A certain substance X has a normal boiling point of 141.7 °C and a molal boiling point elevation constant K,=1.58 °C-kg•mol '. Calculate the boiling point of a solution made of 1.0 x 10 (NH.),CO) CO dissolved in 950. g of X. g of urea Round your answer to 4 significant digits. PC Continue Submit Assignme 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibl MacBook Air 80 44 DII DD F3 F4 F5 P6 F7 PB %24
A certain substance X has a normal boiling point of 141.7 °C and a molal boiling point elevation constant K,=1.58 °C-kg•mol '. Calculate the boiling point of a solution made of 1.0 x 10 (NH.),CO) CO dissolved in 950. g of X. g of urea Round your answer to 4 significant digits. PC Continue Submit Assignme 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibl MacBook Air 80 44 DII DD F3 F4 F5 P6 F7 PB %24
Chapter11: Properties Of Solutions
Section: Chapter Questions
Problem 72E: A solution contains 3.75 g of a nonvolatile pure hydrocarbon in 95 g acetone. The boiling points of...
Related questions
Question
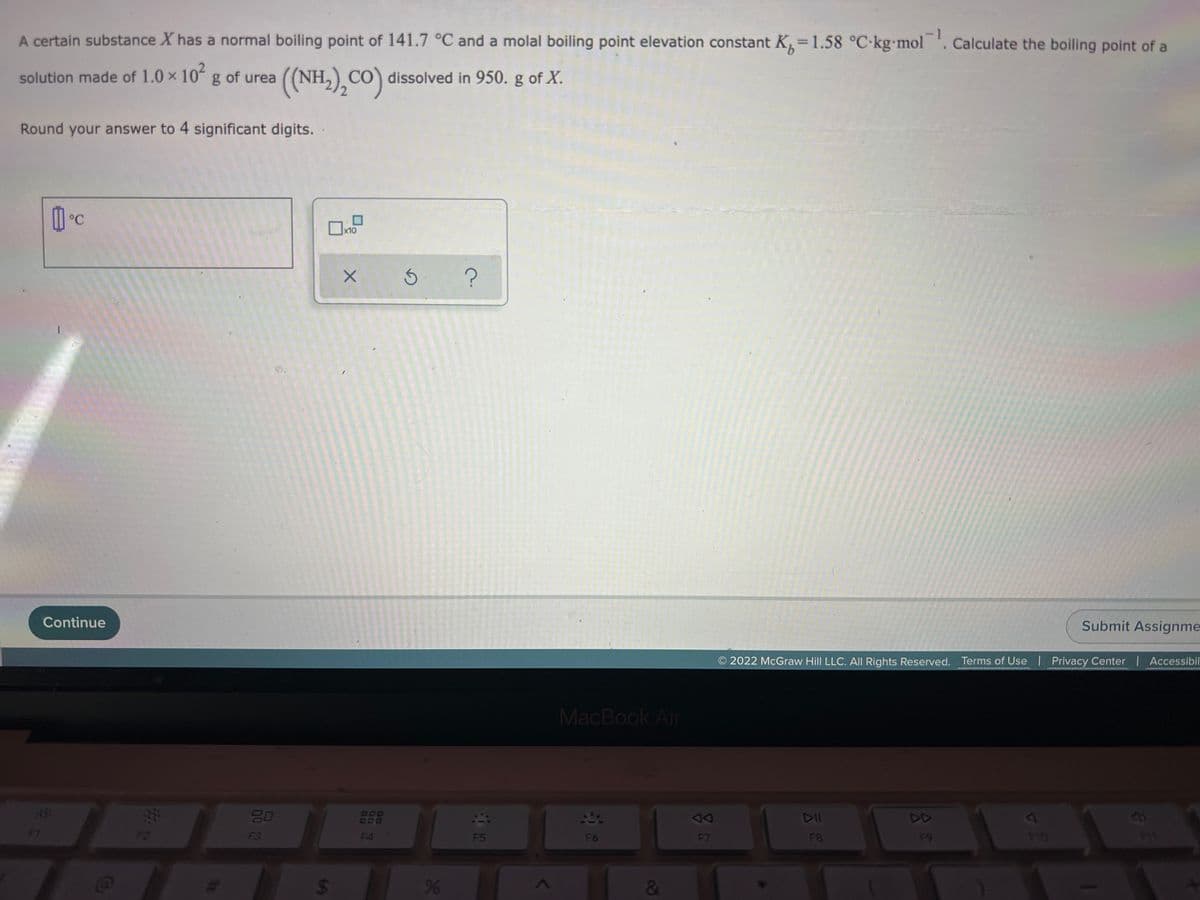
Transcribed Image Text:A certain substance X has a normal boiling point of 141.7 °C and a molal boiling point elevation constant K,=1.58 °C-kgmol . Calculate the boiling point of a
-1
%3D
solution made of 1.0 x 10 g of urea
((NH.),CO)
dissolved in 950. g of X.
Round your answer to 4 significant digits.
°C
x10
Continue
Submit Assignme
2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibil
MacBook Air
80
DII
DD
F3
F4
F5
F6
F7
F8
F9
&
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning