Q: How many mL of 8.00 M HCl are needed to prepare 150. mL of a 1.60 M HCl solution? (a) 30.0 mL (b)…
A:
Q: Question 9 AC 2.5 (1) The halogens can all act as oxidising agents. What is an oxidising agent? (ii)…
A: Oxidising agent, reducing agent ; these are the terms from redox reaction.
Q: What makes ethyl acetate a suitable or unsuitable solvent? What concerns would you have if you were…
A: We have find out the answer.
Q: Given the following unbalanced redox reaction: Fe(OH)3 + OCl- → FeO42- + Cl- What is the oxidation…
A:
Q: Consider the following reaction: Cd(s) + Fe2+(aq) →Cd2+(aq) + Fe(s) E= (Fe2+ / Fe) = −0.4400 V,…
A: Given, Cd(s) + Fe2+(aq) →Cd2+(aq) + Fe(s) The reduction potential is, Cathode - E= (Fe2+ / Fe) =…
Q: The formation of methane from carbon and hydrogen has K, = 8.1 x 108 at 298 K. C(s, graphite) +…
A: Answer: In this question formation reaction of methane is given to us and by formulating ICE table…
Q: What is the electron pair geometry of N in NH3? (a) Tetrahedral (b) Trigonal Planar (c) Square…
A:
Q: 28. Water is heated from 25°C to 100°C. By how much does its chemical potential change. assume the…
A: Given, Initial Temperature = 25oC = (273 +25)K = 298K Final Temperature - 100oC = (273 + 100)K =…
Q: 2. What is the value of AG°f for BaCO3 (s), if AG° = +218.1 kJ for the reaction BaCO3 (s)→ BaO (s) +…
A: The standard formation of gibbs free energy change is defined as the change in gibbs free energy…
Q: 33.79 g of a solid is heated to 100.°C and added to 50.0 g of water in a coffee cup calorimeter and…
A:
Q: 4) Given the reaction and data below: 2 NOCL (g) → 2 NO(g) + Cl2 (g) [NOCI]o (molecules-cm-3) 3.00 x…
A: a. Rate law can be determined from the order of reaction with respect to NOCl. Rate = k[NOCl]^x…
Q: Listen True or false? At equilibrium, the concentrations of all reactants and products are equal.
A: Given : statement about equilibrium
Q: The reaction for the decomposition of PCl5 to chlorine and PCl3 is shown below.…
A: Since,Equilibrium constant or Kc is the ratio of the equilibrium concentrations of product over…
Q: What happens to the Vmo and Vphth if the burette was contaminated with KOH in soda ash sample? A.…
A: Usually, soda ash contains sodium carbonate and often sodium hydroxide and sodium bicarbonate.…
Q: A soda ash sample was contaminated with NaOH. It was made up of 0.00069 moles of NaOH and 0.00115…
A: To calculate the expected volumes of Vphth and Vmo for a soda ash sample consisting of 0.00069 moles…
Q: If a column was run on the sample shown in lane 3, what would be the first compound to elute off of…
A: We have find out the answer.
Q: How many electrons are involved in the half-cell reaction: IO3- → I2?
A: Electrochemistry is branch of chemistry in which we deal with oxidation and reduction reaction…
Q: How many resonance structures, if any, can be drawn for the nitrate ion (NO3-)? (a) 1 (no resonance)…
A:
Q: Given the two reactions 3. PbCl₂ (s) 4. AgCl(s) Pb²+ (aq) + 2Cl(aq), K3 = 1.85x10-10, and Ag+ (aq) +…
A: Characteristics of equilibrium constant are : - If the equation (having equilibrium constant K) is…
Q: According to Le Chatelier’s Principle, does the following reaction shift towards reactants, towards…
A: Answer:- This question is answered by using the simple concept of Le Chatelier principle which…
Q: A reaction has a half-life of 11.6 minutes. Calculate the rate constant for this reaction in sec¹¹.
A: The time required to consume the half of the initial concentration of the reactant in a reaction is…
Q: In order to form a tertiary alcohol, excess CH MgBr could react with which one(s) of these…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: a) Draw the products of each acid-base reaction, using arrow pushing notation to show the bond…
A: We have to perform given Reaction.
Q: In the analysis of soda ash experiment, what will be the expected relationship between Vphth and Vmo…
A: A soda ash sample is given to have 0.0450 moles of NaOH and 0.0125 moles of NaHCO3 . we are required…
Q: 16. What is buffer capacity? a. The maximum amount of H+ or OH- ions that a buffer can absorb…
A:
Q: Use standard reduction potentials to calculate the standard free energy change in kJ for the…
A:
Q: When acetylene gas, C₂H₂, burns, how many liters of O₂ at STP are used for every 7.78 mol of…
A: Given, Moles of the Acetylene = 7.78 mol STP means Standard Temperature and Pressure. Th formulae…
Q: How many grams of N2(g) are in 38.7 L at standard temperature and pressure (STP)?
A:
Q: 57 A radioactive element X decreases from 360 mg to 90 mg in 1800 seconds. What is the half life of…
A: Answer:- The radioactive decay follows the first order kinetics. So, using the integrated rate law ,…
Q: H COOH 1. NaBH4 2.H₂O, HCI 3
A: NaBH4 reduces CHO and ketone to alcohol leaving acid and ester untouched
Q: What is the major product of the following reactions? α Br Br 1) BH3, THF 2) H₂O2, NaOH Br₂, hv…
A: BH3. THF, NaOH, H2O2 with terminal alkyne compound leads to anti Merkonikove addition of H2O…
Q: Dissipation of the proton gradient is required for a. None of the options are true b. Overcoming…
A: Here in this question we are asked a multiple choice question According to the question Ques :…
Q: SN1: Comparison A В HO: HO: НО of Leaving Group: нс Ҷсс нс н с HCC-Br Н,С Н.С. Hcmc-a НС Н.С. H₂CC-F…
A: Unimolecular nucleophilic substitution reaction (SN1): This reaction proceeds in two steps. The…
Q: Examine the following half-reactions and select the strongest reducing agent among the species…
A: Given set of equations: HgO(s) + H2O(l) +2e- ==> Hg(l) + 2OH-(aq) E° = 0.0977 V Zn(OH)2(s) +…
Q: 4 Listen You mix 100.0 mL of a 0.100 M NaOH solution and 150.0 mL of a 0.100 M HCI solution.…
A:
Q: How do I find out how many neutrons an atom has?
A: Neutrons are the building particle of an atom, these are neutral particles it contributes to the…
Q: A sample of a gas at 3.67 atm pressure and 14°C has a volume of 75.0 mL. The pressure is lowered to…
A:
Q: The mass of the object is 298.9 g. It is determined experimentally that it takes 19.2 J to raise the…
A:
Q: 5. A tiny piece of paper (produced from formerly living plant matter) taken from the Dead Sea…
A:
Q: How many moles of MgCl2 are present in 45.0 g of MgCl2?
A: Since, Moles is the ratio of mass to the molar mass of that atom or molecules. Thus,
Q: I got a yield of 30% for a lab where we converted 1-butanol to 1-bromobutane using HBr and sulfuric…
A: The percent yield of a reaction can be obtained by dividing actual yield by theoretical yield and…
Q: Two electrodes are combined to construct an electrochemical for Sn². Calculate Eº for the following…
A:
Q: T Part A A250 mL sample of ozone gas at 15 °C and 0.490 atm is cooled to a pressure of 0 340 atm. If…
A:
Q: What is the potential of a non-standard condition galvanic cell formed by an Al electrode in a 1M…
A: A galvanic cell is a type of electrochemical cell that uses a spontaneous redox reaction to generate…
Q: Calculate the average rate of production of H2 between 0 and 37.9 s. Now calculate the rates for PH3…
A: To calculate the rate average rate of disappearance of PH3 and the rate of production of H2 0 and…
Q: Select all that apply. Wrong answers count against you. Which of the following synthetic strategies…
A: The ether synthesis is mainly done by two processes. 1. dehydration of alcohol in presence of…
Q: rent Attempt in Progress What is the predicted product of the reaction sequence shown? NHCH3 Jymen…
A:
Q: QUESTION 24 Calculate the heat of the reaction, in kJ, using the standard enthalpies of formation…
A: Use formula, standard enthalpy change All data given,
Q: Listen Which of the following is an example of a heterogeneous equilibrium reaction? a) C₂H₂(g) +…
A:
Q: A reaction is observed to have a rate constant of 35 min ¹. Calculate the corresponding half-life…
A: For first order reaction, the unit of rate constant is (time) -1 And the half life calculation can…
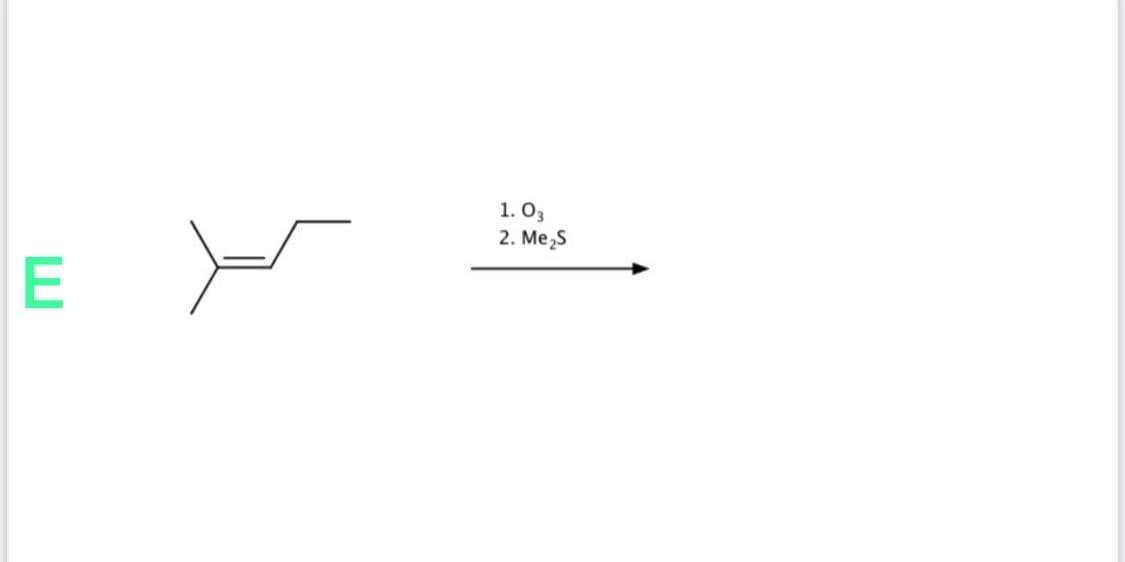
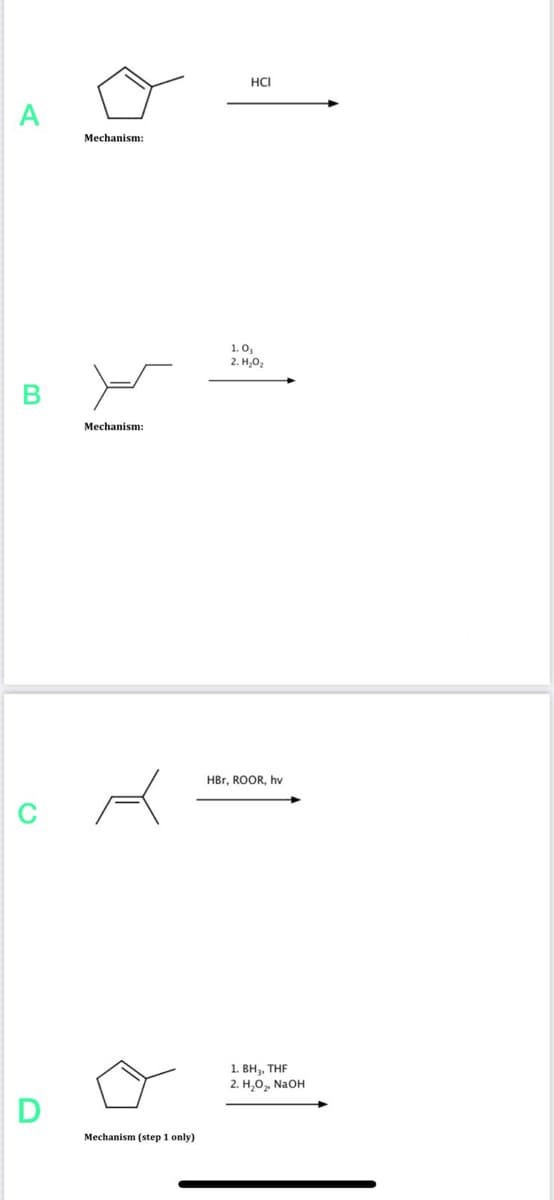
Step by step
Solved in 2 steps with 2 images

- What type of mechanism is exhibited in the reaction? SN1, SN2, E2, E1?The substitution of an Iby a Clon H3C-I can occur by two possiblemechanisms:Mechanism I: step 1. H3CI → H3C+ + I- slow stepstep 2. H3C+ + Cl- → H3CCl fast stepMechanism II: step 1. H3CI + Cl- → H3CClI slow stepstep 2. H3CClI → H3CCl + I- fast stepa.) Write a rate law for each reaction.Give 3 examples of a reaction mechanism of E1 that follows Zaitsev's rule.
- For each reaction indicate the predominant mechanism: SN1, SN2, E1, or E2For which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be true for one or moremechanisms. The reaction rate increases when the solvent is changed from CH3OHto (CH3)2SO.Q. Carry out following conversion. Provide suitable reagents, reaction conditions and also mechanism of the reactions. Do (iii) as soon as possible.
- Q. Carry out following conversion. Provide suitable reagents, reaction conditions and also mechanism of the reactions. Do (ii) as soon as possible.Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?When Is the Mechanism E1 or E2?
- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- ΔH=-75 kJ/mol Rate = k [CH3CH2Br][CH3COO-] Which reaction energy profile would be the best representative of the data provided?See reaction below and write a step-wise mechanism on how this reaction occurs involving the catalyst.Give one example of elimination reaction (E2) type. Show the reactants materials and the product. Draw the reaction mechanism?

