900 800- 700 600 500 400 300 200 100 Vapor pressure (mm Carbon disulfide Methano 0. 10 20 30 40 Ethandl Heptane 50 60 70 80 90 100 Temperature (°C) 110 From the plot of vapor pressures vs temperature above, estimate the boiling point of methanol when the external pressure is 182 mm Hg. "C
900 800- 700 600 500 400 300 200 100 Vapor pressure (mm Carbon disulfide Methano 0. 10 20 30 40 Ethandl Heptane 50 60 70 80 90 100 Temperature (°C) 110 From the plot of vapor pressures vs temperature above, estimate the boiling point of methanol when the external pressure is 182 mm Hg. "C
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter6: Steam Distillation, Vacuum Distillation, And Sublimation
Section: Chapter Questions
Problem 6Q: A mixture of toluene (bp110.8C) and water is steam distilled. Visual inspection of the distillate...
Related questions
Question
Help me please asap. Thanks !
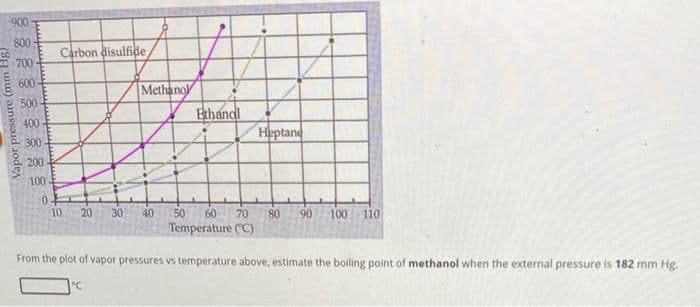
Transcribed Image Text:900
800
Vapor pressure (mm
700
600
500
400
300
200
100
0
Carbon disulfide
10 20 30
Methano
"C
40
Ethandl
50 60 70
Temperature (°C)
Heptane
80 90 100 110
From the plot of vapor pressures vs temperature above, estimate the boiling point of methanol when the external pressure is 182 mm Hg.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning