9-10. Below is the temperature-solubility curve for several salts. 100 90 80 A. 10 grams B. 30 grams C. 60 grams D. 80 grams Solubility (g of salt in 100 g H₂O) 70 60 50 40 30 20 10 NaNO CaCl₂ Pb(NO3)2 NaCl KNO3 KCI KCIO3 K₂Cr₂O₂ Ce₂(SO4)3 0 0 10 20 30 40 50 60 70 80 90 100 Temperature (°C) A solution of potassium chlorate, KCIO3, has 20 grams of the salt dissolved in 100 grams of water at 70 °C. Based on the above diagram, approximately how many more grams of the salt can be added to the solution before it becomes saturated?
9-10. Below is the temperature-solubility curve for several salts. 100 90 80 A. 10 grams B. 30 grams C. 60 grams D. 80 grams Solubility (g of salt in 100 g H₂O) 70 60 50 40 30 20 10 NaNO CaCl₂ Pb(NO3)2 NaCl KNO3 KCI KCIO3 K₂Cr₂O₂ Ce₂(SO4)3 0 0 10 20 30 40 50 60 70 80 90 100 Temperature (°C) A solution of potassium chlorate, KCIO3, has 20 grams of the salt dissolved in 100 grams of water at 70 °C. Based on the above diagram, approximately how many more grams of the salt can be added to the solution before it becomes saturated?
Chapter16: Solubility And Complex Ion Equilibria
Section: Chapter Questions
Problem 10RQ: Figure 15-3 outlines the classic scheme for separating a mixture of insoluble chloride salts from...
Related questions
Question
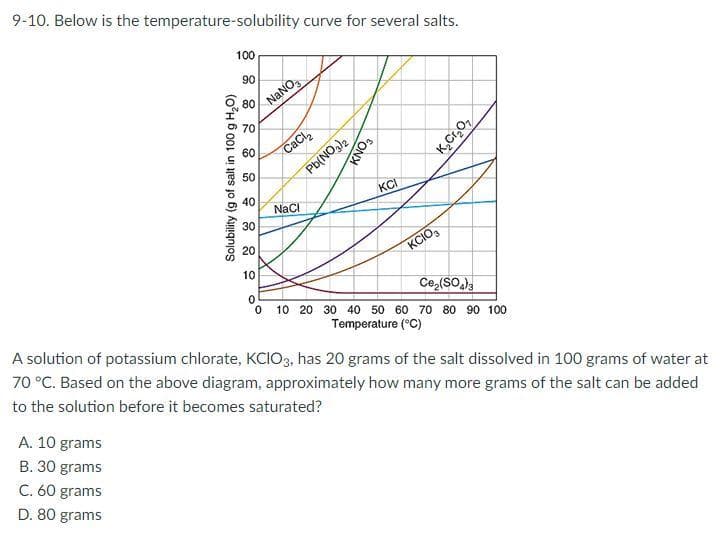
Transcribed Image Text:9-10. Below is the temperature-solubility curve for several salts.
100
90
80
A. 10 grams
B. 30 grams
C. 60 grams
D. 80 grams
Solubility (g of salt in 100 g H₂O)
70
60
50
40
30
20
10
NaNO3
CaCl₂
Pb(NO3)2
NaCl
KNO3
KCI
KCIO3
K₂Cr₂O₂
Ce₂(SO4)3
0
0 10 20 30 40 50 60 70 80 90 100
Temperature (°C)
A solution of potassium chlorate, KCIO3, has 20 grams of the salt dissolved in 100 grams of water at
70 °C. Based on the above diagram, approximately how many more grams of the salt can be added
to the solution before it becomes saturated?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax