A 100 kg of mixture containing 40 w.% Ethyl glycol in water is contacted with an equal amount of pure solvent (Furfural) in an extraction process: A-What fraction of Ethyl glycol can be extracted in a single-stage extractor B-What fraction of Ethyl glycol could be extracted if the fresh solvent were divided in two parts and two cross successive extraction process is used Ethylene glycol Furfural 10 20 30 40 50 90 80 70 60/ 70 80 90 60 50 10 20 30 40 Plait point 50 40 60 70 20 30 80 10 90 A Water
A 100 kg of mixture containing 40 w.% Ethyl glycol in water is contacted with an equal amount of pure solvent (Furfural) in an extraction process: A-What fraction of Ethyl glycol can be extracted in a single-stage extractor B-What fraction of Ethyl glycol could be extracted if the fresh solvent were divided in two parts and two cross successive extraction process is used Ethylene glycol Furfural 10 20 30 40 50 90 80 70 60/ 70 80 90 60 50 10 20 30 40 Plait point 50 40 60 70 20 30 80 10 90 A Water
Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
9
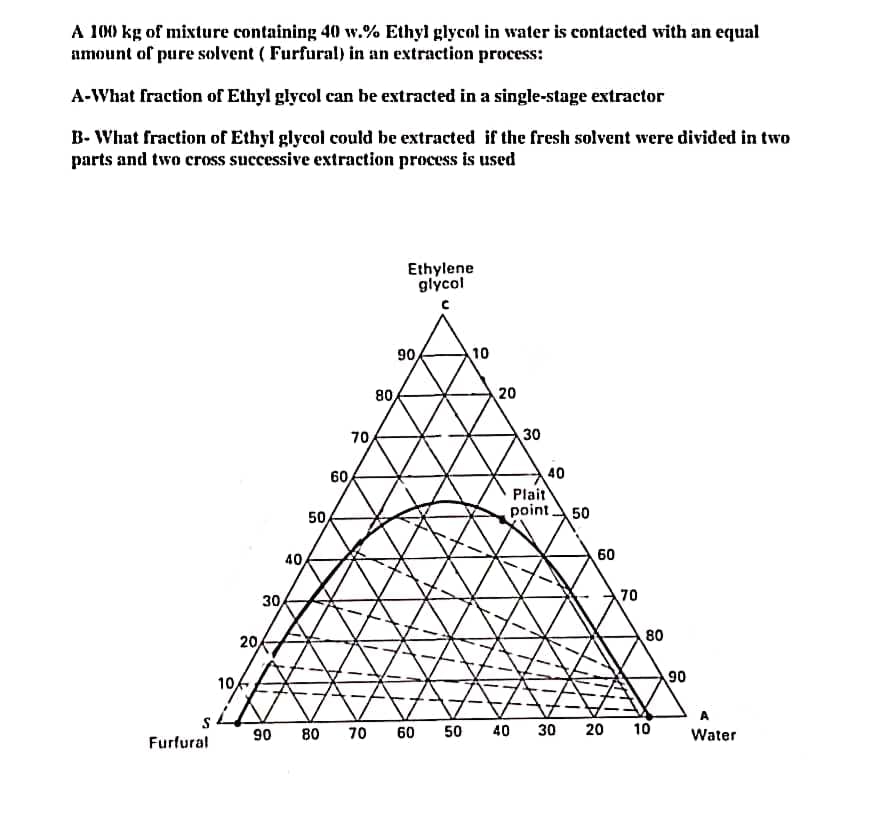
Transcribed Image Text:A 100 kg of mixture containing 40 w.% Ethyl glycol in water is contacted with an equal
amount of pure solvent (Furfural) in an extraction process:
A-What fraction of Ethyl glycol can be extracted in a single-stage extractor
B-What fraction of Ethyl glycol could be extracted if the fresh solvent were divided in two
parts and two cross successive extraction process is used
Ethylene
glycol
Furfural
20
10/
30
40
50
70
60/
90 80 70
80
90
60
50
10
20
30
40
Plait
point 50
40
30
60
20
70
80
10
90
A
Water
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

