Q: Consider a solution that is 2.0×102 M in Fe² and 1.4×102 M in Mg2+ (Kap for FeCO3 is 3.07 x 10-11…
A: Answer: For any sparingly soluble salt, precipitation begins when its ionic product becomes equal to…
Q: Be sure to answer all parts. Draw both the Syl and El products of the following reaction. The Syl…
A:
Q: Use the References to access important values if needed for this question. A 1.60 mol sample of CO₂…
A: Given that, Volume and Temperature are constant We know that, Expression for Kinetic Energy (K.E) =…
Q: & 21.7 Give the structure of the product. LiCu(Me)2
A: Organolithium reagents are organic compounds that contain a carbon-lithium bond (C-Li) and are…
Q: a lethal dose of sodium cyanide (NaCN ) is 1.52E_(21) molecules. How many grams of NaCN is this
A:
Q: 350. mg of an unknown protein are dissolved in enough solvent to make 5.00 mL of solution. The…
A:
Q: What is the correct IUPAC name for the compound shown here?
A: IUPAC nomenclature refers to the standardized system of naming chemical compounds established by the…
Q: rude Oil into its various fractions
A: The process of separating crude oil into its various components is a critical step in the production…
Q: 51.1 Write the cell reactions, electrode half-reactions, and Nernst equations for the following…
A: As per our honour code we can only provide 2-3 parts out of 6 and for the solutions of remaining…
Q: Would NaBH4 also work as a reagent in this reaction?
A: Answer: In this question we have to compare the strength of two reducing agents.
Q: [References] [Review Topics] a. Calculate the number of moles and mass of the positive ions present…
A:
Q: A student is studying the reaction: 2 CrO4 + 2 H3O Cr207 + 3 H2O If CrO4 is yellow and Cr207 is…
A: According to Le Chateliar's principle, the equilibrium shifts in a direction that nullifies the…
Q: What is the pH of 50ml of 00.100M NaoH and 40ml of 0.100M HcooH
A: HCOOH (formic acid) is a weak acid and NaOH (sodium hydroxide) is a strong base. When they react,…
Q: What minimum temperature is required to dissolve 24gof potassium nitrate in 40 g of water?
A: We will calculate the minimum temperature is required to dissolve 24gof potassium nitrate in 40 g of…
Q: For each of the salts on the left, match the salts on the right that can be compared directly, using…
A: as the given compound , calculate the Ksp for all compound and match first dissociate into ions ,…
Q: Suppose a 250. mL flask is filled with 1.8 mol of Cl₂, 1.0 mol of HCl and 1.1 mol of CCI. The…
A:
Q: 9. Write all steps of the mechanism that explains each of these reactions. Don't simply name the…
A: Both the reactions takes place through E1 mechanism because tertiary alkyl halides are involved in…
Q: 4+ Sn²+ (aq) + IO4 (aq) → Sn¹+ (aq) + (aq) Express your answer as a balanced net ionic equation.…
A: Part C: Sn2+ (aq) + IO4- (aq) → Sn4+ (aq) + I- (aq) Oxidation Half : Sn2+ (aq) → Sn4+ (aq) + 2e-…
Q: Two titrations are performed: - One with HCl against NaOH - Another with Crotonic acid…
A:
Q: Calculate the molar solubility for calcium phosphate Ca3(PO4)2. (Ksp is 2.07×10–33)
A: Let molar solubility is S
Q: Provide the key term that corresponds to each of the following definitions. (a) a procedure for…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What will be the major organic product from the dehydration of 2-pentanol in the presence of a…
A: Alcohols in the presence of strong acid and heat undergo dehydration to give alkenes. Alkene form…
Q: a) Which law/rule is violated when filling the energy levels in the diagram below? b) Explain the…
A:
Q: A student did not read the directions to the experiment properly and mixed up where to put the NaOH…
A: Acid base titration is a titration where the concentration of acid or base is determined by adding…
Q: Macmillan Learning Ne(g) effuses at a rate that is ratee ratech times that of Cl₂(g) under the same…
A:
Q: Predict the major organic product of the following reactions and determine whether the SN1/E1, or…
A: We have been given an organic reaction and we have been asked to predict the major organic product.…
Q: A 50.5 g sample of polystyrene is put into a calorimeter (see sketch at right) that contains 200.0 g…
A: Given: Mass of polystyrene = 50.5 g = mpTi of polystyrene = 85.4 ◦C = TpSpecific heat of polystyrene…
Q: Draw a titration curve for a strong acid titrated with a strong base. On the same axes draw a…
A: Answer: When acid is titrated against base then pH of the solution rises as acid gets neutralized…
Q: Which of the following statements is NOT part of Bohr's model of the hydrogen atom? the electron in…
A: #1) According to Bohr's Model , An atom consists of a small, heavy positively charged nucleus in the…
Q: At a certain temperature, Kc equals 1.4 × 102 for the reaction: 2 CO(g) + O2(g) 2 CO2(g). If…
A: Given number of moles of CO2 = 0.400 moles Given number of moles of O2 = 0.100 moles Given volume =…
Q: The Kc of the reaction below is 3.149x10 -4 at 351.0K. What is the Kp value for this reaction. The…
A: The relationship between Kp and Kc is => Kp = Kc (RT)Δn where, T is the temperature in kelvin,…
Q: Draw the structural form of Asparagrine as it transition from low pH to high pH. Indicate the net…
A: Asparagine is a non-essential amino acid that is found in many proteins. It is one of the 20 amino…
Q: Show a mechanism of each of the following hydrolysis A reactions. Pay careful attention to whether…
A:
Q: Which rule or principle states that orbitals of equal energy fill with one unpaired electron each…
A:
Q: A mixture of carbon monoxide, hydrogen, and methanol is at equilibrium. The balanced chemical…
A: Given, A mixture of carbon monoxide, hydrogen, and methanol is at equilibrium. • The balanced…
Q: H2COCH2 Insert the correct amount of each element from the chemical formula H C O_ H2COCH2 Insert…
A: chemical formula identifies each constituent element by its chemical symbol and indicates the…
Q: 15. Calculate the pH of a 0.10 M NH4CN solution 16. Calculate the pH of a solution prepared by…
A: The pH of a solution is a measure of its acidity or basicity. It is defined as the negative…
Q: State the name of each of the following reactions (acid/base, Lewis acid/base, resolutio…
A: Resolution : It is the separation of a racemic mixture into its respective enantiomers. Since…
Q: How did you go from this 0.0639T2.K=T2−293K to get −0.936T2.K=−293K? I'm not understanding how…
A:
Q: What is the maximum concentration of Ag+ that can be added to a 0.00650 M solution of Na₂CO3 before…
A: Answer: At the saturation point ionic product of silver carbonate will become equal to its…
Q: I need help with these problem...
A:
Q: Balancing Act! Balance this equation! (Equation number 1 of 10) Sm₂O3 + 10 Ba →>> - 1 C Sm + Check…
A: We know in a balanced chemical equation there is conservation of mass .
Q: AC3.2 1. Identify the acid, base, conjugate acid and conjugate base for each of the following. a)…
A: We have been given a question related to acid and base concept. We have been given three subparts…
Q: How many moles are in 5.69×1024 atoms of iodine?
A: Mole is equal to number of atom divide by avagadro number
Q: Nitroglycerin is a dangerous powerful explosive that violently decomposes when it is shaken or…
A:
Q: An isotope dilution experiment was performed to determine the chloride content in a given samples. 5…
A: Given An isotope dilution experiment was performed to determine the chloride content in a given…
Q: Titration of Acids and Bases Part B. Determination of the molar concentration of an unknown acid.…
A: To find out the molarity of the unknown acid solution, we need to use the dilution law. Using that,…
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A: Addition of HBr to alekene in presence of peroxide gives anti-markovnicov product. The major…
Q: A 50.8 g sample of brass, which has a specific heat capacity of 0.375 J-g -1.0C-1 ", is put into a…
A:
Q: What is the correct IUPAC name for the compound shown here? Br Br
A:
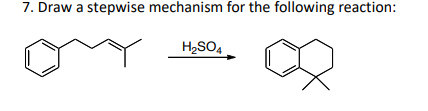
Step by step
Solved in 2 steps with 2 images

- Draw the products formed when A or B is treated with fo following agent. In some cases, no reaction occurs. [1] LiAlH4; [2] H2O#6r Daw a stepwise mechanism for the following reaction:Draw the products formed when A or B is treated with fo following agent. In some cases, no reaction occurs. [1] CH3MgBr (excess); [2] H2O
- Identify the reagent e, f, g and hBromoetherication, the addition of the elements of Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. This reaction has been used in the synthesis of the polyether antibiotic monensin (Problem 21.37). Draw a stepwise mechanism for the following intramolecular bromoetherication reaction.Which reactant in each of the following pairs is more nucleophilic? Explain. (a) -NH2 or NH3 (b) H2O or CH3CO2- (c) BF3 or F- (d) (CH3)3P or (CH3)3N (e) I- or Cl- (f) -C≡N or -OCH3



