A 1.914 g sample of a component of the light petroleum distillate called naphtha is found to yield 5.867 g CO2 (9) and 2.802 g H2O(1) on complete combustion. This particular compound is also found to be an alkane with one methyl group attached to a longer carbon chain and to have a molecular formula twice its empirical formula. The compound also has the following properties: • melting point of -154 °C, • boiling point of 60.3 °C, • density of 0.6532 g mL¯! at 20 ° C, • specific heat of 2.25 J g¬1 °C-1. and 1 A¢H = -204.6 kJ mol¬1 .
A 1.914 g sample of a component of the light petroleum distillate called naphtha is found to yield 5.867 g CO2 (9) and 2.802 g H2O(1) on complete combustion. This particular compound is also found to be an alkane with one methyl group attached to a longer carbon chain and to have a molecular formula twice its empirical formula. The compound also has the following properties: • melting point of -154 °C, • boiling point of 60.3 °C, • density of 0.6532 g mL¯! at 20 ° C, • specific heat of 2.25 J g¬1 °C-1. and 1 A¢H = -204.6 kJ mol¬1 .
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter21: Biochemistry
Section: Chapter Questions
Problem 30QAP
Related questions
Question
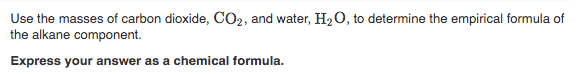
Transcribed Image Text:Use the masses of carbon dioxide, CO2, and water, H2 0, to determine the empirical formula of
the alkane component.
Express your answer as a chemical formula.

Transcribed Image Text:A 1.914 g sample of a component of the light
petroleum distillate called naphtha is found to yield
5.867 g CO2 (9) and 2.802 g H20(1) on
complete combustion.
This particular compound is also found to be an
alkane with one methyl group attached to a longer
carbon chain and to have a molecular formula twice
its empirical formula.
The compound also has the following properties:
• melting point of – 154 °C,
• boiling point of 60.3 °C,
• density of 0.6532 g mL at 20 °C,
• specific heat of 2.25 J g¯1 °C-1,
and
• A:H = -204.6 kJ mol-1 .
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning