawing the bond formed by the movement of electrons…
A: acetanilide Acetanilide bromination reaction Br+ electrophile Bromination at para position most…
Q: draw all significant resonance structures
A: When all the properties of a molecule cannot be explained by a single structure , we draw more than…
Q: O Draw the resonance hybrid } mark appropriate dcharges electon nch N.
A:
Step by step
Solved in 3 steps with 3 images

- See attached image. For this molecule draw the 2 best resonance structures in the boxes. Circle the best.Why are the two resonance structures I circled in green incorrect? My professor's answer key is the picture with the red text.Draw the resonance structure that results from the arrow pushing scheme below:
- For each proposed set of resonance structures: a. (E) Add curved arrows (starting from left) to show how each successive r.s. was generated. b. Cross out any resonance structures that are NOT important, and explain your reasoning.A. Draw the major resonance structure, then draw curved arrows to show how this can be converted to the Lewis structure given. B. Thionitromethane is redrawn for you. Draw curved arrows to show how this can be converted to the resonance structure you drew in part A.How do I figure this out? Compound X is the reactant on the left.
- Draw out all possible resonance forms.Please draw a more stable resonance structure for the following molecule. Use a curved arrow to show how to transform the original structure to the new one and please specify charges.Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Write the resonance structure that would result from pushing the electrons as indicated by the curved arrow.Z

