The boiling points (at 1 atm of pressure) of several organic and inorganic materials are included below: Chemical Chemical Formula ВР (°С) Water H2O 100 Acetone C3H60 56 Dichloromethane CH2CI2 39.6 Diethyl Ether C4H100 34.6 Dodecanoic Acid C10H20O2 269 Ethanol C2H60 78.4 Formic Acid CH2O2 100.8 Hexane C6H14 68 Hydrogen Sulfide H2S |-60 Toluene C7H8 110.8 Xylene C8H10 139 Using the language of intermolecular forces, what insights can you offer to explain the data?
The boiling points (at 1 atm of pressure) of several organic and inorganic materials are included below: Chemical Chemical Formula ВР (°С) Water H2O 100 Acetone C3H60 56 Dichloromethane CH2CI2 39.6 Diethyl Ether C4H100 34.6 Dodecanoic Acid C10H20O2 269 Ethanol C2H60 78.4 Formic Acid CH2O2 100.8 Hexane C6H14 68 Hydrogen Sulfide H2S |-60 Toluene C7H8 110.8 Xylene C8H10 139 Using the language of intermolecular forces, what insights can you offer to explain the data?
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter11: Intermolecular Forces And Liquids
Section: Chapter Questions
Problem 62SCQ
Related questions
Question
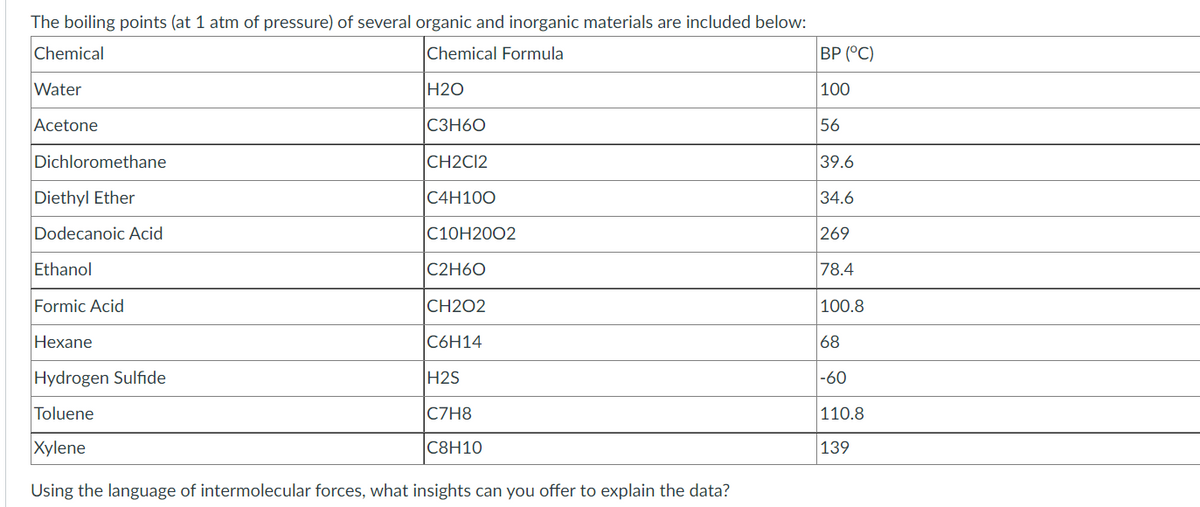
Transcribed Image Text:The boiling points (at 1 atm of pressure) of several organic and inorganic materials are included below:
Chemical
Chemical Formula
ВР (°С)
Water
H2O
100
Acetone
C3H60
56
Dichloromethane
CH2CI2
39.6
Diethyl Ether
C4H100
34.6
Dodecanoic Acid
С10Н2002
269
Ethanol
C2H60
78.4
Formic Acid
CH2O2
100.8
Hexane
C6H14
68
Hydrogen Sulfide
H2S
-60
Toluene
C7H8
|110.8
Xylene
С8Н10
139
Using the language of intermolecular forces, what insights can you offer to explain the data?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning