Q: Draw the major organic product you would expect to isolate from the reaction of 3-ethyl-5-methyl-4-…
A: BH3 is complexed with Tetrahydrofuran(THF) to avoid the dimerization of the borane molecule. This…
Q: a) Outline a synthetic pathway to produce N,N-dimethylaniline from benzoic acid. b) Arenediazonium…
A: Both the final products can be prepared from the respective starting materials via a large no. of…
Q: Provide the full mechanism for nitration of benzene. Show how the electrophile is formed and how the…
A:
Q: Which of the following ethers can be formed in good yield by condensation of thecorresponding…
A: The structure of dibutyl ether (1-butoxy butane) is: It is symmetrical ether.
Q: 1.
A:
Q: Outline a feasible synthetic route for the following:
A: Step-1: Free radical substitution reaction Step-2: Nucleophilic substitution reaction
Q: The analgesic acetaminophen is synthesized by treating 4-aminophenol with one equivalent of acetic…
A: Given 4-aminophenol + acetic anhydride ----> acetaminophen + additional product
Q: Draw a sketch of the structure of the transitionstate,
A: SN2 reaction takes place in single step and in this reaction transition state is formed before…
Q: Examine the sequence of reactions shown below and provide structures (including stereochemistry…
A: The given reaction sequence is represented as follows:
Q: Give detailed answer- Provide example of compound that fulfil each criteria below. Explain why-…
A: Dear student since it is a multiple subparts questions, according to bartleby guidlines here i am…
Q: Solvolysis of 2-bromo-2-methylbutane in acetic acid containing sodium acetate gives three organic…
A: When 2-bromo-2-methylbutane undergoes solvolysis in the presence of acidic acid and sodium acetate…
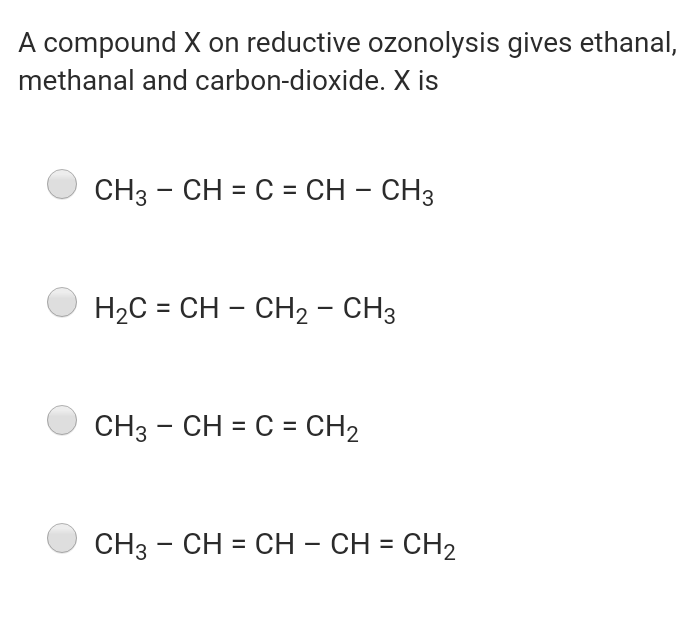
Q: Among the following compounds which compound(s) can involve in Cannizzaro reaction (including…
A:
Q: product of the reaction between benzoyl chloride and excess ammonium hydroxide is most easily…
A: The reactants given are benzoyl chloride and excess ammonium hydroxide.
Q: What is /are organic product(s) expected when 1-butyne is first treated with sodium amide and the…
A: In this reaction, we will start with a terminal alkyne. The first step includes the generation of a…
Q: Draw structural formulas for the product of nitration of compound. Where you predict ortho-para…
A: If the substituents on benzene carbon are electron rich groups they are known as activating groups.…
Q: Doxaprost, an orally active bronchodilator patterned after the natural prostaglandins , is…
A: Given:- Doxaprost, an orally active bronchodilator patterned after the natural…
Q: Briefly explain why treatment of alcohols with hydrogen halides to produce alkyl halides s generally…
A: The alcohols react with hydrogen halide to produce the corresponding haloalkanes. The reaction…
Q: Provide a strategy to control the production of 3-nitroquinoline, from quinoline (with mechanism).
A:
Q: Discuss the fatal flaw in the following reaction and provide a rational solution to alleviate he…
A: Friedel-Craft alkylation reaction:When the benzene reacts with an alkyl halide in presence of Lewis…
Q: Which of the molecules A through D shown below will give mixtures of enantiomers upon reaction with…
A: Answer: 1 (C) and (D) Explanation: Non superimposable mirror images are called enantiomers. If…
Q: What would be the ratio of products in the reaction of chlorine with ethylbenzene ifchlorine…
A: The reaction of chlorine with ethylbenzene if chlorine randomly abstracted a methyl or methylene…
Q: How common electron-donating and electron-withdrawinggroups affect both the rate of reaction of a…
A: The substitution of an atom or group of atom through an electrophile is termed as electrophilic…
Q: Outline a stepwise mechanism of the following reaction: Br NHCH3 CH;NH2 NO2 ÑO2
A: Given Reaction of P-nitro Bromobenzene with Methyl amine Mechanism = ?
Q: The synthesis of one of the rings in remdesivir is accomplished using the reaction below. A. Using…
A: Electrophiles are those species which have deficiency of electrons. It attacks on the those species…
Q: Define the Mechanism - Conversion of Acid Chlorides to CarboxylicAcids ?
A: Acid chlorides: Also known as Acyl chloride is one of the derivatives of acid in which OH group is…
Q: most of the additions of bromine to double bonds gave entirely antistereochemistry. Explain why the…
A: Additions of bromine to double bonds: In most cases, the additions of bromine to double bonds gave…
Q: 1. Discuss a disadvantage of Friedel - Crafts acylation reaction 2. Propose a mechanism for the…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Explain and illustrate, why it is difficult to perfom Friedel-Craft reactions on the unactivated…
A: Friedel Craft reactions are the reactions which attaches the substituents to an aromatic ring. It…
Q: The reactions given in the synthesis scheme showing the synthesis of D, a phremone of the moth…
A:
Q: Give Four main precautions need to be considered when carrying out the following. b) Synthesis of…
A: Benzopinacole (white color precipitate) generation is done via photoreduction method. It is highly…
Q: What does Meso-dibromobutante on debromination gives?
A: On the basis of mechanism, there are different types of chemical reactions such as addition…
Q: Give the main organic product for the following reaction:
A: Carbonyl compound (aldehyde and ketone) reacts with an alcohol to form hemiacetal in the presence of…
Q: Give the major organic product for each of the reactions
A: To solve this problem we have to give the major products formed by the given reaction .
Q: Show the synthesis of butylamine from ammonia in one step. Make sure to specify the stiochiometry of…
A: Alkyl halides undergo substitution reaction with ammonia to give ammonium salts because ammonia act…
Q: Provide the structure of the major organic product of reaction 1 and the major organic product after…
A: Concept introduction; When alcohol treated with thionyl chloride in presence of base OH group is…
Q: AICI3
A: It is an example of Friedel craft acylation reaction, Here a new cyclic ketone formed as the major…
Q: Give the structures of A and B in the following reactions. Pay special attention to stereochemistry.…
A: m-CPBA (meta chloro per benzoic acid)--->> It is used for epoxidation of alkene and also used…
Q: Provide the major organic product of the reaction below and a detailed, stepwise mechanism which…
A: In the presence of a weak base, CH3OH, and heat, the reaction undergoes an E1-mechanism. In the case…
Q: The major product/s that form/s during the nitration of benzenesulfonic acid is? Choose the correct…
A: Nitration of sulphonic acid takes place as per following concepts.
Q: b) Provide a stepwise mechanism for the bromination of nitrobenzene.
A: -NO2 is a electron withdrawing group,it pull electron from ortho and para position by resonance and…
Q: Give
A: In the presence of peroxide HBr adds Br on more number of hydrogen contains carbon. While in absence…
Q: 6. Outline the synthesis route for the product. Any appropriate reagents such as carboxylic acid or…
A: Note : The groups on benzene ring direct the electrophile to ortho, para or meta position. Step 1:…
Q: Which of the following reacts with piperidine to give the compound shown below? piperidine
A: Interpretation: By using piperidine, we have to check which among the following from the given…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- most of the additions of bromine to double bonds gave entirely antistereochemistry. Explain why the addition to phenanthrene gives a mixture of synand anti stereochemistry. When the product from (c) is heated, HBr is evolved and 9-bromophenanthrene results.Propose a mechanism for this dehydrohalogenation.For the dehydrohalogenation (E2) reaction shown, draw the Zaitsev product(s) resulting from elimination involving C2–C3 (i.e., the carbon atoms depicted with stereobonds). Show the product stereochemistry clearly. If there is more than one organic product, both products may be drawn in the same box. Ignore elimination involving C2 or C3 and any carbon atom other than C3 or C2.True or false? 2-bromo-2-methylpropane undergoes SN1 rather than SN2 because the nucleophile experiences steric hindrance, and a stable carbocation can be formed. When hydroxide ion reacts with 1-chloropropane the main product is 1-propanol.
- Explain how the reaction of (CH3)2CHCH(Cl)CH3 with H2O yields two substitutionproducts, (CH3)2CHCH(OH)CH3 and (CH3)2C(OH)CH2CH3The reaction of 2-ethyl-1-pentene with Br2, with H2 + Pd/C, or with R2BH/THF followed by aqueous HO- + H2O2 leads to a racemic mixture. Explain why a racemic mixture is obtained in each case.What product(s) would you expect to obtain from reaction of 1, 3-cyclohexadiene witheach of the following?(a) 1 mol Br2in CH2Cl2(b) O3followed by Zn(c) 1 mol HCl in ether(d) 1 mol DCl in ether(e) 3-Buten-2-one (H2C = CHCOCH3)(f) Excess OsO4, followed by NaHSO3
- Which of the following reagent best accomplish this transformation below? a. BH3, THF, H2O2 b. NaNH2, NH3 c. H2SO4, H2O, HgSO4 d. H2, LindlarProvide the product most likely to form from reaction of each reagent with 1-methylcyclohexene. You may ingore stereochemistry for this question. Br2 H-Br H-Br, PhC(O)OOC(O)Ph N-bromosuccinimide, PhC(O)OOC(O)Ph A. B. C. D.Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answer
- Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the helpCleavage of oct-1-ene with hot, KMnO4 will yield _____________. octanol and carbon dioxide octanone hexanol and 2 carbon dioxides heptanal and formaldehyde heptanoic acid and carbon dioxideDraw the major organic product for the reaction of 1-phenylpropan-1-one with sodium hydride followed by ethyl iodide. Ignore stereochemistry.

