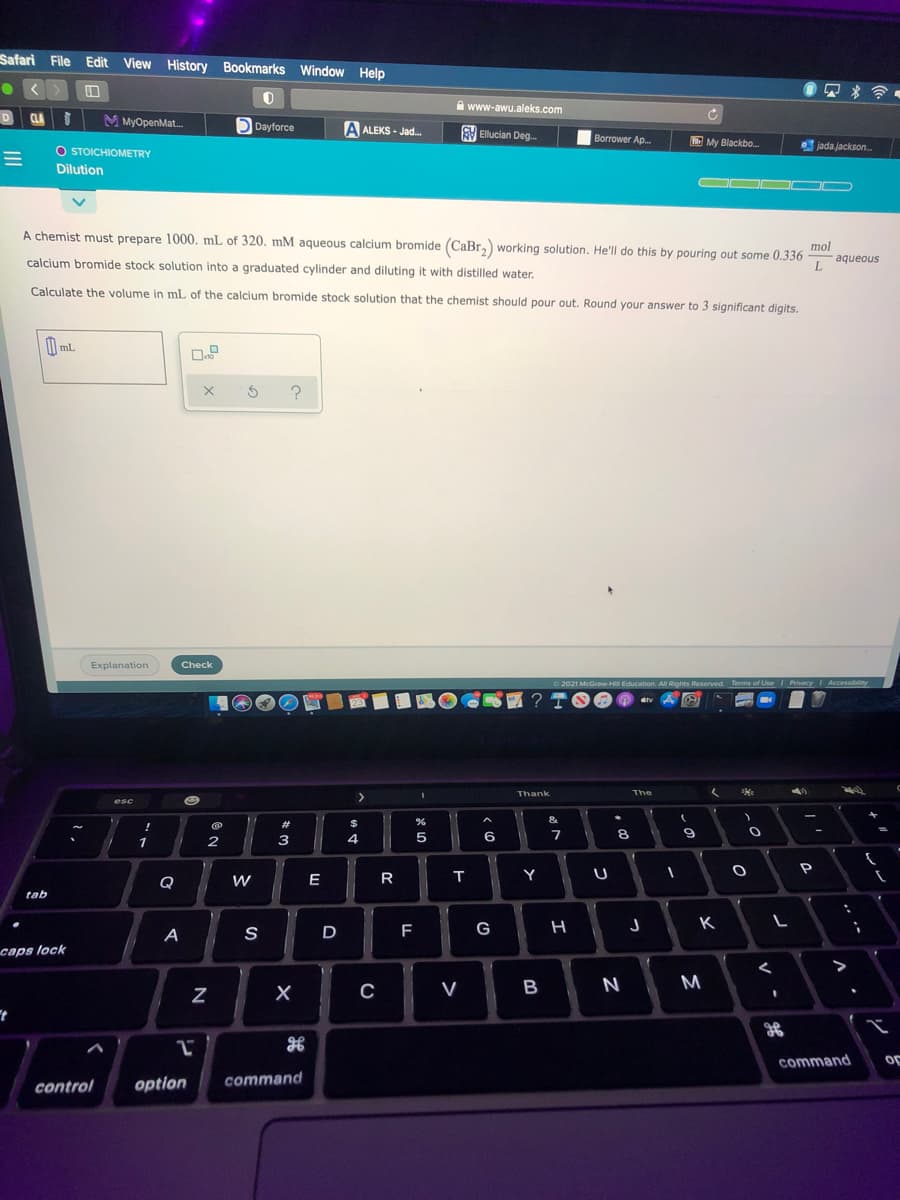
A chemist must prepare 1000. mL of 320. mM aqueous calcium bromide (CaBr,) working solution. He'll do this by pouring out some 0.336 mol calcium bromide stock solution into a graduated cylinder and diluting it with distilled water. aqueous L. Calculate the volume in mL of the calcium bromide stock solution that the chemist should pour out. Round your answer to 3 significant digits.
A chemist must prepare 1000. mL of 320. mM aqueous calcium bromide (CaBr,) working solution. He'll do this by pouring out some 0.336 mol calcium bromide stock solution into a graduated cylinder and diluting it with distilled water. aqueous L. Calculate the volume in mL of the calcium bromide stock solution that the chemist should pour out. Round your answer to 3 significant digits.
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 1P
Related questions
Question
Transcribed Image Text:Safari File Edit
View History Bookmarks Window Help
A www-awu.aleks.com
CLA T
M MyOpenMat.
ODayforce
A ALEKS - Jad.
SV Ellucian Deg..
Borrower Ap.
My Blackbo..
D jada.jackson.
O STOICHIOMETRY
Dilution
OD
A chemist must prepare 1000. mL of 320. mM aqueous calcium bromide (CaBr,) working solution. He'll do this by pouring out some 0.336
mol
calcium bromide stock solution into a graduated cylinder and diluting it with distilled water.
aqueous
Calculate the volume in mL of the calcium bromide stock solution that the chemist should pour out. Round your answer to 3 significant digits.
mL
Explanation
Check
2021 McGraw-H Education. AllRights Reserved. Terms of Use / Privacy Accessibility
dtv
Thank
The
esc
&
2#
-
1
2
3
4.
6
Q
W
E
T
Y
tab
D
F
G
K
A
S
caps lock
M
V
command
or
control
option
command
R
N
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning



Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning
