(7.9) Using the standard reduction potentials listed in the Table, determine following spontaneous under standard conditions. whether the reaction is Support your answer. Hg* (aq) + 21¯(aq) - Hga) +I2(s) From the Table: Hg (aq) * ,+2, - 2e- - Hg1) E°red = +0.85 V I2(s) + 2e- - 21¯(aq) E°red = %3D +0.54 V Answer: Is the above reaction spontaneous? (YES or NO) Briefly support your answer (.
(7.9) Using the standard reduction potentials listed in the Table, determine following spontaneous under standard conditions. whether the reaction is Support your answer. Hg* (aq) + 21¯(aq) - Hga) +I2(s) From the Table: Hg (aq) * ,+2, - 2e- - Hg1) E°red = +0.85 V I2(s) + 2e- - 21¯(aq) E°red = %3D +0.54 V Answer: Is the above reaction spontaneous? (YES or NO) Briefly support your answer (.
Chemistry for Engineering Students
3rd Edition
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter1: Introduction To Chemistry
Section: Chapter Questions
Problem 1.77PAE
Related questions
Question
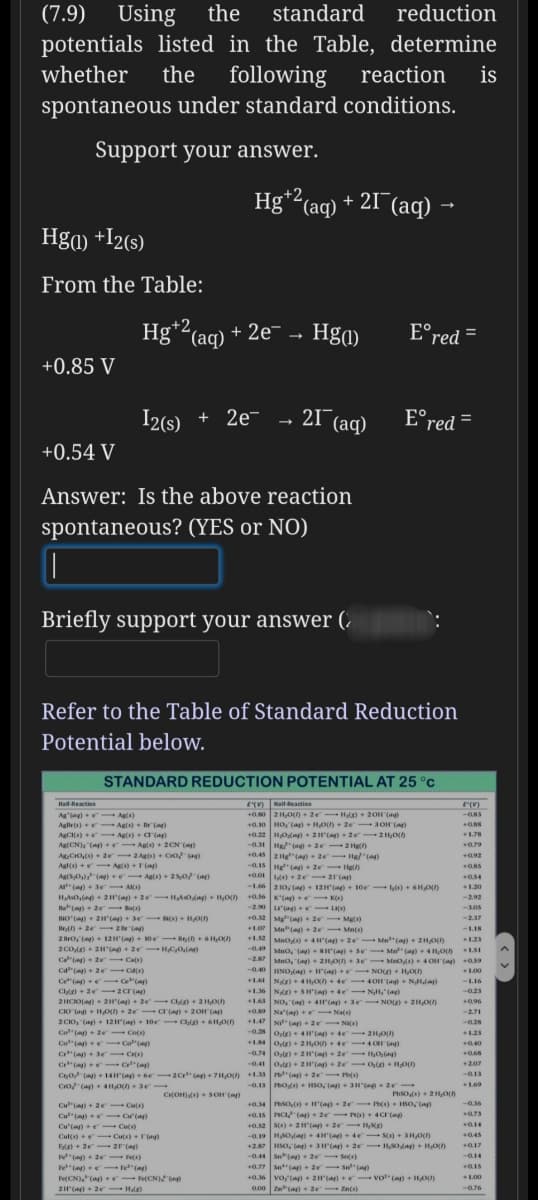
Transcribed Image Text:(7.9) Using
potentials listed in the Table,
the
standard
reduction
determine
whether
the
following
reaction
is
spontaneous under standard conditions.
Support your answer.
Hg*2(aq) + 21¯(aq) →
,+2,
Hga) +I2(s)
From the Table:
Hg*2(aq) + 2e¯ → Hga)
E°red =
+0.85 V
I2(s)
+ 2e- - 21 (aq)
E°red =
+0.54 V
Answer: Is the above reaction
spontaneous? (YES or NO)
Briefly support your answer (.
Refer to the Table of Standard Reduction
Potential below.
STANDARD REDUCTION POTENTIAL AT 25 °C
ETV) atteaction
+0.50 2H,O + 2e-Hlg) + 20H (a
Hait-Reaction
(V)
Ag (ag) +- Agis)
Aglle(s)+- Ag)+ r (a)
AgCIs) - Ag(s) -a(ap)
Ag(CN), (a) eAgs)+ 2CN (a)
-O83
Agi+ r(a)
0.10 HO, (a) + HO) + 2e 3OH (a)
+0.22 HOa) + 2H(ag) 2e 2Ho)
-0.31 He(a 2e 2 Hg)
+0.45 2Hg"(a)+ 2e Hg ()
-0.15 He(a)+ 2e Hel)
) + 2e 21(a)
-1.46 2 10, (a) + 12H(a) + 10e l) +6HO)
+0.56K'(ag) K)
+1.78
a79
AgCro,(s) + 2e 2 Ags) + Cro )
Agl(s) +e- Ag) (a)
AgrS,0(a) + - Agis) + 25,0 (a)
A" (a) + 3e - A)
92
054
1.20
HAs0,n- 2H"(a) + 2e HAOa) + H,o)
--2.92
Ba"(a)+ 2e al)
u'(ag) +e- LA)
+0.32 Mg"(a)+ 2e Mg)
+1.07 Mn ()+ 2e- Mn(s)
-2.37
HO () + 21H" () + 3e- N(s) + HO)
len + 2e- 2 (a)
2 Bro, (a)- 12H (a)+ 10e - Ben+ 6 HOU)
-LI8
+1.52 Mno 4H(a)+ 2e Mn(a)+ 2H0
-0.49 MnO, () BH(a) Se- Ma () 41,On
1.23
Ca"(a) + 2e Cats)
Ca"(a) + 2e cas)
Ce"(a) e -Ce"(a)
Cle) + 2e- 2cr(a)
2HCIO() 2H'(a)+ 2e C 2HOU)
CIO (ag + HO) + 2e Cr (a) + 20H (a)
2 CIO, (a)+ 12H(a) 10e C 6H,On
Co(a) 2e Co(s)
Co"(a) Co"(ay)
Cr"(a) 3e Crs)
Cr"(a) - C"(a)
Co (ay) + 14H(a)+ 6e- 2C(a)+ 7H,O) +1.33 "(a) + 2e- P)
-2.87
Mno, (n) 21O)+ 3e Meo) 4OH(a) a59
-0.40 HNO,(a)+ (a) + r- NO) + HO)
+1.61 N) + 41O)+4e 4OH (ag + NHda)
+1.36 N) SH(a)+4e NH, (ag)
+1.63 NO, tarn+ 4(a)+ 3e- NO)+ 21HOU)
100
-L16
--0.23
+96
+0.9 Na'(a) Na(s)
+1.47 N(a)+2e- N)
-2.71
-0.28
-0.28 otr)4H'() +4e 21HOU)
1.23
+1 oe) + 2 HO) + de -4 OH G)
0.40
-0.74 Or)2H() 2e HOng)
-0.41 o,) + 2(a) + 2e- On + HO)
+207
-Q.13
CO (a) + 4HO 3r
-0.13 o) HSO, ) 3Hp- 2e-
1.69
Ce(OH)+ SOH (a
+0.34 rtso) + H"(ag)+ 2e - I) + SO, (a)
+0.15 PCI (a) + 2e P) + 4Cr(a
-0.36
Cu"(a) + 2e Culs)
Cu"(a) Cu'(a)
Cu'len +e Cuts)
a73
+0.52 S(s)+ 2H'(a)+ 2e H)
Cul(s) +e- Cu(s) + T(ag)
Ede) + 2e- 2F(a)
(a)- 2e Fe)
045
-0.19 HSOag) + 4H"(a) + 4e- S) + 3H,On
+27 HSO, ng) 3H"(a) + 2e HS0 ag) + HO)
-0.44 S(a)+ 2e Sns)
+0.77 Set(a)+ 2e- S ()
--a14
FeCN) (a) + FeCN) Gn
2H(ag)+ 2e Hl)
vo,(a) + 2H (a)e vo" (a) + HO)
a00 Z a)+ 2e- Zn(s)
+0.
100
--a76
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning