A 5.27-g sample of an ant-control preparation was decomposed by wet ashing with H₂SO4 and HNO3. The arsenic in une residue was reduced to the trivalent state with hydrazine. After the excess reducing agent had been removed, the arsenic(III) was oxidized with electrolytically generated 12 in a faintly alkaline medium: HASO + 1₂ + 2HCO HASO + 2 + 2CO + H₂O The titration was complete after a constant current of 105.2 mA had been passed for 10 min and 43 s. Express the results of this analysis in terms of the percentage As2O3 in the original sample.
A 5.27-g sample of an ant-control preparation was decomposed by wet ashing with H₂SO4 and HNO3. The arsenic in une residue was reduced to the trivalent state with hydrazine. After the excess reducing agent had been removed, the arsenic(III) was oxidized with electrolytically generated 12 in a faintly alkaline medium: HASO + 1₂ + 2HCO HASO + 2 + 2CO + H₂O The titration was complete after a constant current of 105.2 mA had been passed for 10 min and 43 s. Express the results of this analysis in terms of the percentage As2O3 in the original sample.
Chapter17: Complexation And Precipitation Reactions And Titrations
Section: Chapter Questions
Problem 17.31QAP
Related questions
Question
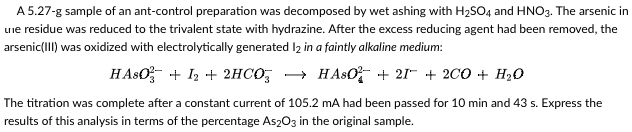
Transcribed Image Text:A 5.27-g sample of an ant-control preparation was decomposed by wet ashing with H₂SO4 and HNO3. The arsenic in
une residue was reduced to the trivalent state with hydrazine. After the excess reducing agent had been removed, the
arsenic(III) was oxidized with electrolytically generated 12 in a faintly alkaline medium:
HASO + 1₂ + 2HCO3 → HASO +21+ 2CO + H₂O
The titration was complete after a constant current of 105.2 mA had been passed for 10 min and 43 s. Express the
results of this analysis in terms of the percentage As2O3 in the original sample.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

