Q: The equilibrium constant, Kc, for the following reaction is 6.30 at 723 K. 2NH3 (9) N2(g) + 3H2 (9)…
A: Given,The equilibrium equation:The equilibrium constant at 723 K = 6.30At equilibrium,moles of NH3…
Q: Draw a complete mechanism for the formation of your product via Friedel Crafts Acylation.Reaction…
A: Friedel Crafts Acylation reaction is an Electrophilic Aromatic Substitution Reaction.Acylium ion…
Q: Draw one of the two enantiomers of the major product from this reaction. Use a dash or wedge bond to…
A: Answer:When an alkene reacts with Br2, a dibromo alkane will be formed. The reaction proceeds via…
Q: Predict the major products of the reaction in the drawing area below. Be sure to use dash and wedge…
A: Answer:Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide…
Q: Identify the alkyne. O a. CH3CH2CH2CH3 O b. CH2CH2 CH2CH2 O c. CH3CHCHCH3 Od. CH3CCCH3 Clear my…
A:
Q: HO OH Give correct detailed Solution with explanation needed don't give Handwritten answer. What is…
A: Rules to write the chemical formula of a organic compound: • Identify the symbol of each atom, In…
Q: Which of the following conclusions can you draw from this mass spectrum (choose one) 100 88 80- Rel.…
A: A molecule with one Cl atom in it show M and (M+2) peaks with the intensity ratio of 3:1 as the…
Q: single bonds. of the starting material to draw the major product(s). Use the single bond tool to…
A: The objective of the question is to find the structure of the major product for the given reaction…
Q: Provide the structure of the major organic product in the reaction below. H FIN 1. CH3I (excess) 2.…
A: The given reaction scheme is shown below.In this reaction, first occurs N-alkylation on amine by…
Q: Draw the stereoisomers of 4-chloro-2-methylhexane. Use a dash or wedge bond to indicate the…
A: Stereoisomers are molecules with the same molecular formula and same connections between the…
Q: O= & 1. NaOH, H2O, heat 2. HCI, H2O (workup) O= & OH 애애 OH
A: Given an organic compound and reagent .Determine the mechanism
Q: (a) For Co(NH3)63+, give the LFT diagram, with electrons included as well as Aoct, HOMO, LUMO, LFSE…
A: The full form of LFT is Ligand Field Theory (LFT). To construct this LFT diagram for Co(NH3)63+, we…
Q: 2. Calculate the volume, in milliliters, of a 0.390 M KOH solution that should be added to 5.50g of…
A: In given question buffer is prepared from strong base KOH and weak acid HEPES. We will use Henderson…
Q: Draw the major product of this elimination. Consider regiochemistry and stereochemistry. Ignore…
A: The objective of the question is to find the major product of the given elimination reaction by…
Q: .Draw Lewis Dot diagrams for the PF3 b) SiCl4 SeH2 d) Sl₂ e) SiSez
A: We have to draw the Lewis dot structure
Q: 1. 222 86 Rn decays by emission of an alpha particle. The product of this decay is 2. Uranium-238…
A: Please note: As per our company guidelines we are supposed to answer only three parts of one…
Q: What is (are) the major product(s) for the following reaction? EtOH, heat Br O More than one of…
A: The given reaction scheme is shown below.We have to provide the major products of the given…
Q: 4. Propose a synthesis for the following compound using benzene or toluene and any other reagents…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: 1. Consider the 3-step reaction below. Assume that the halide and Mg are in excess of the ethyl…
A: B is the major product.Explanation:The major product of the reaction will be product B.The complete…
Q: ▾ Part A (s) (M) In [A] 1/[A] 0.00 0.500 -0.693 2.00 20.0 0.389 -0.944 2.57 40.0 0.303 -1.19 3.30…
A: Let's say, we have the following reaction:Where, A is the reactant and P is the product.Now, if we…
Q: Name the following compound by IUPAC rules. CH3 CH2CH3 CHCH2CH2-O CH2CH2CH2CH3
A: A structure of an organic moleucle is given in the question, We have to interpret IUPAC name of the…
Q: Which of the following can be best explained by temporary dipole moments? Choose one or more: A.…
A: Temporary dipole moments, also known as London dispersion forces or instantaneous dipole moments,…
Q: Determine whether each item is describing an endothermic or exothermic reaction by checking the…
A: The answers are checked accordingly: EndothermicExothermicForming new bonds ✓Breaking bonds✓ Boiling…
Q: You have 50.0mL of 0.541M NH3. Kb = 1.8x10-5 You add 15.0mL of a 0.05M HCl solution. What is the pH…
A: The objective of this question is to calculate the pH of a solution after the addition of a certain…
Q: Br Br דיי CH₂ONa H₂C H₂C OH CH H₂SO4, A
A: We have to predict the correct product.
Q: Name the following
A: The objective of this question is to predict the name of the given organic compounds.The process of…
Q: intermediate. tion products obtained in the reaction shown. Assume that both are derived from the…
A: Conjugated dienes can give 1,2 or 1,4 addition products in the electrophilic addition reactions. In…
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Zn(s) + O2(g) → 2ZnO(s)…
A: Enthalpy change for a reaction is the amount that is evolved or absorbed by a reaction at different…
Q: H H H. H H H How many bonding pairs and nonbonding pairs of electrons does each carbon atom contain?…
A: The objective of the question is to determine the number of bonding and nonbonding pairs of…
Q: Predict the major, organic product for the following reaction sequence. E
A: Given is organic reaction. The starting compound is aromatic halide.
Q: Determine the structures of compounds A-F. Br i 1. LAH PBr3 1. PPh3 H A 2. H₂O 2. nBuLi 1. D 1. E Br…
A: Lithium Aluminium Hydride (LAH) reagent is versatile reducing agent which reduces almost all…
Q: Draw the two major products obtained in the reaction shown. Br2 (one equivalent) CH2Cl2
A: Given compound is brominated in the presence of CH2Cl2
Q: b. Propose a synthesis of the following product from the given starting materials. N ے ہی Br…
A:
Q: Draw the products of the reactions. + H₂O H+ +1 equiv OH- products Select Draw Templates More Erase…
A: Given are organic reactions.The given starting compounds are ester.The given reactions are ester…
Q: 。 A Lewis structure for the carbonate ion (CO¹³½³-) is shown, but incomplete. Complete the structure…
A: Answer below this question Explanation:
Q: 2. If a 10.0g sample of the unknown you used in Part II is dissolved in 5.00mL of water and if the…
A: The objective of the question is to find out how much potassium nitrate (in grams) from the original…
Q: what will be the steps involved to yield the product in this reaction
A: Given is organic synthesis reaction.The given starting compound is acid chloride.
Q: Write a balanced equation to represent the reaction shown. H HT H H H-C-S Нон S C-H H HS-H H A…
A: Given data:In balance reaction number of each atom on reactant side is equal to number of each atom…
Q: The preparation of an aqueous solution is described in the table below. For this solution, write the…
A: Information about the question
Q: A chemistry student needs 55.00 g of acetone for an experiment. He has available 0.20 kg of a 33.2%…
A: Given that,Mass of acetone=Mass of solution= Mass of solution student use= ?
Q: Predict the expected product for each reaction and provide IUPAC name for the correct starting…
A: Box 1. 2-ethyl-1-buteneBox 2 Option BExplanation:
Q: Buffer System ID Acetic Acid Volume of ~0.50 M HC2H3O2 (mL) 1 30 Sodium Acetate Volume of ~0.50 M…
A: The objective of the question is to calculate the values of pH for the given buffer system ID.
Q: H Η H H OH H NaOH, EtOH, reflux H2SO4
A: Given are organic reactions. The station compounds for reaction 1 are aldehydes and reagent is…
Q: Give an IUPAC name for the compound below. Accepted names for branched alkyl groups are isopropyl,…
A: Given cyclic compound can be named by considering following points.I) identify parent cyclic ring…
Q: If you dilute a 50 mM stock solution by 1:500, what concentration do you end up with expressed in…
A: Make a 1:500 dilution.IMPLIESTake 1 part of your stock solution and add 500 parts of solvent.The…
Q: That is the product of the following reaction? a. b. Oc. CN Od. NaCN CN
A:
Q: Complete the Mechanism with all intermediate steps (hint: what can be done can be undone; what do…
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: 2) For each pair of molecules, CIRCLE the one with the lower pKa value for losing the indicated…
A: The acidity of a compound is related to its tendency to donate H atoms as H+. The higher tendency to…
Q: An analytical chemist is titrating 165.8 mL of a 0.9300M solution of propionic acid (HC₂H₂CO₂) with…
A:
Q: Hypothetical elements X, Z, and E are nonmetals. X and Z are in the same period and Z and E are in…
A: Now let us examine the factors:Because Z is more electronegative than X, it is more likely to be…
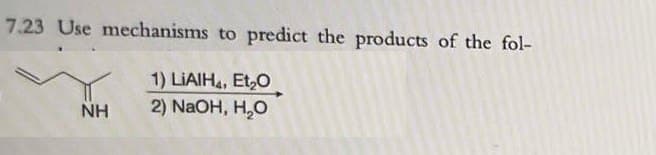
Step by step
Solved in 3 steps with 1 images

- Suppose you have compounds A–D at your disposal. Using these compounds, devise two different ways to make E. Which one of these methods is preferred, and why?6.Give the products:(a) Identify the missing major products A and B from the reactions below. ( b) For the catalyst used for each transformation, draw the structure and state the formal electron count and oxidation state (showing your workings)
- The alcohol compound given the formula below is used in perfume making. Bromine benzene and 1- synthesis of this compound using the necessary organic and inorganic chemicals starting from butene, show.what predictions can we make of the following reactionsthe reactions listed below do not take place.Explain WHY.(for each one seperately) (DO NOT TRY TO BALANCE them , balancing is not the problem) 3Ag + H3PO4 → Ag3PO4 + 3/2 H2
- A) Provide the reagent and reaction mechanism to show how the reactants and products in the following reaction can interconvert B) under what conditions would the reaction I) favour reactants, II) favour the products and III) why?Oct-1-yne (HC≡CCH2CH2CH2CH2CH2CH3) reacts rapidty with NaH, forming a gas that bubbles out of the reaction mixture, as one product. Oct-1-yne also reacts rapidty with CH3MgBr, and a different gas is produced. Write balanced equations for both reactions and identify the gases formed.6. Predict the products for the following reaction.

