Q: A principal shell with a value of n=3 would contain s,p,d, and f orbitals true or false
A: A principal shell is indicated by the principal quantum number.
Q: which of the sub-atomic particles is commonly represented by this symbol:"e-"?
A: An ATOM is the smallest unit of ordinary matter that forms a chemical element. Every solid, liquid,…
Q: What forces of attraction hold the subunits in a quaternary structure?
A: Protein play wide variety of essential function in our body. They provide strength and structural…
Q: Identify the names of the following structure. (with alpha/beta and L-D designation) CH,OH CH2OH OH…
A: The D and L forms are isomers and form mirror images. The orientation of -H and -OH groups on the…
Q: Draw structures showing the reaction of glycerol with linoleic acid to form the triglyceride…
A: Triglycerides are formed by the joining of three molecules of fatty acids and a molecule of glycerol…
Q: How many electrons are in the outer shell of each of the following atoms?
A: Since, the question is incomplete, not provided with complete options, so here giving the…
Q: What is the difference between molecular mass and formula mass? Between molecular mass and molar…
A: Atoms are the basic building blocks of matter. Molecules are defined as the particle contains two or…
Q: Elemental analysis of a compound with molar mass 342.3 g/mol gives the following mass percent…
A: Given Percentage of C= 42.11 % Percentage of H=6.48 % Percentage of O=51.41% To Calculate:…
Q: Which of the following substances will have the greatest lattice energy? MgS NaF MgO CaO
A: Lattice energy can be defined as the energy required to convert one mole of an ionic solid into…
Q: Beyond the differences in molecular mass, what is different about determining the molarity of a…
A: Concentration of the solution is also expressed in terms of molarity. Molarity refers to the number…
Q: For the following peptide: group 3 O. C-NH, CH, Н 4 2 group 1 HO NH, 3 CH2 1 group 4 N. N-H group 2…
A: The given separation method has to be isoelectric Focusing or IEF as it is a method of separating…
Q: Why does the structure H-C=C-H fail to make sense chemically?
A: A hydrocarbon is an organic compound that is composed of hydrogen and carbon atoms. The four classes…
Q: Which of the following structures is a triglyceride? *
A: Triglycerides are esters derived from glycerol and three fatty acids. Ester bonds are formed…
Q: Kevin was experiencing heartburn prior to his heart attack, for which he was taking an antacid…
A: In the antacid tablets, calcium carbonate acts as an active ingredient. This ingredient is…
Q: Assume we have water contained in a beaker. How many types of forces are there in this setup at…
A: The shape of the water molecule is V shape. It consists of two hydrogen atoms and an oxygen atom. A…
Q: If one compound has the formula C5H10 and another has the formula C4H10, are the two compounds…
A: Isomers are molecules or compounds with identical molecular formulas which means they have empirical…
Q: Estimate the molecular weight of a B-a-B supersecondary element that is 3 nm high?
A: Supersecondary structures (also called motifs ) are characteristics folds in proteins composed of 2…
Q: What structure is indicated by the number 2 in the image below?
A: Question - what structure indicated by number 2 ?
Q: In the Equation: HCO3- + HCl --------> Conjugate Acid + Conjugate Base a ) Which reactant…
A: An acid base reaction is a type of chemical reaction that involves transfer of one or more hydrogen…
Q: Glycosphingolipids are?
A: Biomolecules are organic molecules comprised mainly of carbon, hydrogen, and oxygen. These molecules…
Q: How does the atomic structure or composition of the versions of sodium in question C above differ…
A: According to the question, We have to explain how does the atomic structure or composition of the…
Q: A compound with empirical formula C2H5O was found in a separate experiment to have a molar mass of…
A: Molecular formula gives the kind and number of atoms of each element present in the molecule of the…
Q: How many H2O particles were produced by creating the following structure? * R H R R H-N-C,-c-…
A: Amino acids are monomers of protein they are linked with each other by forming peptide…
Q: HH нно н но HHO H-N-CC-OH H-N-C C-N-C-C-OH H-NC C-OH CH2 CH2 HC-CH3 CH3 CH2 CH2 HC-CH3 CH3 CH3 ČH3…
A:
Q: What kind of isomers are they
A: Carbohydrates are simple monosaccharides that are made up of carbon, hydrogen, oxygen and have…
Q: The configuration of the given structure below is: * CH,OH он ÓH O alpha-L O alpha-D O beta -L O…
A: The carbohydrates can be represented by two types of configurations: L-isomer and D-isomer. When the…
Q: Column A shows the names of some of the important functional groups. Column B shows their…
A: A specific group of atoms that is present within a molecule and is responsible for the…
Q: Why does the water on glass (left) look flat, while the water on the wax paper (right) looks…
A: Water is a unique solvent because of its unique physical and chemical properties.
Q: What is Secondary Structure?
A: The science of chemical reactions that take place inside or relate to live beings is known as…
Q: Is silica polar or nonpolar?
A: Chromatography is a technique that is used to separate a mixture of substances that are dissolved in…
Q: A Transmission Electron Microscopy image of Gold nanoparticles is shown below. Name a physical or…
A: Introduction Nanoparticles are one-of-a-kind structures with one-of-a-kind features due to their…
Q: what is chemical kinetic and how can you relate chemical kinetics in a real-life situation?
A: Chemical reactions are reactions that convert one chemical compound into another by the action of…
Q: How many electrons does fluorine have? How manyelectron shells? Name the orbitals that are…
A: Fluorine is a chemical compound with the symbol F and the atomic number is 9.
Q: What is sievert?
A: Radiation It is the emission or transmission of energy in the form of waves or particles through…
Q: Exactly what causes the charges to form on atoms in ionic bonds?
A: Atoms lose or gain electrons to complete their octet.
Q: what subatomic particle is present in the nucleus of an atom?
A: The subatomic particles are present inside the atom. Each subatomic particle has a unique charge…
Q: Why does butter is solid in room temperature, while vegetable oil is liquid?
A: Fats and oils are commonly called as triglycerides. Mainly these are of two types saturated and…
Q: H C-C-N-H エ-
A: Condensed structure formula is written in a line text and it is less extended form than normal…
Q: Are the following items chiral or achiral? Give a justification for your answers.(a) A fork (b) This…
A: Chiral : A chiral object in not identical in all respects with its mirror image. Achiral : An…
Q: What characteristic must a compound have to experience the following intermolecular forces?
A: There are three characteristics : London dispersion forces Dipole-dipole forces Hydrogen bonding
Q: What do you mean by woronin bodies?
A: Ascomycota is a phylum of kingdom fungi. The members of phylum ascomycota are known as sac fungi…
Q: Indicate whether the following familiar objects are chiral or nonchiral: (a) a glove; (b) a tennis…
A: Introduction: A chiral atom is a sort of particle that has a non-identical representation.
Q: HH H H H C-C- Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η H H H-C-O C- онн C-C- H H H H H H H CH, H H…
A: There are many macromolecules present in the living organism. They include carbohydrates, lipids,…
Q: Identify all of the chirality centers in the structure. `NH OH a h
A: The chiral center or atom (in this case carbon) has four different groups or atom attached to it.…
Q: нннн ннннН с-С-с-с-с-С-С-С-С-с-н HHHH HHHH H H. Ising the graphic above, answer the following…
A: Introduction: Life is made up of lifeless chemical molecules. The natural compounds known as the…
Q: Consider the structure shown below. он CH, O 3 5 CH; O CH; O 1…
A: The amino acids serine (Ser), threonine (Thr), cysteine (Cys), asparagine (Asn), glutamine (Gln),…
Labeling anatomical directions
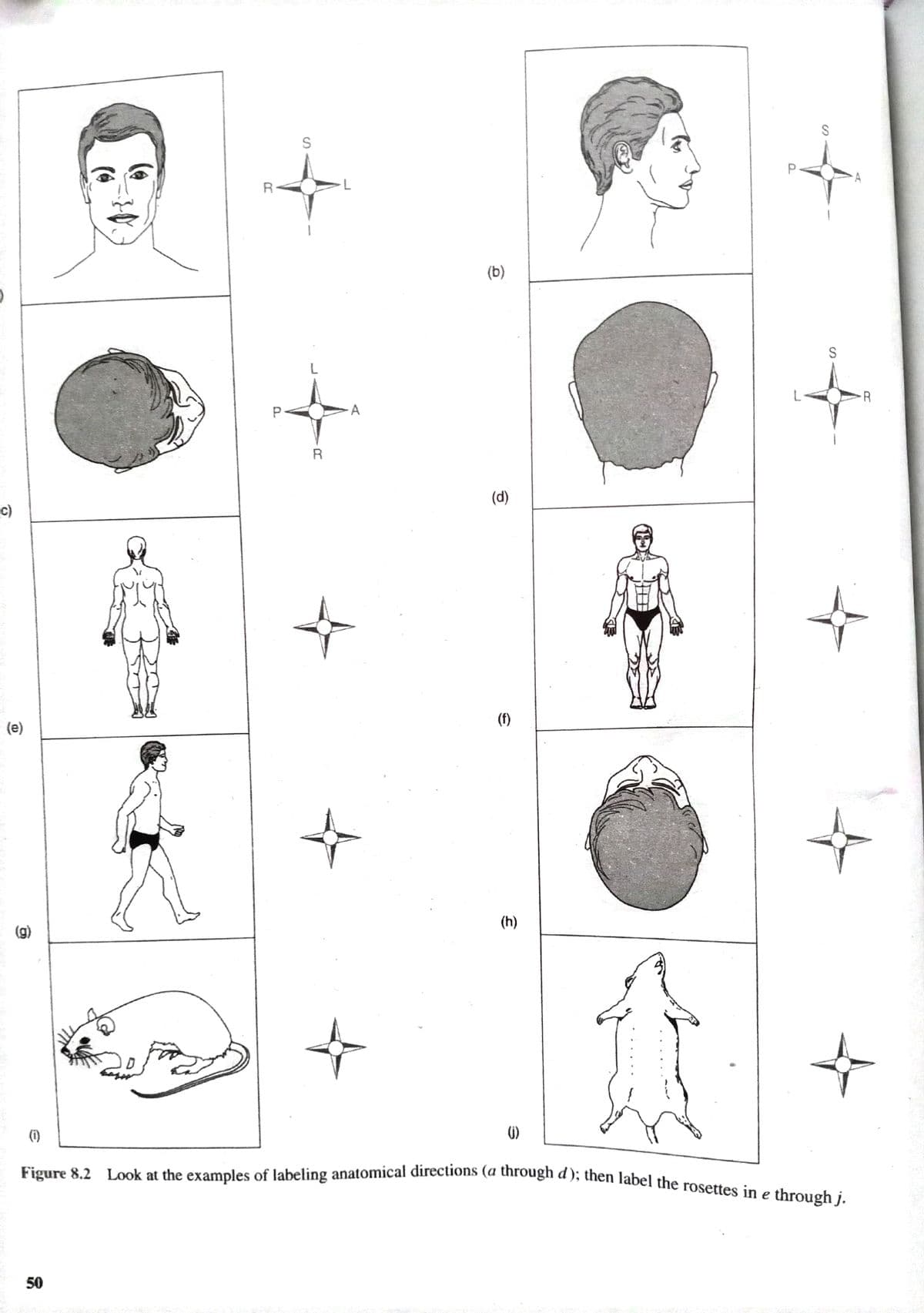
Step by step
Solved in 2 steps

- Number 3?the pedal region is _____ and ____ to the patellar region?When using word components to relate medical terms to the structure and function of the human body, why is it important to be able to break down medical terms into component parts? Why not just memorize each term? What is the point of learning all these Latin terms, anyway? Aren't they just an outmoded tradition?

