7.55 The following reaction gives a product with the molecular for- mula C4H3O2. Draw the structure of the product. Br Br ONa C,HO2 + 2 NaBr NaO
7.55 The following reaction gives a product with the molecular for- mula C4H3O2. Draw the structure of the product. Br Br ONa C,HO2 + 2 NaBr NaO
Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 74AP: Identify the reagents represented by the letters a-e in the following scheme:
Related questions
Question
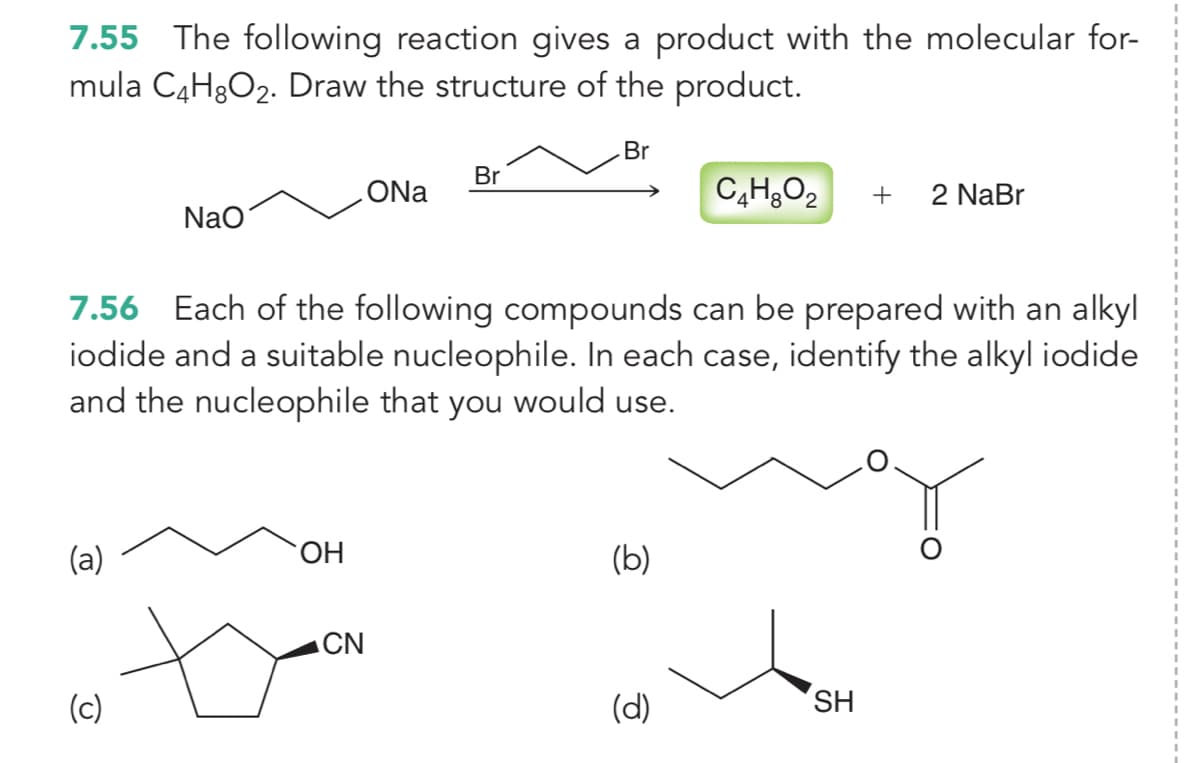
Transcribed Image Text:7.55 The following reaction gives a product with the molecular for-
mula C4H3O2. Draw the structure of the product.
3D
Br
Br
ONa
+
2 NaBr
Nao
7.56 Each of the following compounds can be prepared with an alkyl
iodide and a suitable nucleophile. In each case, identify the alkyl iodide
and the nucleophile that you would use.
(a)
HO.
(b)
ACN
(c)
(d)
SH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

