A carbamate can be prepared by treating an isocyanate with an alcohol, as shown here. This type of reaction is used to synthesize polyurethanes- polymers that have a wide variety of industrial applications, such as surface sealants, high-performance adhesives, and synthetic fibers. Propose a mechanism for this transformation. R'—ОН R. `N=C=0 R. OR' An isocyanate A carbamate (Substituted urethane) O=C
A carbamate can be prepared by treating an isocyanate with an alcohol, as shown here. This type of reaction is used to synthesize polyurethanes- polymers that have a wide variety of industrial applications, such as surface sealants, high-performance adhesives, and synthetic fibers. Propose a mechanism for this transformation. R'—ОН R. `N=C=0 R. OR' An isocyanate A carbamate (Substituted urethane) O=C
Chapter19: Aldehydes And Ketones: Nucleophilic Addition Reactions
Section19.SE: Something Extra
Problem 36MP: It is not uncommon for organic chemists to prepare acetals by an exchange-type process known as...
Related questions
Question
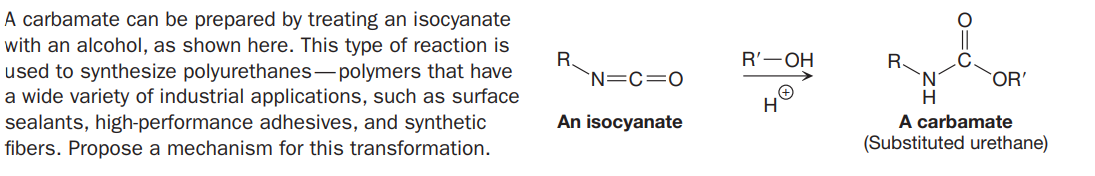
Transcribed Image Text:A carbamate can be prepared by treating an isocyanate
with an alcohol, as shown here. This type of reaction is
used to synthesize polyurethanes- polymers that have
a wide variety of industrial applications, such as surface
sealants, high-performance adhesives, and synthetic
fibers. Propose a mechanism for this transformation.
R'—ОН
R.
`N=C=0
R.
OR'
An isocyanate
A carbamate
(Substituted urethane)
O=C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning