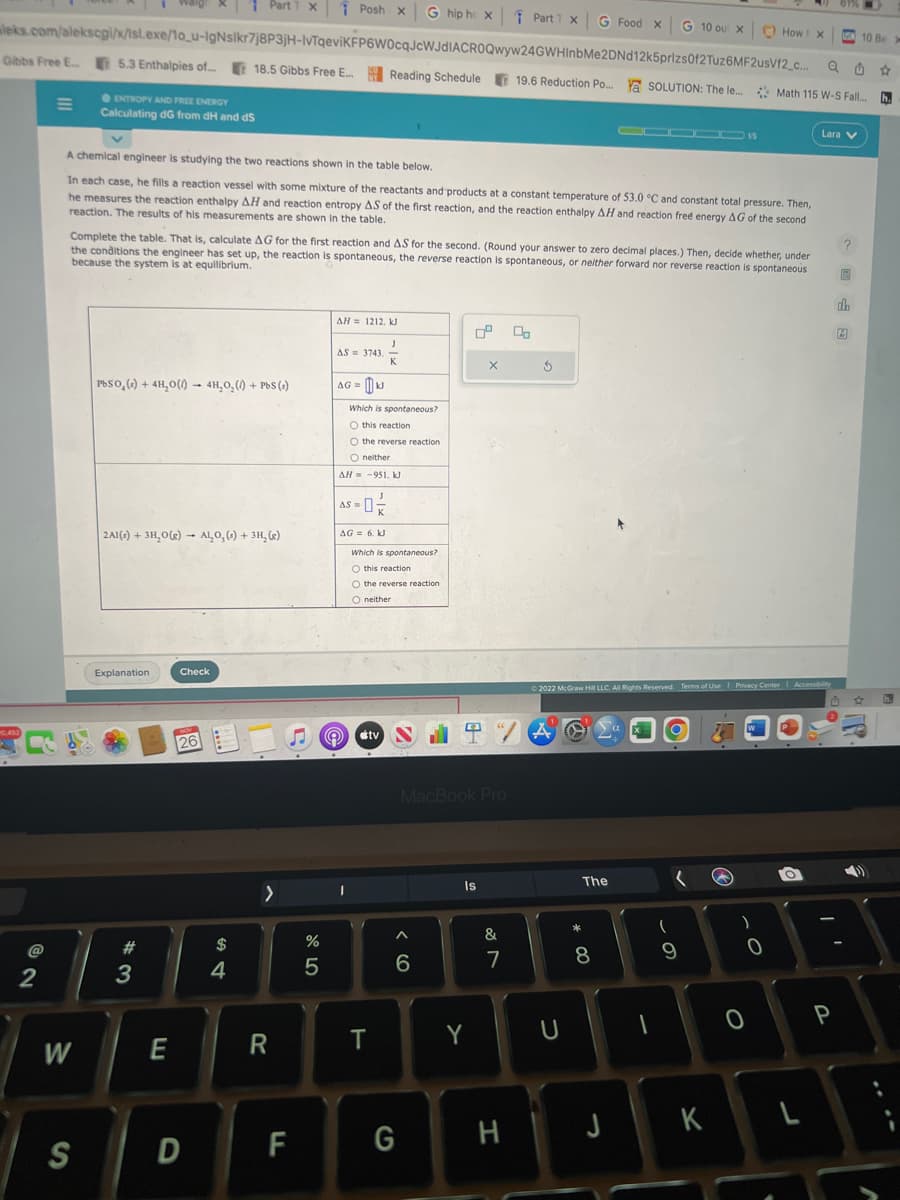
A chemical engineer is studying the two reactions shown in the table below. In each case, he fills a reaction vessel with some mixture of the reactants and products at a constant temperature of 53.0 °C and constant total pressure. Then, he measures the reaction enthalpy AH and reaction entropy AS of the first reaction, and the reaction enthalpy AH and reaction free energy AG of the second reaction. The results of his measurements are shown in the table. Complete the table. That is, calculate AG for the first reaction and AS for the second. (Round your answer to zero decimal places.) Then, decide whether, under the conditions the engineer has set up, the reaction is spontaneous, the reverse reaction is spontaneous, or neither forward nor reverse reaction is spontaneous because the system is at equilibrium. PbSO () + 4H₂O()- 4H₂O₂()+ PbS () 2Al(s) + 3H₂O(g) AL₂O, (s) + 3H₂(e) Explanation Check MIN 26 AH= 1212. kl AS = 3743. AG = UJ D)) Which is spontaneous? O this reaction O the reverse reaction O neither AH-951. kl AS=0/2 AG = 6. KJ J K Which is spontaneous? O this reaction O the reverse reaction O neither tv X Ś MacBook Pro Ⓒ2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center 1 Accessibilty TVA dh 2
A chemical engineer is studying the two reactions shown in the table below. In each case, he fills a reaction vessel with some mixture of the reactants and products at a constant temperature of 53.0 °C and constant total pressure. Then, he measures the reaction enthalpy AH and reaction entropy AS of the first reaction, and the reaction enthalpy AH and reaction free energy AG of the second reaction. The results of his measurements are shown in the table. Complete the table. That is, calculate AG for the first reaction and AS for the second. (Round your answer to zero decimal places.) Then, decide whether, under the conditions the engineer has set up, the reaction is spontaneous, the reverse reaction is spontaneous, or neither forward nor reverse reaction is spontaneous because the system is at equilibrium. PbSO () + 4H₂O()- 4H₂O₂()+ PbS () 2Al(s) + 3H₂O(g) AL₂O, (s) + 3H₂(e) Explanation Check MIN 26 AH= 1212. kl AS = 3743. AG = UJ D)) Which is spontaneous? O this reaction O the reverse reaction O neither AH-951. kl AS=0/2 AG = 6. KJ J K Which is spontaneous? O this reaction O the reverse reaction O neither tv X Ś MacBook Pro Ⓒ2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center 1 Accessibilty TVA dh 2
Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
Transcribed Image Text:Posh x G hip hc x
How x
leks.com/alekscgi/x/isl.exe/1o_u-IgNslkr7j8P3jH-lvTqeviKFP6W0cqJcWJdIACROQwyw24GWHInbMe2DNd12k5prizs0f2Tuz6MF2usVf2_c... Q
0
18.5 Gibbs Free E... Reading Schedule 19.6 Reduction Po.... Ya SOLUTION: The le... Math 115 W-S Fall....
h.
Gibbs Free E...
0,452
E
CO
2
5.3 Enthalpies of...
W
●ENTROPY AND FREE ENERGY
Calculating dG from dH and dS
S
A chemical engineer is studying the two reactions shown in the table below.
In each case, he fills a reaction vessel with some mixture of the reactants and products at a constant temperature of 53.0 °C and constant total pressure. Then,
he measures the reaction enthalpy AH and reaction entropy AS of the first reaction, and the reaction enthalpy AH and reaction free energy AG of the second
reaction. The results of his measurements are shown in the table.
Complete the table. That is, calculate AG for the first reaction and AS for the second. (Round your answer to zero decimal places.) Then, decide whether, under
the conditions the engineer has set up, the reaction is spontaneous, the reverse reaction is spontaneous, or neither forward nor reverse reaction is spontaneous
because the system is at equilibrium.
PbSO, () + 4H₂O()- 4H₂O₂() + PbS (s)
2Al(s) + 3H₂O(g) Al₂O, (s) + 3H₂(g)
Explanation
#3
E
Check
HON
126
D
Part TX
$
At
4
>
R
FL
%
5
AH = 1212, kJ
AS 3743.
AG=kJ
Which is spontaneous?
O this reaction
O the reverse reaction
O neither
AH-951. KJ
|AS = 0²/2
AG = 6. KJ
I
J
K
Which is spontaneous?
O this reaction
O the reverse reaction
Oneither
#tv
T
BH
A
6
G
MacBook Pro
X
Is
Y
TA
&
Part 1 x G Food X
7
Do
H
Ś
U
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
0
The
* 00
G 10 ou X
▬▬▬OD 15
8
J
(
9
K
)
0
0
.
Lara V
61%
P
?
db
10 Be
h.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

