A 9.00 L tank at 9.44 °C is filled with 5.94 g of boron trifluoride gas and 17.9 g of sulfur hexafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits. mole fraction: boron trifluoride partial pressure: atm mole fraction: sulfur hexafluoride partial pressure: atm Total pressure in tank: atm
A 9.00 L tank at 9.44 °C is filled with 5.94 g of boron trifluoride gas and 17.9 g of sulfur hexafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits. mole fraction: boron trifluoride partial pressure: atm mole fraction: sulfur hexafluoride partial pressure: atm Total pressure in tank: atm
Chapter5: Gases
Section: Chapter Questions
Problem 152CP: You have an equimolar mixture of the gases SO2 and O2, along with some He, in a container fitted...
Related questions
Question
100%
Please do NOT round your answer until the end. Thank you
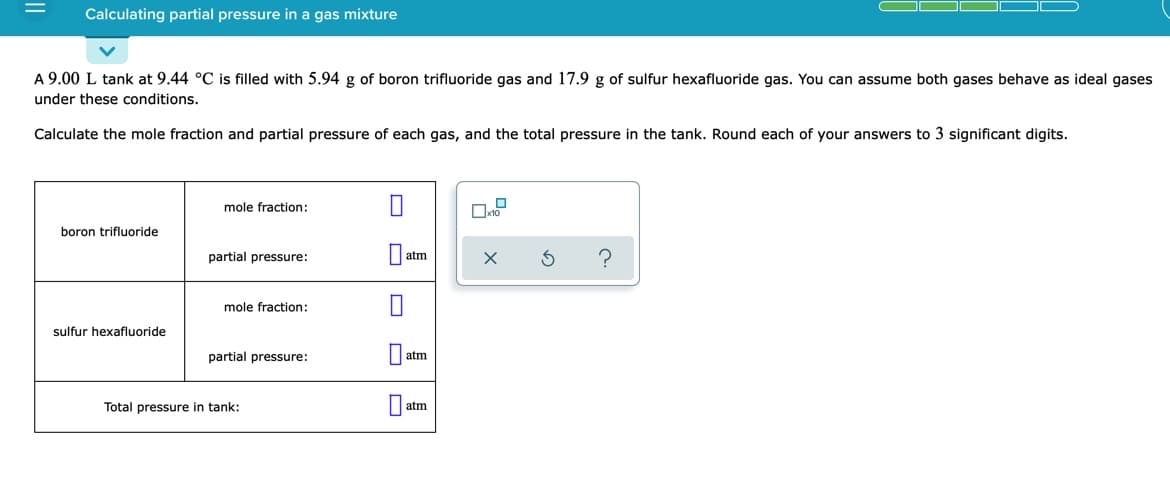
Transcribed Image Text:Calculating partial pressure in a gas mixture
A 9.00 L tank at 9.44 °C is filled with 5.94 g of boron trifluoride gas and 17.9 g of sulfur hexafluoride gas. You can assume both gases behave as ideal gases
under these conditions.
Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits.
mole fraction:
boron trifluoride
partial pressure:
atm
mole fraction:
sulfur hexafluoride
partial pressure:
atm
Total pressure in tank:
atm
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning