Q: Determine which of these two compounds is more basic CH3CH2NH2 or CF3CH2NH2? Discuss further
A: Given amine substituted Compounds are - CH3CH2NH2 , CF3CH2NH2 Here amines are weakly basic in…
Q: Draw chair conformations for a-d-galactopyranose and b-d-galactopyranose. Label the anomeric carbon…
A: Carbohydrates are biomolecules which are composed of certain monomer units, called as…
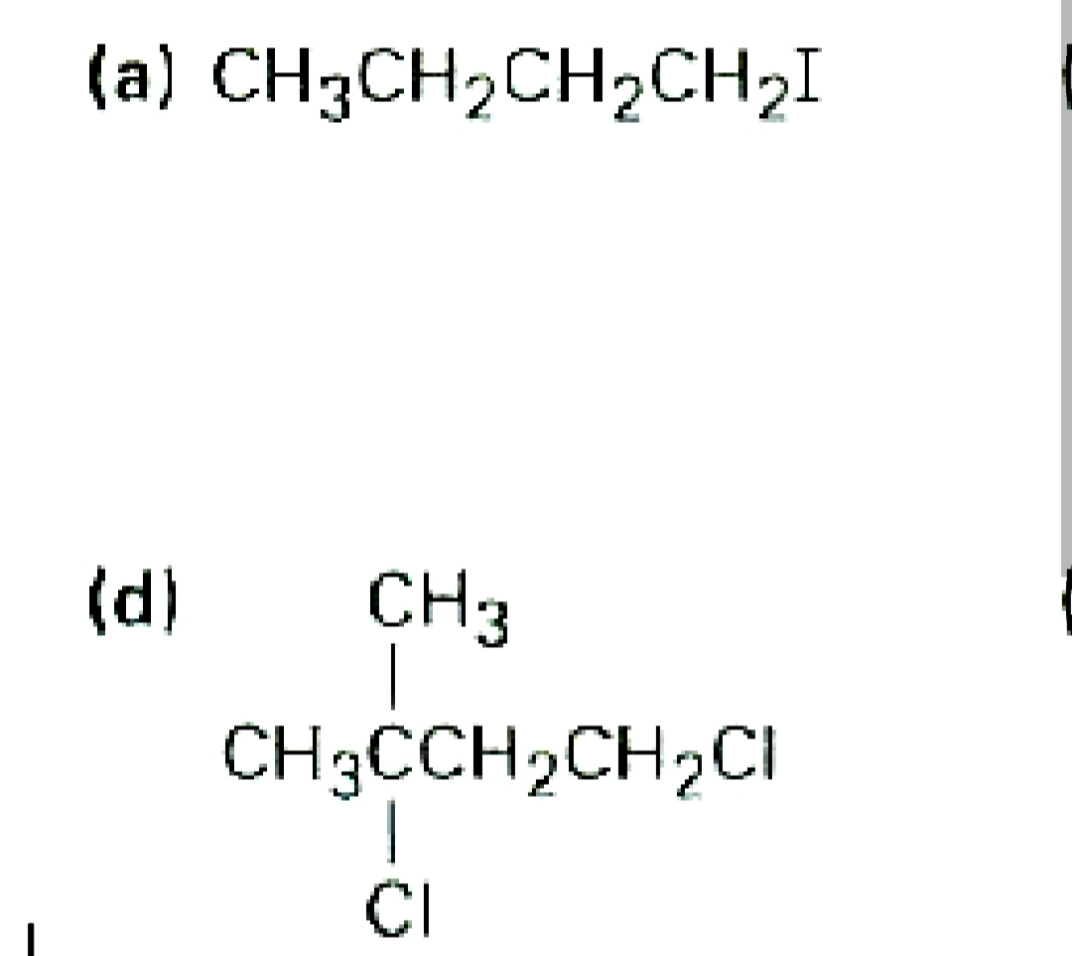
Q: . CH3CH2F B. CH3CH2CH21 - CHoCHoOH
A: The M+2 peak in a mass spectrum arises from the presence of chlorine or bromine atoms i.e a halogen…
Q: Draw and identify the structuresof glucose, its anomers, and itsepimers, both as Fischer…
A: Anomers and epimers are types of diastereomers. In epimers there is difference in configuration at…
Q: Tinal
A:
Q: provide chari conformations and chair flip, which one is most stable and why?
A:
Q: ed through an aque- the yolume of Or
A: m = ( M • I • t ) / nF m/M = ( I • t ) / nF n = I • t / nF Where, m = mass and M = molar mass and…
Q: Which reacts faster in an SN2 reaction?
A: In SN2 reaction the order of reactivity of alkyl halide is given below. Primary alkyl halide >…
Q: The pK, of CH3CH2CH2OH is than that of CH3CH2CH2SH.
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Which is more stable?
A: The stability of alkenes is majorly influenced by hyperconjugation. Hyperconjugation increases the…
Q: Define Stereogenic Centers in Cyclic Compounds ?
A: In a compound the center that bears substituents that are different from one another is called a…
Q: (b) Br CH3CHCH2CH2COH (c) CO2H CH3CH2CHCH2CH2CH3
A: IUPAC names : (b) 4-Bromopentanoic acid (c) 2-Ethylpentanoic acid
Q: Without referring to the chapter, draw the chair conformations of N-acetylglucosamine, glucose with…
A: The open structure of N-acetylglucosamine is shown below.
Q: Trehalose and isomaltose are both dimers of glucose. However, they have considerably different…
A:
Q: las a the of lactic acid and mark the chiral atom with *. Draw three-dimensional diagrams of the two…
A: The question is based on the concept of stereochemistry. we have to identify the chiral carbon &…
Q: Draw the two chair conformations of menthol, and tell which is more stable
A: Menthol is the derivative of cyclohexanol. Chemical formula for menthol is C20H10O and IUPAC name…
Q: Compound P was discovered by a scientist. She discovered that P is a ripeptide that is optically…
A: Given data are P compound =C21 H25 N3 O5 Q compound = C21 H25 N3 NaO5 S compound =C21 H23 Cl2 N3 O3…
Q: Without referring to the chapter, draw the chair conformations of b-d-galactopyranose (the C4 epimer…
A: SOLUTION: Step 1: Gluco pyranose and galacto pyranose are C-4 epimers, which means they both differ…
Q: iso-butylchloride with CH3COONa
A: The structure of iso-butylchloride is as follows: This alky halide gives a nucleophilic…
Q: Give the IUPAC name of CH, CH₂CH₂CH CH₂ Include stereochemistry.
A:
Q: Suggest a reason why decane(CH3CH2CH2CH2CH2CH2CH2CH2CH2CH3) does not exhibitliquid crystalline…
A: Since to exhibit liquid crystalline behaviour the molecules should not tend to coil in any random…
Q: Draw the structure of cholic acid, showing the rings in their chairconformations, and label each…
A: The structure of Cholic acid is:
Q: Monosaccharide Structure and Stereochemistry For D-arabinose: Draw its enantiomer.
A: Given: A monosaccharide, D-arabinose.
Q: Explain "Gauche conformations are generally higher in energy than anti conformations because of…
A: The stability of any conformation depends upon their energies as higher is the energy ,lesser is…
Q: a) Which structures are Enantiomers? b) Which structures are Diastereomers? c) Which structures are…
A: Enantiomers are chiral molecules that are mirror images of one another and non-superimposable on one…
Q: Does lactic acid have enantiomers or stereoisomers? And explain why ...
A: Lactic acid have enantiomers because lactic acid is a chiral molecule. Lactic acid has one chiral…
Q: In addition to ß-D-glucopyranose (see Problem 4.67), glucose can exist in another cyclic form,…
A: Carbohydrates exist in multiple cyclic forms, one is a six-membered ring and the other is a…
Q: Locate with an asterisk the stereogenic centers (if any) in the following structure: C.…
A: The compound given is CH3CHBrCF3.
Q: Which of the attached cyclic molecules are meso compounds?
A: If a compound, despite the presence of two or more chiral centres in its molecule is achiral due to…
Q: Does methylcyclopentane have a stereogenic center?
A: Methyl cyclopentane has no stereogenic center, because no any carbon atom is connected to four…
Q: Select the group with the highest IUPAC priority.. (a) -CHCI2 -CH2B (c) -CH2CH2I
A: The priority of the groups depend on the atomic number of the elements present in it .
Q: clearly explain what type of isomerism is being shown and how it arises. i) Br2C=CH2 ii)…
A: positional isomerism, it is also called position isomerism - isomers have the same functional groups…
Q: what is the N factor of disodium ethylenediaminetetraacetate (C10H14N2Na2O8)?
A: N factor is defined as the number of H+ ions replaceable by 1 mole of acid in a reaction i.e.., in…
Q: Classify attached pair of compounds as constitutional isomers or stereoisomers ?
A: The given pair of compounds classified as constitutional or stereoisomers has to be given.
Q: B с D C A CEC-CH2CH3 H N-H
A: Bond angle is the angle between two bonds. It can be easily predicted if you know the hybridization…
Q: Determine which of these two compounds is more basic CH3CH2NH2 or CF3CH2NH2? Elaborate why.
A: Given compounds are : CH3CH2NH2 or CF3CH2NH2 Determine which of these two compounds are more basic…
Q: Without referring to the chapter, draw the chair conformations of(a) b-d-mannopyranose (the C2…
A: In chair conformation two planes present. Two possible positions for hydrogen is there axial and…
Q: Which of the following cyclic molecules are meso compounds?
A:
Q: How many constitutional isomers of acyclic monosaccharides are there that can be classified as…
A: Constitutional isomers are the compounds which have the same chemical formula but different…
Q: (b] CH3 CH3CHCH2CH2CІ {e) I CH2CH2CI CH3CHCHCH2CH3
A: Give the IUPAC name of the given structures-
Q: How many chiral centers are in B-d-glucopyranose and a-D-galactopyranose? How many stereoisomers of…
A: Beta -D-glucopyranose and alpha-D-glucopyranose are isomers. Chiral centers is carbon having four…
Q: a. Which structure(s) contain(s) sphingosine? b. Which structure(s) is a (are) glycolipid(s)? c.…
A: 1) Sphingosine (2-amino-4-trans-octadecene-1,3-diol) is an 18-carbon amino alcohol with an…
Q: The pK, of CH3CH;CH2OH is than that of CH3CH2CH2SH.
A: Here, we have to find whether the pKa of CH3CH2CH2OH is greater or lesser than that of CH3CH2CH2SH.…
Q: CI ч CH₂CH₂CH₂CH3 CH3 CH3
A:
Q: How many asymmetric carbons and stereoisomers are there for an aldohexose? For aketohexose?
A: Carbon atom that is connected to four different types of groups or atoms is known as asymmetric or…
Q: Vanillin (4-hydroxy-3-methoxybenzaldehyde), the principal component of vanilla, occurs in vanilla…
A: In the structure of beta D glucopyranoside, there is O linkage between glucose and other group…
Q: draw each of the chair configurations of myo inositol. predict which conformation will be more…
A: Axial position is highly less stable due to repulsion while equitorial position is more stable…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images


