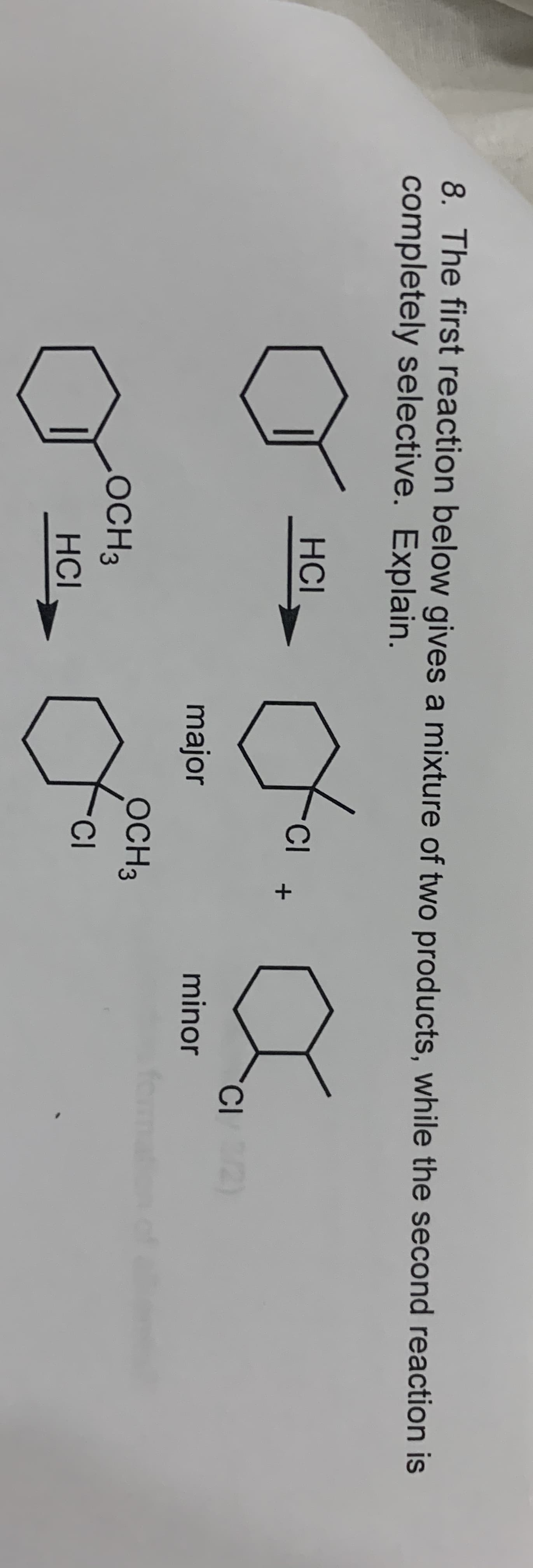
8. The first reaction below gives a mixture of two products, while the second reaction is completely selective. Explain. НCl CI major minor ОСНЗ ОCHЗ TCI НCI
Q: Draw a mechanism to explaim the observation show the movement of electrons with a cwwwed w Draw all…
A: In the given process, formation of electrophile is quite important, which proceeds the further…
Q: Predict the major product of the following reaction. NH₂ 1a. OLI O 1b. MeCl 1a. CH3I (excess) K₂CO3…
A: -> Nucleophile can give Michael addition reaction to the alpha,beta - unsaturated carbonyl…
Q: 3.3 Explain the reasons why the reactions provided below DO NOT work and show the other expected…
A: Reasons: a. The initial reaction will not work because nitrogen of aniline will react fast with…
Q: Predict the product for the following reaction. 1. O3 2. DMS 3. Na,Cr,0,/H9SO,H,0 HOH LOH HO- HO. II…
A:
Q: Br Br 2 Nal 6. CH;COCH3 Heat H2, Pt (CH3);CONA 7. Br (CH3);COH HCI, Heat H3O+ 8. Heat Hg(OAc)2, H2O…
A: When alkene react with HBr in presence of peroxide, then the negative part of the addendum attach to…
Q: 46. Which mechanism(s) would be favored under the following reaction conditions? -CI NaBr DMSO A)…
A: The substrate is a haloalkane and NaBr salt can dissociate to form Br- ion. This Br- can act as…
Q: Identify the reagents needed for the given reaction. CH2=CH-CH,CHO → CH2=CH-CH2CH2OH Select one: O…
A: Sodium borohydride, NaBH4 is a reducing agent and it can selectively reduce a carbonyl functional in…
Q: 4. Consider the following reaction: HBr 40°C a) Show a detailed mechanism for the reaction and give…
A: Hello. Since the question has multiple subparts, the first three subparts have been solved for you.…
Q: 24. Predict the major product of the following reaction. OH HNO3 H,SO, d. а. O,N. HO, OH NO2 NO2 е.…
A: Benzoic acid is an aromatic carboxylic acid. It behaves as an electron-withdrawing group through the…
Q: GENERAL SN1, SN2, AND ELIMINATION Which product (or products) would you expect to obtain from each…
A:
Q: CH3 HNO3 KMNO4 A В H2SO4 H*, Heat Br
A: When benzene ring possesses two different groups among which one is activating and the other is…
Q: N-H ? Catalytic H EH,0) но, 'N NH2 HO N- (a) (b) (c) (d)
A: When a cyclic ketone , cyclopentanone treated with tetrahydropyrrole (or pyrrolidine) in…
Q: Identify the product(s) of the reaction below. hv ? NBS Br Br Br Br
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: 13. Give mechanisms for three of the following four transformations. You will be penalized if you do…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: Which mechanism best describes the deuteron-bromination of the following cyclohexadiene derivative…
A: The thermodynamically favourable pathway for the given transformation is governed by the stability…
Q: What are the products for the following ozonolysis reaction? 1.0₂ 2. DMS III A. I and II B. II and…
A:
Q: 10. Treatment of 1,2-dibromoethane with the dithiolate dianion shown in the reaction below leads to…
A: Sulphur ion is a good Nucleophile and thus attacks the carbon attached with Bromine atom as below.
Q: Help
A: Answer is given as follows
Q: Draw the products of the following ozonolysis reactions: 03 (1) Me₂S 03 (2) Zn, HCI ?+ ? ?+ ?
A: In this question we have to tell the product of the reaction.
Q: The reaction of 2,2-dimethyl-1-propanol [(CH3)3CCH2OH], also known by the common name neopentyl…
A: Neopentyl alcohol is derived from neopentane. It has a prefix neo because of the presence of…
Q: Although dehydrohalogenation occurs with anti periplanar geometry, some eliminations have syn…
A: Syn elimination: In This reaction, leaving groups are present in syn periplanar with each other. Syn…
Q: (b) Write out the product(s) for the reactions below. Analyze the compounds (i) and (ii) below and…
A:
Q: Part 4: Reactions: Complete the following reactions by providing the correct product(s), starting…
A: 17. Hydroboration–oxidation reaction: Alkene gives an electrophilic addition reaction with borane.…
Q: 1. What is the product (s) of the following Reaction? HBr Br Br A B D a. B&D b. C&D A&C d. A&B&C&D…
A: I have solved only qus no. 1 as asked in the question.
Q: a) Will the following reaction proceed primarily by Sn1 or SN2? Show the product/products. NaOCH3…
A: Secondary alkyl halide with strong base undergo SN2 (retention of product) Secondary alkyl halide…
Q: . Provide the major organic product of the reaction below: Brz hv Br Br Br None A. 36. What is the…
A: I sloved first three. Remaining repost
Q: What is the missing product from this reaction? 32p → 32s + 15 16 A) He B) -1 D) e E) "P e O A В OD…
A:
Q: LDA CH3-I B 0°C
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: please show the detailed mechanism for each step for the reaction below. OTBDPS Me TMANO (anhydrous)…
A: Step 1 : alklyne addition to two coblat atoms. Step 2 : olefin addition to one of the coblat atom.…
Q: Please draw the product below. You need to show stereochemistry using dashes and wedges. OH \CO2CH3…
A: In organic conversions, appropriate reagent converts the reactants into products. Reagents attack…
Q: What is the intermediate of the following reaction? Submit your answer as a SMILES string. Be sure…
A: The TBF is used for the Deprotection of of alcohol from silyl group. Second step is oxidation step
Q: d. 2 ea Brz e. 1 eq cl2 f.
A:
Q: 15. Draw the major product(s) of the following reactions. Write N.R. if there no reaction. 1)…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: 43-46. Consider the reaction below. Which of the following reagents is suitable for the synthesis of…
A:
Q: which of the following electrophiles would yield the fastest reaction rate in an SN1 reaction
A: In SN1 reaction, the order of stability of carbocations is 3○> 2○>1○. Resonance effects…
Q: Predict the product of the following reaction and classify the reaction.…
A: The complete balanced chemical equation can be written as follows: Pb(NO3)2 + FeSO4 → PbSO4 +…
Q: 3. Predict the compounds in each pair that wll undergo the S,2 reaction faster. Fully Justify your…
A: We have to give rate of sN2 reaction
Q: a) Br HO но ? H20 04150 b) CH3CH,Br CN c) CH3CH,CI H20 ?
A:
Q: 3-The reaction of 2,2-dimethyl-1-propanol with HBr is very slow and gives 2-Bromo-2-methyl butane as…
A:
Q: 5. One of the two diastereomers shown below reacts with potassium butoxide significantly faster than…
A: Given reaction is elimination reaction.
Q: 13. Identify the compounds A to E in each of the following reactions. conc. H2SO4 a) CH3CH,CH,OH A +…
A: conc H2SO4 is a dehydrating agent. It takes away water molecule from any compound making a double…
Q: CO2Et 120 °C ? 1c. CO2Et .CO,Et CO,Et CO2Et cO,Et CO2Et a d 1d. Identify the reagent(s) that will…
A: Since you have posted multiple subparts so I am solving first three subparts for you.If you want to…
Q: Order the following substrates in descending order of reactivity in the following reaction: 1) PhLi…
A:
Q: What is the mechanism of the major product in the following reaction? C d) Sp CH₂ Q ? -) Sul and El…
A: -> In presence of strong base there occur E2 elimination reaction.
Q: 2. Draw the expected product for the following multi-step reaction. 1. Br2, acetic acid 2. IZ
A:
Q: Br + Na cl. Br + K + Но + KCI c2. F Nao "CI dl. + + Nao- NaBr + HO- d2. 7
A: In zaitsev Elmination most substituted alkene is major product. If the base is bulk,less substituted…
Q: 7. For each reaction below, determine whether the primary reaction is SN1, SN2, E1, or E2, and then…
A:
Q: 1) МezNH 2) НСНО, H* 3) CH3I 4) Он-
A: The reaction is the shift base formation.
Q: Draw the products of the following ozonolysis reactions: 03 (1) Zn, HCI 03 (2) Zn, HCI ?+ ? ?
A: It is ozonolysis of alkene followed by Zn/HCl which gives aldehyde and ketone depending upon…
Q: Rearrangment (р 1283) CH, H,C. ZnBr, H,C CH, Br HBr CH 93% yield он (Manning P. Cooke, Jr.) I need…
A: The mechanism of reaction is provided below in attach image.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d below9. Product of the reaction of nitrobenzene with sulfuric acid in the presence of oleum at 60 °C:a) o-nitro benzenesulfonic acidb) m-nitro benzenesulfonic acidc) p-nitro benzenesulfonic acidd) None of the above 10. Due to their ability to move through ducts or pipes, they are generally called fluids:a) Solidsb) Solid mixturesc) Gasesd) Liquids and gases1) The Predict the product(s). 2)Label any kinetic/thermodynamic product(s). 3)Give reason towards the selection of kinetic product(s).
- Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answerwhat are the reagents to carry out this multi-step reaction and what are their inetermediates? explain regioselectivity.How to prepare the Organometallic Reagents ?
- Consider the reactions shown below. For each reaction state if it would favor S N 1 or S N 2pathway and give the major product of the reaction.Which of the following reagent best accomplish this transformation below? a. Li/NH3 b. H2SO4, H2O, HgSO4 c. BH3, THF d. potassium permanganateWhat steps are needed to prepare phenylacetylene, C6H5C = CH, from each compound: (a) C6H5CH2CHBr2; (b) C6H5CHBrCH3; (c) C6H5CH2CH2OH?
- Predict the product(s) for each reaction belowThiols such as ethanethiol and propanethiol can be used to reduce vitamin K epoxide to vitamin KH2, but they react much more slowly than dihydrolipoate. Explain why this is so.Write the major product of each reaction and what kind of mechanism it is: sn1, sn2, e1, e2.



