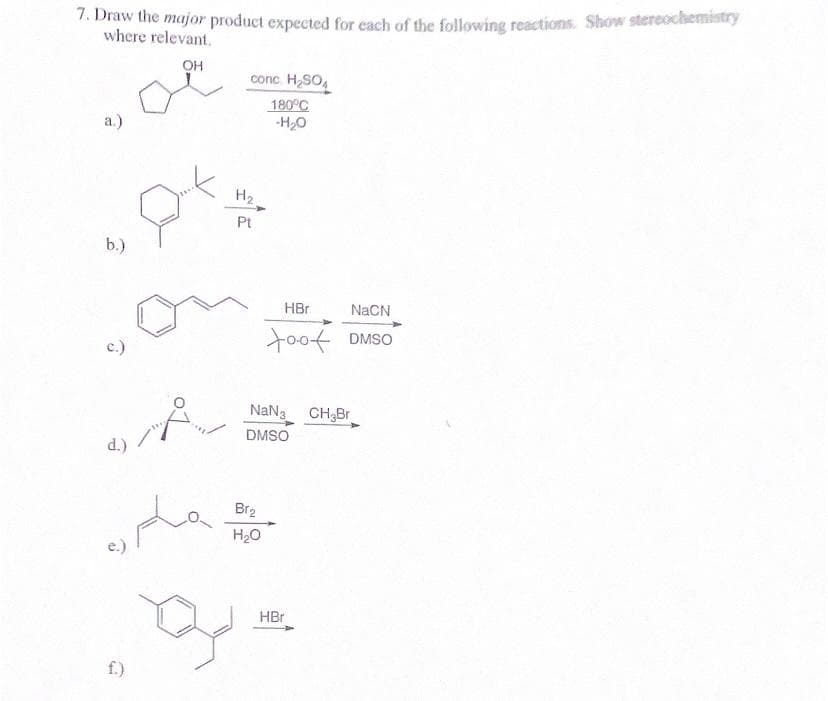
7. Draw the major product expected for each of the following reactions. Show stereochemistry where relevant. OH a.) b.) c.) conc. H₂SO4 180°C -H₂O H₂. Pt HBr NaCN too DMSO
Q: Determine the % of acetic acid (MM = 60.05 g/mol) in a 2.00 mL vinegar sample that has a mass of…
A: Given that, a 2.00 mL vinegar sample that has a mass of 2.10 g. A 16.36 mL of a 0.155 M NaOH…
Q: Calculate the value of Eºcell (in volts) for the following hypothetical reaction from data given in…
A: The given problem is based on the standard reduction potential values.
Q: Why does catalytic hydrogenation of alkenes proceed through syn addition? Provide a detailed…
A: The catalytic hydrogenation of alkenes proceed through syn addition due to the physical arrangement…
Q: 3c)Referring to the intermediates you drew in problem below explain in detail why no meta product is…
A:
Q: Combustion of hydrocarbons such as dodecane (C₁2H26) produces carbon dioxide, a "greenhouse gas."…
A:
Q: 2. Draw the product(s) of each reaction a. H₂C- b. H₂C C. H₂C= d. O₂N CH3 CH3 CH3 CH3 [1] 03 [2]…
A: chemical reaction is a process in which one or more substances known as the reactants are converted…
Q: What mass of ammonium sulfate, (NH4)2SO4 must be added to prepare 400.0 g of a 7.0% solution of…
A:
Q: Consider the structural formula with the name 2-butyne. Are there two geometrical isomers that exist…
A:
Q: The 'standard' maximum level for entry into a confined space is: O a. 0% LEL O b. 10% LEL O c. 50%…
A: Confined Spaces are inherently dangerous workplaces that exist in a variety of industries. An…
Q: Balance the equation in acidic conditions. Phases are optional. equation: H3 AsO3 + I₂ → H₂ AsO4 +…
A: we have to balance the given reaction in acidic solution
Q: Which metals will dissolve in HCI? Select one or more: Au(s) Ba(s) Sn(s) Cd(s) Ag(s)
A: Answer:- This question is answered by using the simple concept of chemical reactions of metals with…
Q: I. II. OH & & A ola NO₂ B OH B NO₂ CH3 of ob C III. Outline a method based on extraction techniques…
A: Extraction is a technique to separate components of a mixture. In this technique, a solvent is…
Q: 5. Both oxygen atoms in acetic acid (CH3CO₂H) are capable of hydrogen bonding, a feature that…
A: Hydrogen bonds are one of the intermolecular forces which are formed when a hydrogen atom bonded to…
Q: which of the following molecules have a lone pair electrons and/or non identical central bonds?…
A: Lone pair electrons - Electrons pair which do not take part in bond formation known as lone pair…
Q: formation of the C-O a-bonding molecular orbital? HÖ:/ Problem viewing the image. Click Here O…
A: Hybridisation is the concept of mixing atomic orbitals to form hybrid orbitals which are suitable…
Q: I. AHrxn for the reaction below was determined using bond dissociation energies (BDES) to be -57 kJ.…
A: Bond dissociation energy is calculated as the amount of energy required to disintegrate a chemical…
Q: Where this question came from it says its answer is actually B is this possible?
A: Disproportionation reaction is a reaction where a species having intermediate oxidation is converted…
Q: What is the pOH of a 0.033 MHNO3 solution? O 10.59 1.48 3.41 12.52 15.48 14
A:
Q: of Identify all symmetry elements and point group CHF3
A: The symmetry elements and point group of CHF3 are given below
Q: The following ionic solids have measured distances between their cations and anions. (1 Angstrom, a…
A: Lattice energy : It is the amount of energy required for the separation of constituent particles…
Q: How many chiral/asymmetric carbon(s) in the following structure? Problem viewing the image. Click…
A:
Q: Sodium hydrogen carbonate (NaHCO3), also known as sodium bicarbonate or "baking soda", can be used…
A:
Q: Which of the following steps is not part of structure determination by X-ray diffraction? A) Growing…
A: A common method for determining the crystallinity and structure of solid samples is X-ray…
Q: 8. Which of the following statements is the best explanation for the effect of increase in…
A: Chemical kinetics can be defined as the branch of chemistry that deals with rates of the chemical…
Q: What is the total concentration of hydrogen ion, H+, present in a water sample if 100. mL of the…
A: Question is about acid base neutralisation. If 8.1 ml of 2.1×10–3 M NaOH is needed to neutralize the…
Q: What is the pH of a 0.35 M solution of a Base with a Kb = 4.4 × 10–4 at 25oC? Identify the step(s)…
A: Concentration of base = 0.35 M Kb = 4.4 × 10-4 pH of solution = ?
Q: This reaction utilizes nucleophilic substitution reactions to get from the aziridine to the…
A: Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: Which of the following statements is incorrect? O One reason why HCI is a stronger acid than HF is…
A: We have to tell among the given statements, which are incorrect.
Q: Which of the following pairs of species is not a conjugate acid-base pair? O H₂CO3, HCO3- O 0²-, OH-…
A:
Q: Cr₂O7²- + S₂03²- → Cr³+ +S406²- when balanced in an acidic medium, would involve a total of process?…
A: Given that, a reaction is Cr2O72- + S2O32- → Cr3+ + S4O62-. We have to tell the total number of…
Q: In the titration of a weak monoprotic acid with a strong base, the pH of the titration solution…
A: pH is measure of acidity or basicity of a solution.If pH of solution is less than 7, then solution…
Q: The equilibrium pressure of hydrogen H2 over solid uranium U and solid uranium hydride UH3 at 500 K…
A: We have find out standard Gibbs energy of formation of UH3 (s) in the reaction: U(s) + 3/2 H2 (g) →…
Q: 1) The activation energy for the following first-order reaction is 102 kJ/mol. N₂O5(g) → 2NO2(g) +…
A:
Q: A solution contains 0.08 M acetic acid, HC₂H3O₂ (Kµ = 1.7 × 10¯5 at 25°C), and 0.15 M sodium a What…
A: Answer :-. Given data - A solution contain 0.08 M acetic acid ( Ka = 1.7× 10-5 at 25°C) And 0.15…
Q: Only specific amount of energy can be absorbed and emitted by an atom. Energy Photon E5 (n=5) E4…
A: Electron spectroscopy refers to a group which is based on the analysis of the energies of emitted…
Q: Draw the missing products or reagents in the following multistep synthesis. Ignore any inorganic…
A: We have to draw the missing products of the given reaction scheme. Introduction: Radical…
Q: HBr (1 equiv) H₂O₂ esc Select to Draw ! 1 O F1 143.177 XX @ 2 F2 # 3 DEC 31 6 80 F3 $ 4 Q F4 CI % 5…
A: Markovnikov rule, the hydrogen atom is attached to the carbon atom with the highest number of…
Q: Carbon-14 has a half-life of 5730 years and the radiocarbon activity of wood recently cut down is…
A:
Q: The heat of vaporization AH, of acetonitrile (CH3CN) is 29.8 kJ/mol. Calculate the change in entropy…
A:
Q: H₂SO4, heat
A: We have to give the major product of the below-given reaction Introduction: Electrophilic…
Q: Kindly answer the following questions below. You may use illustration to help explain your answer a.…
A:
Q: HCO3* Lewis Structure (with formal charges) Hybridization VSEPR Shape of each central atom Bond…
A: The concept used here is from chemical bonding of inorganic chemistry. Here we have to talk about…
Q: A model of an alkane appears in the window below. ball & stick ♥ - + labels Which of the following…
A: The compounds with the same molecular formula but different bond connectivity are structural…
Q: Determine the numbers of protons, neutrons, and electrons in each of the following species: (a)…
A:
Q: A 6.00 L tank at 14.5 °C is filled with 10.2 g of sulfur tetrafluoride gas and 5.04 g of carbon…
A:
Q: Which of the following qualifies as a state function? the change in potential energy when a book is…
A: State function: It is a property whose value depends only on the initial and final state only but…
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A: NBS undergoes homolytic cleavage to produce bromo radical. Bromo radical abstract benzylic proton to…
Q: HCO, Lewis Structure (with formal charges) Hybridization VSEPR Shape of each central atom Bond types…
A: We have to give the Lewis structure of HCO3-.
Q: Hydrogen sulfide (H2S)reacts with oxygen (O2) to produce sulfur dioxide (SO2) and water. How many…
A: It is an example of stoichiometric reaction Here we are required to find the mass of water produces…
Q: Provide a synthetic route for the following transformation.
A: Clemmensen reduction is used only for reducing aldehyde and ketones into respective alkane...
Step by step
Solved in 2 steps with 2 images

- Draw the major organic product of the reaction. Ignore stereochemistryInterestingly, reduction of the complex shown above mostly gives thealcohol enantiomer shown below after workup. a. Draw the other minor diastereomeric alcohol product. b. Briefly explain the diastereoselectivity of the reduction – why is oneenantiomer heavily favored? Draw a structure or structures to support youranswer.A. Provide the necessary reagents. B. Provide the necessary eragnets and the step by step mechanism.
- The following compound readily eliminates CO; to form a conjugated six membered ring. (a) Complete the reaction with drawing the possible structure and (b) explain why this reaction goes steadily.please explain clearly and in detail. thank you! 2-butene reacts with NBS yields and forms two products, 1-bromobut-2-ene and 3-bromobut-1-ene. explain how this happens using mechanisms.Give the major organic product(s) for each step of the following reactions (a,b,c)
- A. For each reaction1) Provide the missing reagents/conditions or major organic products as appropriate2) Assign each reaction as Oxidation or Reduction unless otherwise directed3) Pay attention to stereochemistry including racemic mixtures unless specifiedComplete the following reactions with the majority products and the missing reaction conditions. Indicate stereochemistry if applicable. Also indicate the type of reaction that took place (SN1, SN2, E1, E2, E1bc)Orgonic Chmeistry II: 2,4,6-Trinitrotoluene (TNT) is synthesized by trinitrating toluene. The first nitration proceeds much faster than the second two. Briefly explain.
- The reaction of cyclohexene with iodobenzene under Heck conditions forms E, a coupling product with the new phenyl group on the allylic carbon, but none of the “expected” coupling product F with the phenyl group bonded directly to the carbon–carbon double bond. Draw a stepwise mechanism that illustrates how E is formed.Hello. Kindly help me understand if this is a possible schematic of the reaction. The specific question is in Organic Chemistry Principles and Mechanisms by Joel Karty 2nd Edition, Problem 12.11 on page 614. I believe the predicted major product should come as well as with its enantiomer hence 4 products predicted for this reaction as there is no easy rotation about the cyclic structureC) provide the drawing of stereochemical model explaining the obtained stereochemistry of product here C) provide the drawing of stereochemical model explaining the obtained stereochemistry of product here

