Q: The change in entropy will be positive for which of the following reactions? a.2NO2(g) → N204(g) b.…
A: Entropy is a measure of disorder or randomness in the system. Gas has more entropy than solid and…
Q: Label each pair of compounds below as: a. conformational isomers b. stereoisomers C. constitutional…
A: We have to determine the relation between the given structures
Q: Through titration with a known standard, we counted that an unknown solution contains 0.048 moles of…
A: Given, Moles of acetic acid = 0.048 mol. The original volume of acetic acid = 120.0 mL. The…
Q: Determine the initial pH of a 15.5mL HCl solution that was titrated with 16.9mL of 0.075mol/L NaOH…
A: Sodium hydroxide reacts with hydrochloric acid, to form sodium chloride and water. The equation for…
Q: Consider the following chemical reaction: Al2O3 (s) + 6HCl(aq) ---------->2AlCl3(aq) + 3H2O(l)…
A:
Q: List of Acids HF CH3COOH H2SO4 HI HCl H2S H2CO3 1. Which acid/s will produce an ion with a single…
A: Given following acids - HF, CH3COOH, H2SO4, HI, HCl, H2S, H2CO3 Here HF, CH3COOH, H2S and H2CO3…
Q: What is the most likely product from the following sequence
A: The first step of the reaction is the Friedel-Kraft acylation reaction and the second step of the…
Q: Question # 3: Draw or name the following organic compounds. You may draw complete structural or line…
A:
Q: C OD. O B. OC. CH3 A Br CH₂ B AC... CH3 CH₂ Br H3C Br C CH₂ معاكم
A: For the most stable conformation, bulky group (Br) should be placed at equitorial position and…
Q: Indicate whether the pair of structures shown represent stereoisomers, constitutional isomers,…
A: Relationship between the following molecule.
Q: Draw the chair flip of the following conformation. Select to Draw Chair Flip Q I 1 I I
A: In this question, we will draw the chair flip conformation for cyclohexane ring. You can see the…
Q: How is this reaction classified? R DG & Reaction progress Free energy O a Endergonic O b.…
A: the ∆Grxno of this reaction is negative which we can see in this graph.
Q: A particular water sample that is saturated in CaF2 has a Ca²+ content of 105 ppm (that is, 105 g…
A:
Q: Review items 1 to 6 and answer questions a and b: The activities were designed to produce an EX4…
A:
Q: Which structure has a formal charge of -1 on the carbon shown with a "C"? Note that the atom of…
A:
Q: How many π (pi) bonds in the structure shown below? 01 05 04 03 2
A:
Q: An argon gas occupies 56.2 L at 760 torr. If the volume of gas is decreased to 1/4 of the original…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Burger Barn makes a dipping sauce by mixing 4 spoonfuls of honey with 1 spoonful of mustard.…
A: The constituents of the dipping sauce are mustard and honey. Since mustard is present in smaller…
Q: A solution that is 20.0% ethanol by volume has a density of 0.977 g/mL. What is the mass percent of…
A:
Q: Assign R, S configurations to each indicated chirality center in the molecules below. OH B. HO COOH…
A:
Q: A potential strategy for the synthesis of livermorium-293 Z = 116 involves bombarding hassium-277 Z…
A: A nuclear reaction can be balanced by the summation of mass number and atomic number. The atomic…
Q: A certain reaction has the following reaction Constant values at various temperatures k, min-¹ Ï°C…
A:
Q: 1. Draw the curved arrow mechanism for the given reaction and write the product. CF₂CO₂H
A:
Q: 3 Cu + 8HNO3 -->3 Cu(NO3)2 + 2 NO + 4 H₂O In the above equation how many moles of water can be made…
A:
Q: When an orbital is specified as 3d, what quantum numbers correspond to the orbital?
A:
Q: GUIDE QUESTIONS: 1. What are other oils, besides coconut, that are used to make bath soaps? 2. Based…
A: (1) There are some other oils besides coconut oil - * Sun flower oil * Sweet almond oil * Olive oil…
Q: Choose the reagent(s) that would be most likely to complete this reaction.
A: Given; reactant and product
Q: Fill-up in the blanks: Common Name 1. N-tert. pentyl- valeramide 2. (5) IUPAC Name (1)…
A: 4) amide 8) anhydride
Q: 2. Sketch (as accurately as you can) steady state i vs E curves for solutions prepared from: a) 1 mM…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Four samples of radionuclides listed in the table below. Initially, each sample contains 16.00 µmol…
A:
Q: For each reaction listed, determine its standard cell potential at 25 °C and whether the reaction is…
A: The standard cell potential of a cell can be determined by the oxidation and reduction potential of…
Q: Chemistry What is the molarity of a solution formed when 37.79 g of copper II nitrate is dissolvedin…
A: Given that - Mass of Copper II nitrate = 37.79 g Volume of water used = 170 mL Then, from…
Q: Of the following compounds, rank each series by nucleophilicity using 1 for the most nucleophilic…
A: Nucleophilicity is nothing but the tendancy of attack over the electron difficient species . The…
Q: Draw a correct Lewis dot structure for the following: (The way the formula is typed MIGHT give you a…
A: Lewis dot structure is the two dimensional representation of valence electrons between two…
Q: 15 ice cubes at 0 °C, each containing 1.0 mole of H₂O, are used to cool tea. How much energy do the…
A: given, number of ice cubes = 15 1 ice cube = 1.0 mole water
Q: Provide the IUPAC name for the following. You may use either the 'prefix-2-alkene' or the…
A: We have to name the given structures
Q: (R) (S) ОНС XIII
A: R/S configuration is given by CIP rule. Initially the priority is assigned to all the four groups…
Q: Ethylene glycol (EG), CH₂(OH)CH₂(OH), is a common automobile antifreeze. It is water soluble and…
A:
Q: In a terrible accident, a solution consisting of 2.37 kg of nitric acid, HNO3, was spilled on the…
A: Given: The mass of HNO3 that was spilled = 2.37 kg The mass of Na2CO3 added = 2.00 kg We have to…
Q: What is the mole fraction of acetone (C3H6O. MW-58.08 g/mol) in the gas phase resulting from a…
A: In this question, we will determine the mole fraction of acetone in gas phase from the liquid phase…
Q: Identify the strong acid in the list below: - HCl - HBr - H2SO4 - All of these
A: • The given list of acida are - HCl , HBr , H2SO4 • We need to identify the strong acid from the…
Q: 3/variants/434513/take/4/ MATCHING LIST Question 3 The oxidation number of nitrogen for three…
A: Oxidation number: • Oxidation number of atoms in pure element or Atoms in free or uncombined…
Q: Draw all the Newman projection of (2R, 3S)-dibromobutane sighting down the C2-C3 bond. Identify the…
A: All the conformers are shown below.
Q: The standard reaction free energy AG = -910. kJ for this reaction: 6 C(s) + 6H₂(g) + 3O₂(g)…
A:
Q: What is the product of this reaction? (A) (C) O .H H O & (B) (D) 1. LDA, -78, °C O 2. 3. H3O+ O H
A: Organic reactions are those in which organic reactant react to form organic products. LDA is lithium…
Q: 1. Fill in the blanks with the most appropriate term: A are the tells the story of a chemical…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 3. b) Give the common names of the above structures and indicate whether they contain an aldehyde of…
A: If RCHO group is there then it is aldehyde and if RCOR group is there then it is ketone. For more…
Q: Calculate the max using Woodward-Fieser Rules te
A: The woodworld fisher rule is used to calculate the Lambda maximum value of the given compound.
Q: If the given reaction occurs spontaneously, which of the following is the balanced reduction…
A:
Q: In a displacement reaction, we a cation (either H" or a metal cation) with an elemental metal…
A: Metathesis reactions is another name for the double-displacement reaction. The displacement reaction…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

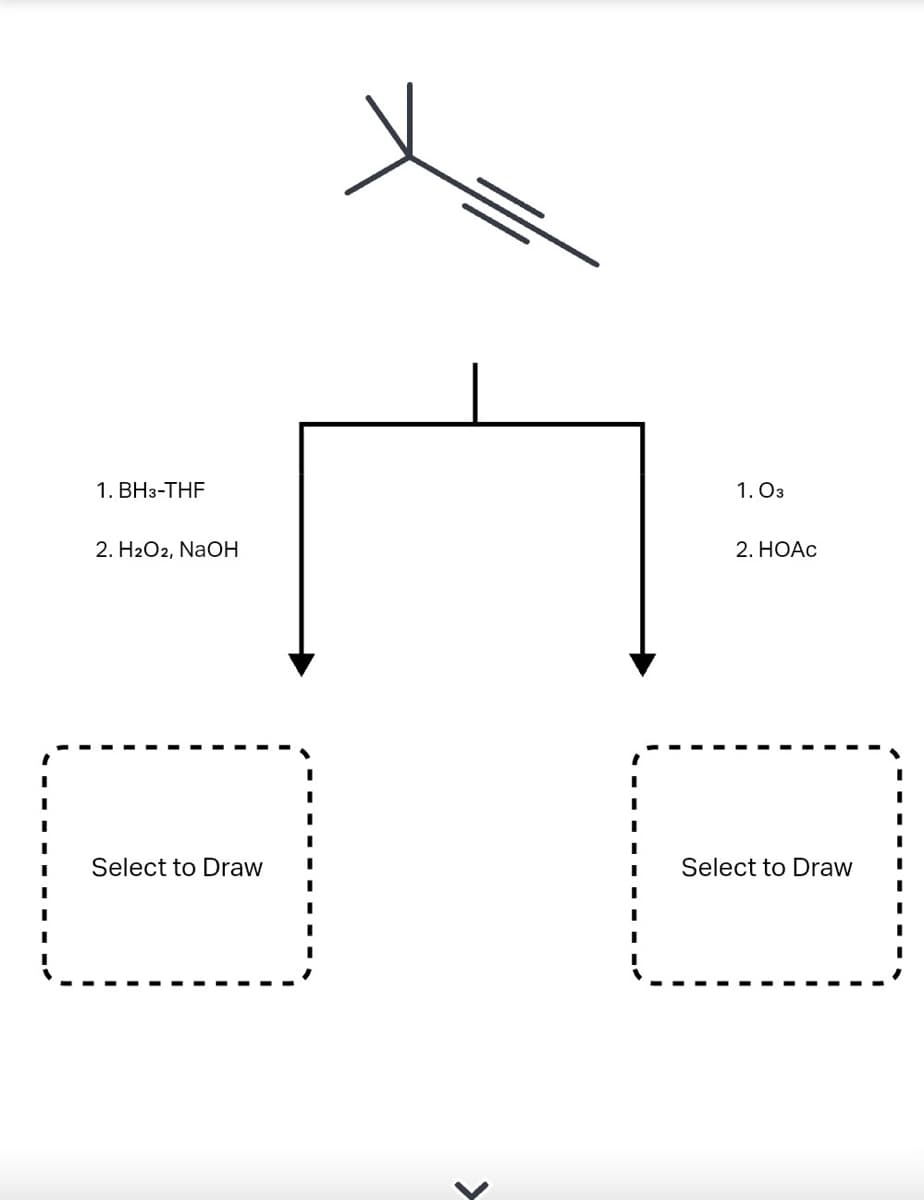
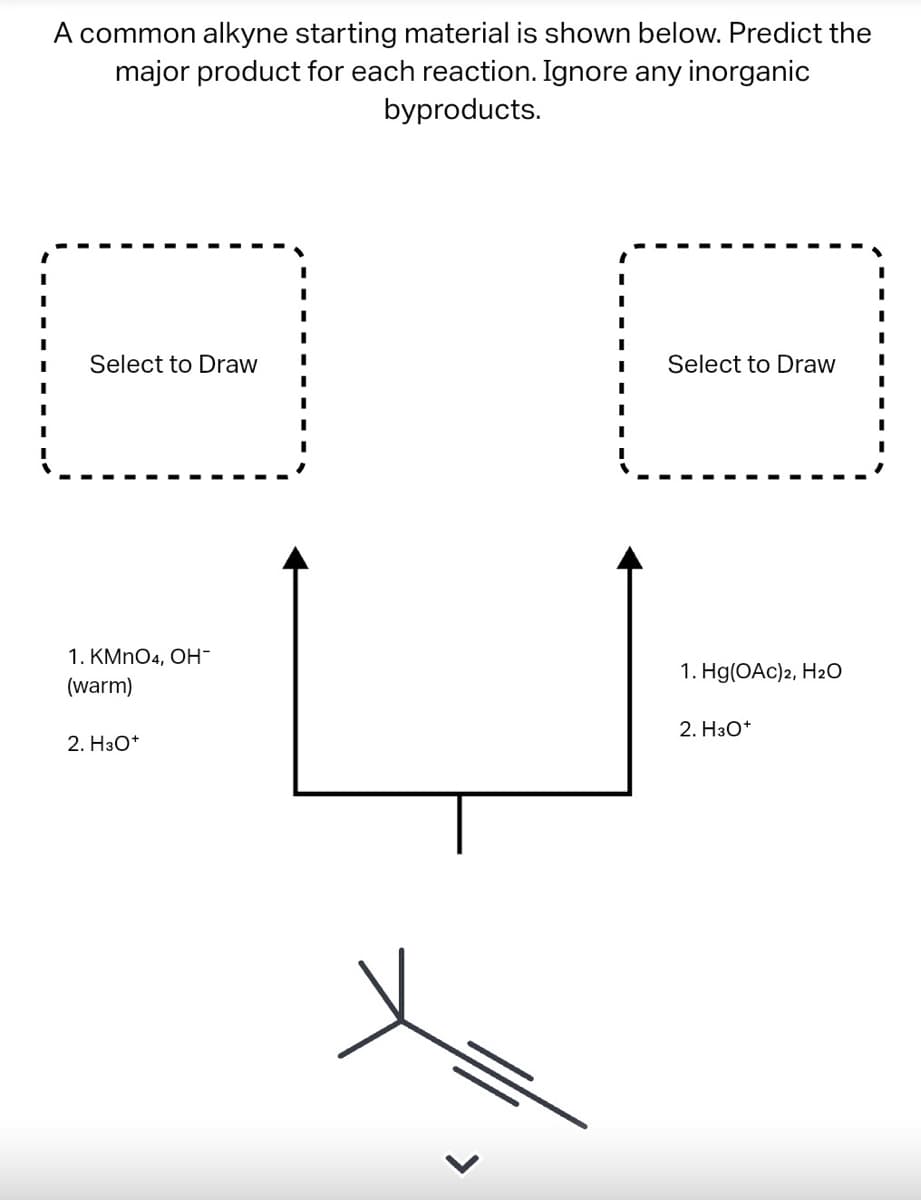
- A common alkyne starting material is shown below. Predict the major product for each reaction. Ignore any inorganic byproducts.. The question is asking for 4 drawings. thanksA common alkene starting material is shown below. Predict the major product for each reaction. Ignore any inorganic byproducts.(a) Analyze SN1 or SN2 is preferred in the reaction below. Explain your choice and express themechanisms.
- What is the structure/s of the major organic product/s for the reaction below?How would you synthesize the followingcompounds from benzene using thereagents from the table? Each requires 3-4steps.A common alkyne starting material is shown below. Predict the major product for each reaction. Ignore any inorganic byproducts.
- Following is a balanced equation for bromination of toluene.(a) Using the values for bond dissociation enthalpies given in Appendix 3,calculate ∆H0for this reaction.(b) Propose a pair of chain propagation steps and show that they add up to theobserved reaction.(c) Calculate ∆H0for each chain propagation step.(d) Which propagation step is rate-determininA. propose the efficient synthesis and the mechanism B. give the products for each step and the final productIn SN2 reactions of haloalkanes, the order of reactivity is RI>RBr>RCl>RF. Alkyl iodides are considerably more reactive than alkyl fluorides, often by factors as great as 106. All 1-halo-2,4-dinitrobenzenes, however, react at approximately the same rate in nucleophilic aromatic substitutions. Account for this difference in relative reactivities.
- Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Compounds A, B, and C have the same molecular formula C4H8. They all react wotu H2/PtO2 to give the same compound. The reaction of A or B with H2O/H2SO4 or with BH3-THF, followed by treatment with a basic solution of hydrogen peroxide, gives the same compound, namely D. The reaction of C with H2O/H2SO4 also gives D. However, the reaction of C with BH3-THF, followed by HO-, H2O2 gives a new compound, E. Provide the identity of A, B, C, D, and E along with explanations of reactivity.Carbocations often rearrange, as shown below. Draw in the hydrogens on the two carbons involved in the rearrangement, and show formal arrow-pushing to illustrate the transformation.

