A certain weak acid, HA, with a Ka value of 5.61 × 10- is titrated with NaOH. Part A A solution is made by titrating 8.00 mmol (millimoles) of HA and 3.00 mmol of the strong base. What is the resulting pH? Express the pH numerically to two decimal places. femplates Symbols uAdo redo reset keyboard shortcuts help pH = Submit Request Answer Part B More strong base is added until the equivalence point is reached. What is the pH of this solution at the equivalence point if the total volume is 74.0 mL ? Express the pH numerically to two decimal places. femplates Symbols uado redo reset keyboard shortcuts help, pH =
A certain weak acid, HA, with a Ka value of 5.61 × 10- is titrated with NaOH. Part A A solution is made by titrating 8.00 mmol (millimoles) of HA and 3.00 mmol of the strong base. What is the resulting pH? Express the pH numerically to two decimal places. femplates Symbols uAdo redo reset keyboard shortcuts help pH = Submit Request Answer Part B More strong base is added until the equivalence point is reached. What is the pH of this solution at the equivalence point if the total volume is 74.0 mL ? Express the pH numerically to two decimal places. femplates Symbols uado redo reset keyboard shortcuts help, pH =
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter13: Acids And Bases
Section: Chapter Questions
Problem 191MP
Related questions
Question
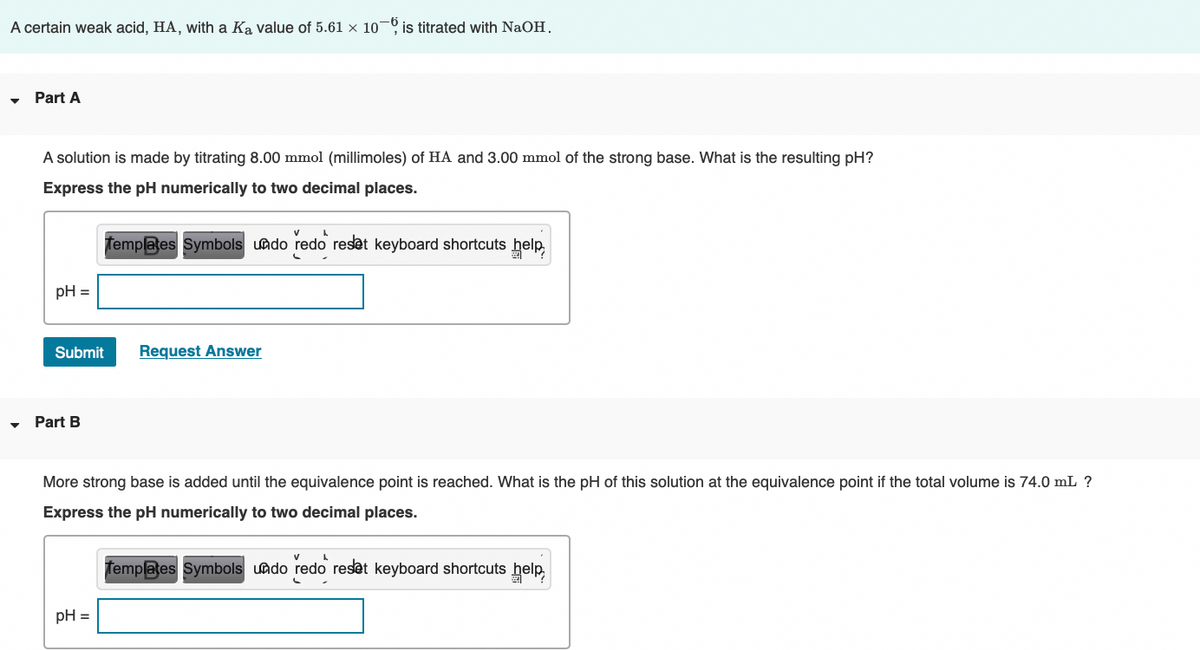
Transcribed Image Text:A certain weak acid, HA, with a Ka value of 5.61 × 10 is titrated with NaOH.
Part A
A solution is made by titrating 8.00 mmol (millimoles) of HA and 3.00 mmol of the strong base. What is the resulting pH?
Express the pH numerically to two decimal places.
Templates Symbols undo redo reset keyboard shortcuts help,
pH =
Submit
Request Answer
Part B
More strong base is added until the equivalence point is reached. What is the pH of this solution at the equivalence point if the total volume is 74.0 mL ?
Express the pH numerically to two decimal places.
Templates Symbols uado redo reset keyboard shortcuts help,
pH =
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER