Q: What are the most important inorganic molecular substances for living beings?
A: Also, the chemical compounds of living things are known as organic compounds because of their…
Q: Which of the following statements is not true concerning the three molecules shown below? A B I. The…
A: The molecules having the same molecular formula but the structural arrangement of atoms in the…
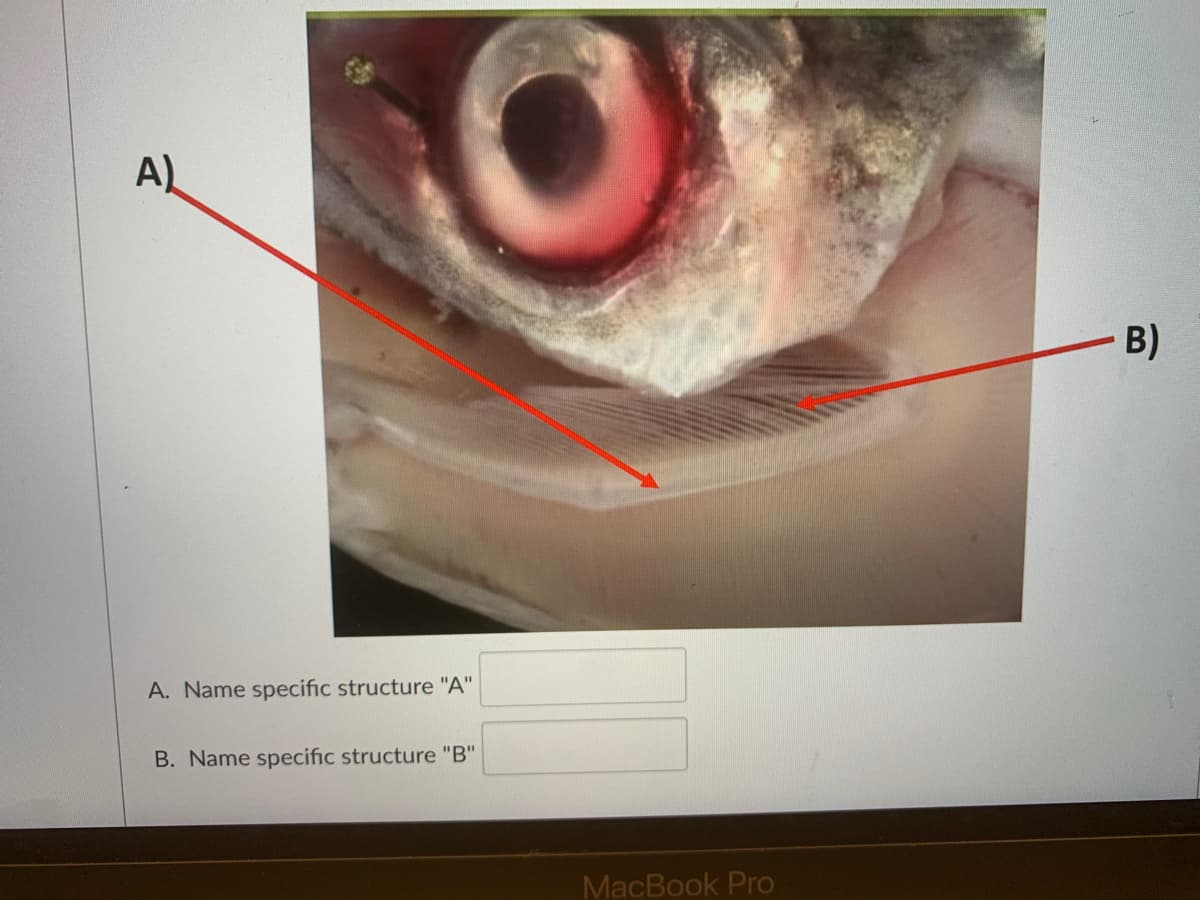
Q: Name specific structure "A"
A: Plant anatomy is used to study the internal structure of the plants. A typical plant body mainly…
Q: Which among the four bonds is strongest? And which one is weakess? Explain.
A: Biomolecules are the substances present in living organisms that play important roles in chemical…
Q: How Do Scientists Determine the Primary Structureof Nucleic Acids?
A: The sequence of bases in the nucleic acid chain gives the primary structure of DNA or RNA. Nucleic…
Q: why lung and soft tissue appear different on a radiograph when they have both an effective atomic…
A: The radio graphic image will appear according to the effective atomic number.
Q: Which macromolecule could be identified if the structure showed it had 6 carbons, 12 hydrogens, and…
A: macromolecules is a term usually used to identify biopolymers like carbohydrates , proteins , lipids…
Q: Which of the bond in the structure is NOT free to rotate?
A: Peptide bond formed between carboxyl group of one amino acid and the amino group of other amino…
Q: What atoms must be present in a molecule for it to be consideredorganic?
A: There are two types of molecules- Organic and inorganic.
Q: Which of the following is a representation of a covalently bonded molecule that shows the relative…
A: Structural formula is generally used to describe the molecular structure of a chemical compound…
Q: B
A: Structural representations help to identify different structures by observing the types of atoms,…
Q: Compare the structures of amino acids, dipeptides, polypeptides, and proteins.Which has the largest…
A: Proteins are complex molecules that are involved in the growth and maintenance of the body. The…
Q: which mineral element is found in nucleic acids ?
A: Nucleic acids are compounds essential for living organisms. There are two types of nucleic acids…
Q: Identify specific functional groups commonly found in biological molecules and describe their…
A: The carbon skeleton of biomolecules is composed of hydrogens and carbon atoms. But, in the…
Q: What structure is indicated by the number 2 in the image below?
A: Question - what structure indicated by number 2 ?
Q: Which macromolecule, along with its examples, is described in the table?
A: Macromolecules are very large molecules or biochemical compounds made up of several monomers…
Q: What causes atoms to form chemical bonds? Why do some elementsnot bond readily?
A: Chemical bonds are formed due to attraction between elements.
Q: Describe Tertiary Structure? Give an example?
A: Introduction Proteins are formed by 20 standard amino acids with the limited amino acids…
Q: Define macromolecule, polymer, and monomer.
A: An atom can be defined as the smallest component unit of matter which comprises a chemical element.…
Q: Define the following terms: a. metalloprotein b. primary structure c. secondary structure d.…
A: Proteins are considered as the organic compounds, which are made up of polypeptide chains.
Q: Which are functional units?
A: Functional units mean that part of the body that performs all the essential metabolic and other…
Q: The configuration of the given structure below is: * CH,OH он ÓH O alpha-L O alpha-D O beta -L O…
A: The carbohydrates can be represented by two types of configurations: L-isomer and D-isomer. When the…
Q: IDENTIFY THE FUNCTIONAL GROUP PRESENT IN THESE COMPOUNDS. A = ? B = ? C = ?
A: A group of atoms with specific chemical properties and forms a part of an organic molecule is…
Q: Which structure is highlighted?
A: As per the guidelines we are supposed to answer only the first question. Please repost the other…
Q: Why is it important to know the primary structure?
A: Introduction: Amino acids the macromolecules that contain two functional groups. The one group…
Q: What is Primary Structure?
A: The primary structure means the basic structure. The cell has DNA, which codes its information into…
Q: How does the bonding involved in a compound (nanoscopic interactions) influence the macroscopic…
A: A chemical bond is an attraction between atoms. This attraction may be seen as the result of…
Q: Is silica polar or nonpolar?
A: Chromatography is a technique that is used to separate a mixture of substances that are dissolved in…
Q: Give 2 examples of primary structure and secondary structure and explain the main difference between…
A: Chemical compounds have different structural organization in the nature. The compounds can have…
Q: _____________ are macromolecules with no fixed molecular weights.
A: A macromolecule is a biomolecule which large in size and high molecular weight.
Q: (b) (d) (f) (e) (h) (g) () () atomical directions (at
A: Anatomical positions are used to describe any region of the body in a specific stance. The different…
Q: Use diagrams to explain differences in macromolecular structural analysis using crystallography
A:
Q: write a brief desription of hydrogen bonds.
A: The temporary connections that hold two atoms, ions, or molecules together to form a chemical…
Q: what is second structure?
A: Secondary structure is defined as the local three-dimensional structure of sheets as helices and…
Q: Match the molecule with its correct description
A: mRNA - Carries a copy of the genetic "message". tRNA - Has anticodons Aminoacyl tRNA synthetase -…
Q: Why is the 3-dimensional structure of naturally occurring molecules important
A: The 3-D structure of naturally occuring molecules is important because :- The three-dimensional…
Q: If you were a pharmaceutical researcher, why would you want to learn the three-dimensional shapes of…
A: It is the application of the life sciences, physical sciences, mathematics and engineering…
Q: Many consumer products are almost identical in chemical composition but are sold under different…
A: Drugs are frequently given multiple names. A chemical name is given to a drug when it is first…
Q: Indicate which orbitals overlap to form the s bonds in each molecule.a. BeBr2 b. HgCl2 c. ICN
A: a)There is an overlapping of SP-hybridized orbital of beryllium molecule with the P orbital of the…
Q: What would be the simplest way to tell the difference in non polar covalent bonds, polar covalent…
A: The complete transfer of electrons leads to ionic bonds formation, whereas, sharing of electrons…
Q: HH H H H C-C- Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η H H H-C-O C- онн C-C- H H H H H H H CH, H H…
A: There are many macromolecules present in the living organism. They include carbohydrates, lipids,…
Q: When polymers are broken down into monomers, what would your body do with those monomers?
A: The biological polymers are the macromolecules that are formed by linking of small molecules called…
Q: m Which level of structure does the aß/aß and aßS/aßS in HbA and HbS describe?
A: The structural organisation of protein can be divided into four categories: i) Primary structureii)…
Q: I. Research for structure (submit on short bond paper, handwritten only)
A: 1. Phenolphthalein, (C20H14O4), organic compound of the phthalein family commonly used as an…
Q: What is structure formula of:
A: IUPAC name - Tert-butylcyclohexane
Q: Describe the nature of structure A C CO A,
A: The structure A here is the plasma membrane or the cell membrane. A plasma membrane is a bilayer of…
Step by step
Solved in 2 steps

- Convert the following structural formulas into condensed structures.X-ray crystallography is time-consuming and technically difficult. Why is the effort to understand the structure of biological molecules worthwhile? What is the recompense of such technology?Draw the structure and write the IUPAC name of sertraline
- In order to study protein structures and functions, many protein techniques have been developed for specific applications. Which of the following pair of technique: application is INCORRECT? a. Mass Spectrometry: protein molecular weight b. SDS-PAGE: protein structure c. X-ray crystallography: protein structure d. NMR: protein structure e. Peptide mass fingerprinting: protein presence and identificationWhat is the highlighted structure? BlueChemistry A homotetramer (lacking intermolecular covalent interactions) has a native molecular size of 60 kDa. What is the expected molecular size of this protein when analyzed by SDS-PAGE? a) 30 kDa b) 240 kDa c) 60 kDa d) 15 kDa
- Why does a sufficiently bright flash of light delivered over a short period of time produce a sensation of temporary blindness? Explain the molecular and biophysical properties in detail.Explain Four Levels of Protein Structure with examples and talk about the importance of each structure (Primary, Secondary, Tertiary, and Quaternary structure). Please write long answers for each structure.One advantage of the protein structures solved by NMR comparing to the x-ray structures is: a. It can determine the absolute, unique structure. b. It can solve protein structures in solution, which might be more physiological relevant. c. It is much easier to solve the structure. d. It has higher resolution. e. It can solve proteins with higher molecular weight.

