Q: The maximum recommended single dose for acetaminophen is 15.0mg per kilogram of body weight for…
A:
Q: Give the reaction mechanism for the following: a. Aldol condensation b. Claisen condensation…
A: In an aldol condensation, the enolate ion combines with a carbonyl molecule to produce a…
Q: 2 3 Compound Structure H CIH H |||| H-C-C-C-C-H |||| HHH CH3 C₂H₂ H F TIL H CH3-C-C-C=C || H F H Br…
A: The correct nomenclature of the above organic compounds according to the IUPAC rule is given below
Q: The Ozone gas at STP contains (6.022 x 10^23) molecules. O Flase O True
A: Solution : The fomula of ozone gas is O3. STP means Standard Temperature pressure. The conditions of…
Q: Calculate the molality of (7.45 g) KCI, which dissolved in 100 g water. (molar mass: H-1, O=16, K=…
A:
Q: In the laboratory you dilute 4.46 mL of a concentrated 6.00 M hydrochloric acid solution to a total…
A: If M1 and V1 are the initial molarity ( concentration )and volume and M2 and V2 are the final…
Q: Part of Dalton's theory were correct is * O All matter is composed of atoms O Atoms are indivisible…
A: Dalton who is a greek philosopher gave us a idea about the concept of the indivisible particle of a…
Q: Given the organic molecule, determine whether it is polar or nonpolar. Explain your answer.
A: Ketones can be defined as the organic compounds that contain carbonyl group in a compound.
Q: If a solution containing 109.90 g of mercury(II) nitrate is allowed to react completely with a…
A: Here, we have to find the mass of the precipitate that will be formed when 109.90 g of mercury(II)…
Q: mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude…
A:
Q: Foods are composed of a variety of biomolecules. Classify each food item based on the primary type…
A: In the given problem we have to classify each food item based on based on primary type of molecules…
Q: which if the following compounds is an electrophile? H20 (PO4)3- NH3 BF3
A:
Q: IR Spectrum (liquid film) 4000 100 80 Lalalalalalala % of base peak 3000 40 13C NMR Spectrum (50.0…
A: Given the molecular formula of the unknown is C7H7Br We are given the IR spectrum, mass spectrum,…
Q: Atoms which form covalent bonds not have any valence electrons. O False O True
A: Covalent bonds are form by sharing of electrons from the bonded atoms.
Q: Q2. Give a list ONLY for the following items: 1. The main bases on which crystallographic systems…
A: We have asked to give only list for the item given and the answer to this question is given in…
Q: 3 3 H [ H H T H T I H OH + -OH 2 K2C1207 8 H2SO4 Cr (VI) 2 C2)SO43_ +11H20 + 2K2$04 Cru + + ܀
A:
Q: Calculate the stoichiometric ratio of Fe3O4 in Fe2O3.
A: Iron (II, III) oxide undergoes oxidation to form Iron (II) oxide.
Q: IR Spectrum 4000 100 3000 40 13C NMR Spectrum (500 MHz, CDC, solution) 80 DEPT CH CH. CH proton…
A:
Q: a le tion of mbrane Part A What is the height difference between the two water surfaces at a…
A: As per the guidelines, I am allowed to answer one question at a time. Please re upload the rest.
Q: Chemistry mutation asp in 1mry?
A: In the question I suppose we need to describe what it means mutation asp in 1mry.
Q: Oxidation process can happen between metal and organic solvent. O True O False *
A: Here we have to predict the true and falseness of the following given statement.
Q: Q12. Convert 876.9 in.³ to m³. (a) 0.01437 m³ (c) 5.351 x 107 m³ 3 (b) 22.27 m³ (d) 0.014 m³ me
A: Convert in3 to m3
Q: 0.080 m K₂ CO3 Choose versus lowest vapor 0.096m kel pressure & explain
A:
Q: For the first-order reaction 1/2 N204 -> NO₂(g); AH = 28.6 kJ the rate constant is k= 3.24 x 10^5…
A:
Q: Determine the number of molecules of oxygen (0₂) required to complete the following chemical…
A:
Q: For a compound with the molecular formula C6H8O, which of the following must be false? The compound…
A:
Q: What is the evidence that a chemical reaction took place?
A:
Q: In the periodic table: Elements in the same group have the same valence charges. O True O False
A:
Q: : In some application, there are used C,Hy as a fuel to produce energy, during analysis gases of…
A: Fuel gas means gas generated at a petroleum refinery or petrochemical plant and that is combusted…
Q: In the relation, W=JxQ, 1 calorie = 4.18400 joules false true
A: Thermodynamics is branch of chemistry in which we deal with amount of heat evolved or absorbed by…
Q: Draw the displaced formula for CH3CH2COCH2I Also name of CH2CH2COCH2I
A:
Q: Tuture is 220 R. B- The pressure in a vessel containing methane and water at 70°C is 10 atm. At the…
A: Given : Temperature = 70°C Pressure = 10 atm Vapour pressure of water at 70°C = 0.3117 bar…
Q: You are given 2 test tubes. One of it consists of glucose and the other maltose. Which of the tests…
A: Glucose is a monosaccharide which is an aldose sugar. Maltose is a disaccharide that is made of two…
Q: The periods of the periodic table shows periodic trends True False M 2 *
A:
Q: Isotopes means; same element can have different masses. O False O True
A: Please find your solution below : An atom is the smallest unit and basic building block of matter…
Q: Sulfur dioxide reacts with carbon dioxide to form an equilibrium with sulfur trioxide and carbon…
A:
Q: If someone consumes 21 g of sodium chloride per day, what mass (in grams) of sodium does that person…
A:
Q: Isotopes means; same element can have different masses.
A: Isotopes mean; same element can have different masses: True Explanation: Isotopes are a family of…
Q: 2. Using structural diagrams to explain the synthesis of butyl ethanoate from an alkane and an…
A:
Q: In the periodic table: Elements in th same group have the same valence charges. O True O False
A: Given statement : Elements in same group have same valence charges.
Q: Experiment Temprature/K/ diff temp Bate Constant/s! 3ܩ3 - ܢܠ 333 ܒܕ ܐ ܣܛ559ܩܙ -ku ܠܰܩܠܫܬ7 ܕܥ ܘܤܠܒܘ ܐ…
A:
Q: Draw the skeletal formula of a functional grouo isomer of 2-methylbut-1-ene
A:
Q: How much CO2 is formed by adding 5 g of baking soda to 75.0 ml of vinegar
A:
Q: Give a clear handwritten answer with explanation..complete the following reaction with balancing…
A: In this question, we will completed the given three reactions by finding the product. You can see…
Q: . Show 2 repeating units of the polymer produced by styrene - refer the diagram attached H₂C-C H-O…
A:
Q: The work done by the system on the surrounding is nRT Inv2/v1 O +jvdp O-Jvdp O +jpdv DRT Inv2/x1
A: Dear student , since you have posted multiple questions we will allow to solve only first question…
Q: 1. Write the hybridization of each carbon atom:
A: Organic compounds are the compounds that contain carbon and hydrogen atoms.
Q: Q1/ Complete the following reactions and mentioning name of the reaction? Et NaOH &. d ? H + CH3l 2)…
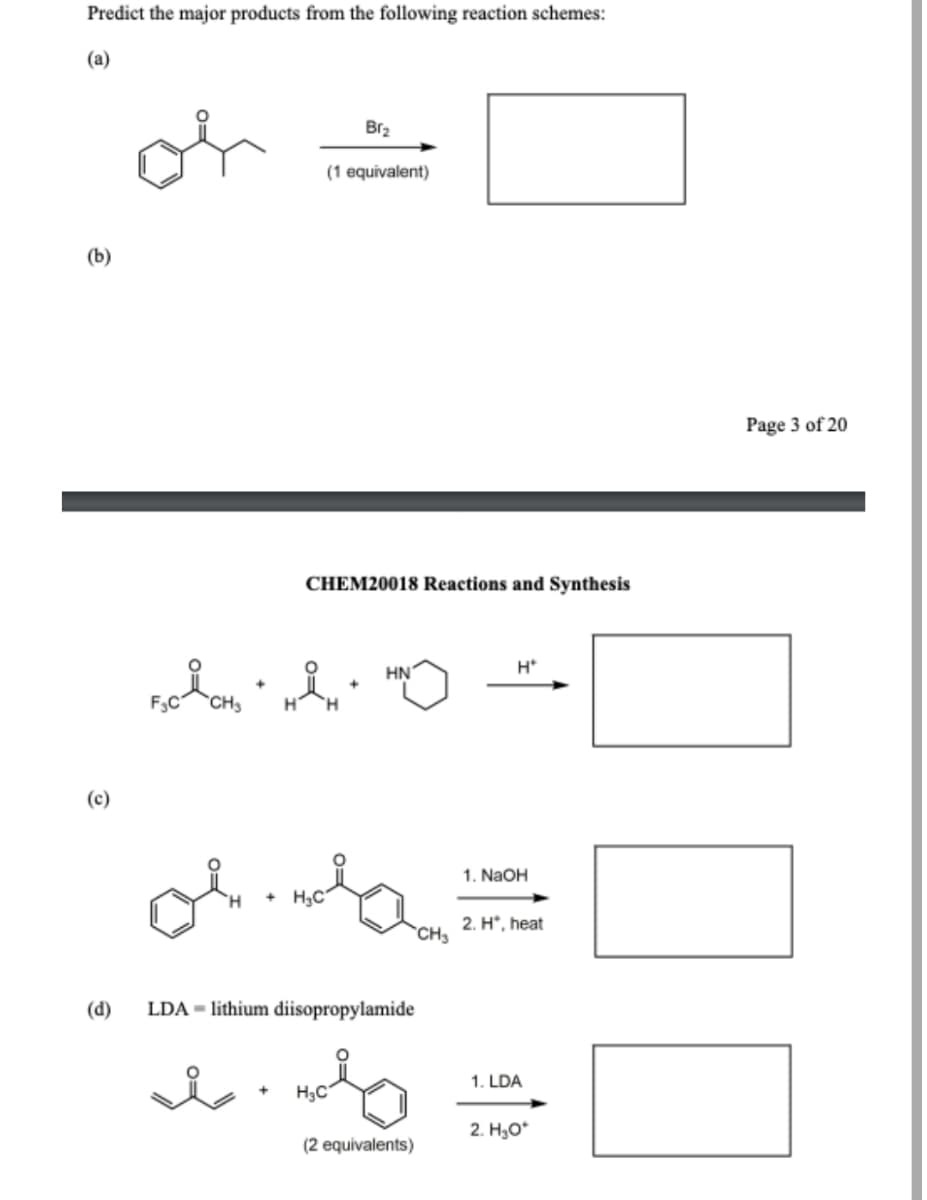
A: 1) Robinson annulation reaction - The method uses a ketone and a methyl vinyl ketone to…
Q: CH₂ H -9 10 C OCH, step 1 CH, Ge Ch step 2 CH₂-C C step 3 orts CH,OH
A: To answer : Three curly arrows to complete the given mechanism Type of reaction involved Role of…
Q: B) Give correct name for each of the following compounds: CH₂ H₂C OH 1) OH CH 3) 27 CH₂ 4) NH₂ Lor…
A: Using IUPAC namenclature rule.
Step by step
Solved in 2 steps with 1 images

- I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?20- In the lab, you prepared phenylmagnesium bromide reagent using 0.81 g magnesium turnings (MW- 24.31 g/mol) and 3.6 mL bromobenzene (MW- 157.01 g/mol, density- 1.50 g/mL) in 15 mL diethyl ether (MW-74.12 g/mol, density - 0.713 g/mol). You then added 2.04 g benzophenone (182.217 g/mol) which was previously dissolved in 6.0 mL of diethyl ether solution to the phenylmagnesium bromide solution.What is the theoretical yield for triphenylcarbinol (MW- 260.33 g/mol) in grams? with the appropriate number of significant figures.Pls solve this question correctly in 5 min i will give u like for sure Balance the chemical equation of a reaction formed by the bromination of trans-cinnamic acid if we started with 0.3088 grams of trans cinnamic acid and 1mL of bromine. The product of this reaction is 2,3 - dibromo - 3- phenylpropanoic acid
- Give the name/s only (structures not required) of the product for the reaction of (i) A and (ii) B with the reagents listed below. Your answers should be given using A1 -A10 and B1-B10 as indicators.Eg. the product for your first reaction should be stated as A1 = name of product etc. b2h6/h202 n-bromosuccinimide kmno4/oh-In synthesizing primary (1⁰) amines by alkylation of NH3, explain how we can prevent the formation of 2⁰ and 3⁰ amines as by-products? You can give an example to help you in explaining but it is not required.Given: Mass spec for 4-acetylbiphenyl. Knowing that 196 m/z is the major product, how would you assign 181 m/z and 152 m/z? (i've also attached a picture of the reaction scheme)
- please provide the machanisms of 1a, 1e, 1fHi,I need help with this chemistry question, please and thank you From the reaction between pyrrolidine and 2-methylcyclohexanone in an acid medium, enamines I and II can be formed. Describe which of the two is formed preferably and explain why?An organic lab student carried out the reaction of methylmagnesium iodide with acetone(CH3COCH3), followed by hydrolysis. During the distillation to isolate the product, she forgot to mark the vials she usedto collect the fractions. She turned in a product of formula C4H10O that boiled at 35 °C. The IR spectrum showed onlya weak O¬H stretch around 3300 cm-1, and the mass spectrum showed a base peak at m>z 59. The NMR spectrumshowed a quartet (J = 7 Hz) of area 2 at d 3.5 and a triplet (J = 7 Hz) of area 3 at d 1.3. Propose a structure for thisproduct, explain how it corresponds to the observed spectra, and suggest how the student isolated this compound.
- Give answer ASAP Thanks What is the role of 10% ammonium hydroxide, sodium hydroxide, and 2 M and 6 M HCl in the synthesis of Pentaamminechlorocobalt(III) chloride derived from Chloropentamine Cobalt (III) Chloride?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…

