Q: m) CH3CH₂-C-CH₂CH3 +2CH3CH₂OH OCH₂CH3 CH3CH₂-C- CH₂CH3 OCH2CH3 + H₂O
A:
Q: Porantherine (molecule M) is the main alkaloid of the low woody shrub Poranthera corymbosa…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: MgBr + CH3CH₂ND₂ (A) I (B) II -H (C) III diethyl ether III (D) IV -ND₂ IV -NDCH₂CH3
A: The given reaction explains the reaction between grignard reagent and deuterated amine to form…
Q: НО. НО. H* (catalytic)
A: When salicylic acid is treated with acetic anhydride, acetylsalicylic acid is produced which is…
Q: g) OH CH3CHCH2CH3 11 CH3CH=CHCH3 + H₂O alkent
A: Dehydration reactions can be defined as the chemical reactions in which a water molecule is…
Q: 4. Calculate [H,O'], [OH ], pH, and pOH for 6.5 x 10 M H₂SO, solution (at 25 °C). a. [H,O*]. b. [OH]…
A:
Q: Give the balanced overall chemical equation for photosynthesis. Express your answer as a chemical…
A: In photosynthesis reaction carbon dioxide and water react in presence of sunlight and chlorophyll to…
Q: A solution is made by dissolving 33.5 g of Ba(NO₂)2 in 500.0 mL of water. Does Ba²+ have any acidic…
A: The reaction for the dissolution of Ba(NO2)2 in water can be written as Ba(NO2)2 + H2O → Ba(OH)2…
Q: 4. Calculate the concentration of nitrogen gas in a 1.00 L container exerting a partial pressure of…
A:
Q: What is the IUPAC name for the following alkane?
A: The given compound is a cyclopropane derivative with two methyl groups. The detailed nomenclature is…
Q: QUESTION 1 A buffer solution contains a weak acid, HA, and its conjugate base,A". The buffer…
A: Information
Q: The sugar D-gulose is a sweet-tasting syrup. Identify the Fisher projection for its mirror image,…
A:
Q: mechanism for each step in the relevant place. ? ? products, reagents an ? ? OH ||||| ... OH 어....
A:
Q: - h) H OH CH3-CH-CHCH₂CH3 T CH3 014 H3O+ CH-CHICCHACH HO-H CH3 major product CHO CHỊCHCH,CHSAH-O-H…
A: Saytzeff's rule : It predicts the regioselectivity of the alkene formed by the elimination reaction…
Q: A current of 3.68 A is passed through a Sn(NO3)2 solution. How long, in hours, would this current…
A:
Q: 2u Cl₂ + 3H₂O + PPh ₂ → Ru (PPh z)z Cz озна тос през balance ++++ + H₂O₁
A: The explanation is given below-
Q: 3. Develop a scheme to separate tha", K and Ag" cations in solution and drew a flow chart that…
A: The question is based on the concept of qualitative analysis. We have to make a scheme for…
Q: {C-№ :O: H H ② 'NH2
A: This is the resonance canonicals of the benzamide molecule.
Q: Calculate the pH for each of the cases in the titration of 25.0 mL of 0.140 M pyridine, C5H5N(aq)…
A: The molarity of 25.0 mL solution of pyridine is 0.140 M. The molarity of HBr solution is 0.140 M.…
Q: Enter your answer in the provided box. Determine the concentration of a solution of ammonium…
A: Information
Q: What is the solubility of MgCO3 in water? (Ksp of MgCO3 is 3.5 x 10-⁹)
A:
Q: 100.0 mL of HCl required 5.831 g Mg(OH)2 to titrate. The molar mass of Mg(OH)2 is 58.31 g. What is…
A:
Q: Consider the following reaction and standard molar entropy values for each substance at 25°C.…
A:
Q: What are enantiomers? What instrument can you use to distinguish the enantiomers? What is racemic…
A: “Since you have asked multiple question, i will solve the first question for you. If you want any…
Q: 3. Calculate the boiling point of a solution made from 227 g of MgCl2 dissolved in 700. g of water.…
A: As mentioned we are solving question 3, we have to calculate the boiling point of the solution
Q: IZ ost D 1. NaOH, A 2. H3O+ 1. NaH 2. 요요 C A 1. CH₂N2 MgBr 2. H3O+ B O H
A:
Q: b. Complete and balance the following equations and explain your results. BaCl₂ (aq) + H₂SO4 (aq) →…
A: The question is based on the concept of chemical reactions. we need to complete the given reactions.
Q: PART A To calculate the half-life, plug the value for k� into the half-life equation and solve.…
A: Given, A. Rate constant = 8.20 x 10-4 s-1 The half life of a reaction is: B. Time taken for…
Q: kinetics experiment is performed to determine the activation energy of a reaction. The following…
A: We know that Activation Energy : the minimum energy needed for reactant molecule to form product.…
Q: Which of the following is the formation reaction for HCIO3 (g)? Group of answer choices A.) 2 H2 (g)…
A: Heat of formation is defined as the enthalpy change when 1 mole of a compound is formed from its…
Q: a. Scandium, Sc. A silvery-white metallic transition metal, it has historically been sometimes…
A: Answer: Electronic configuration is an arrangement of electron in increasing energy order inside an…
Q: urved arrows are used to illustrate the flow following reaction u Select to Add Arrows ~ صلاح Select…
A:
Q: 1. What is the major elimination product of the following reaction? Br NaOCH 3 CH3OH Br 2. List all…
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: Draw the mechanism for this reaction. If no reaction takes place, check the box under the drawing…
A: The reaction may passes through the formation of stable carbocation intermediate. To find out the…
Q: Calculate the pH of the solution after the addition of each of the given amounts of 0.0542 M HNO3 to…
A: Given, Molarity of Aziridine = 0.0750 M Volume of Aziridine = 60.0 mL Molarity of HNO3 = 0.0542 M
Q: The solubility of lead (II) chloride (PbC12) is 1.6 × 10-2 M. What is the Ksp of PbC12? X
A: Molar solubility of PbCl2 (s) = 1.6×10-2 Ksp for PbCl2 =?
Q: Consider the following Haworth structure of a disaccharide and identify the type of glycosidic bond…
A: First monosaccharide given here is alpha D glucose and second monosaccharide is Beta D fructose .…
Q: T Brz Brz 120 CC14 →>
A: The given reactions discuss about reaction of alkene with halogens in presence of CCl4 and water to…
Q: MgBr 1. PhCHO, then sat. NH4Cl(aq) 2. Jones reagent 3. Saegusa-Ito oxidation 4. Me₂CuLi, then workup…
A: To find out the product we have to look the stepwise reaction as follows
Q: Given below is the Fisher projection for a threose enantiomer. What will be the Fisher projection…
A:
Q: Write the complete mechanism for the acid-catalyzed hydrolysis of acetonitrile to ethanoic acid
A: Acetonitrile on acid-catalyzed hydrolysis gives ethanoic acid. We have to write the mechanism of…
Q: number of pi bonds? 1,1'-Diethyl-2,2'-cyanine iodide 1,1'-Diethyl-2,2'-cyanine iodide…
A: The pi bond is formed due to the sidewise overlap (perpendicular to the axis) of orbitals.
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Given reaction involves the acyl substitution by ethyl amine.
Q: 2. While a series of events take place within the airbag system, what ultimately causes the airbag,…
A: Since you have posted multiple questions, we will provide the solution only to the first question as…
Q: Predict the products of the following reactions: (a) NaBH(OAc)3 butan-2-one + diethylamine (c) (e)…
A: We have find out the answer.
Q: Formation of ionic compounds are 'enthalpy driven'. Briefly explain using CaO as an example.
A: The question is based on the concept of ionic bonding. ionic bonds are formed by complete transfer…
Q: 28. Refer to the diagram below to determine a. the boiling point of ethanol when it is at normal…
A: The temperature at which liquid and vapour exist in dynamic equilibrium and vapour pressure of the…
Q: 11. NaOH is a common strong base in chemical laboratories. It is sold as solid pallets or as…
A: Given amount of Ba (OH2) is 2.267grams Volume of the solution is 200.00 ml Now We have to find out…
Q: Complete the table. (All solutions are at 25 °C.) Solution [H3O+] [OH-] pH 1 3.19 2 3 4 3.2 x 10-9…
A:
Q: 1.3 3 (i) In the Haber process for the production of ammonia, the following reaction occurs: 3H₂(g)…
A: The equilibrium constant - of a reaction is the ratio between the product's concentration and the…
please fill in the twobreactants to complete the
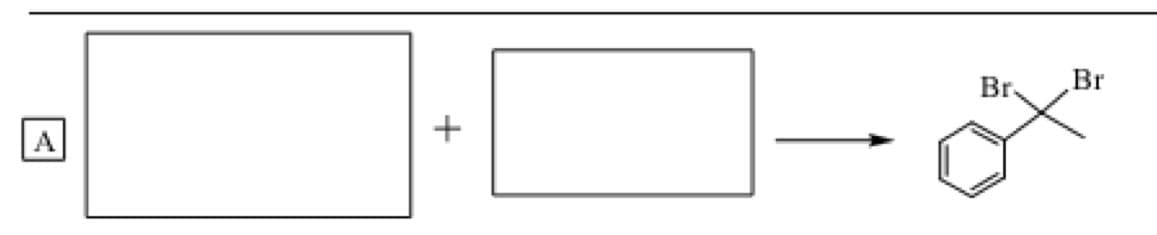
Step by step
Solved in 3 steps with 3 images


