8. What is the relationship between the structures shown below? Br CH3 Br H OH H3C CH3 OH H CH2CH3 (a) identical compounds (b) enantiomers (c) diastereomers (d) constitutional isomers
8. What is the relationship between the structures shown below? Br CH3 Br H OH H3C CH3 OH H CH2CH3 (a) identical compounds (b) enantiomers (c) diastereomers (d) constitutional isomers
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter3: Stereoisomerism And Chirality
Section: Chapter Questions
Problem 3.36P
Related questions
Question
No need to upload any image just give the answer with proper explanation will upvote.
Give explanation of the correct option and explanation of the incorrect options
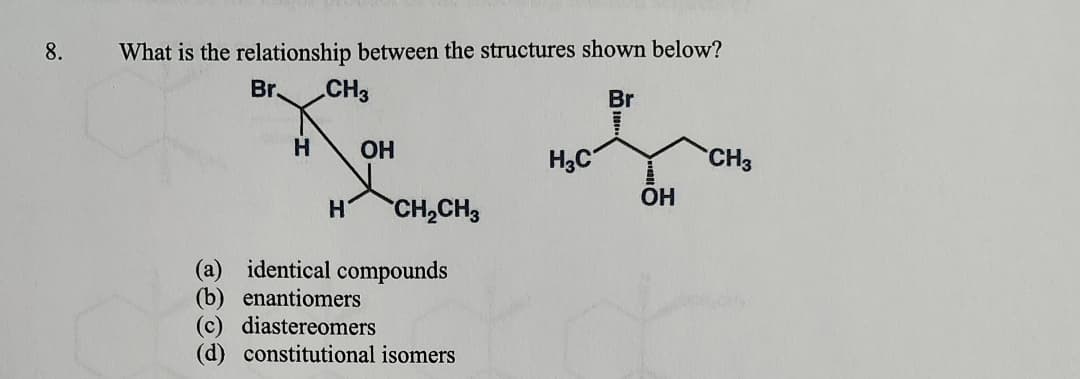
Transcribed Image Text:8.
What is the relationship between the structures shown below?
Br
CH3
Br
H
OH
H3C
CH3
OH
H CH2CH3
(a) identical compounds
(b) enantiomers
(c) diastereomers
(d) constitutional isomers
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
