a) Discuss hybridization of carbon in ethyne, and its orbital diagram and bond formation. 1. b) Draw line bond structure of two compounds with the molecular formula, C2H&O. Comment on their ability to form hydrogen bond. c) Assign formal charges to the atoms in each of the following molecules: „H CH, CH;- N-CH, CH, CH,C=C: CH,C=N-H CH,CH, (i) (ii) (ii) (iv)
a) Discuss hybridization of carbon in ethyne, and its orbital diagram and bond formation. 1. b) Draw line bond structure of two compounds with the molecular formula, C2H&O. Comment on their ability to form hydrogen bond. c) Assign formal charges to the atoms in each of the following molecules: „H CH, CH;- N-CH, CH, CH,C=C: CH,C=N-H CH,CH, (i) (ii) (ii) (iv)
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter4: Molecular Structure And Orbitals
Section: Chapter Questions
Problem 56E: Many important compounds in the chemical industry are derivatives of ethylene (C2H4). two of them...
Related questions
Question
6
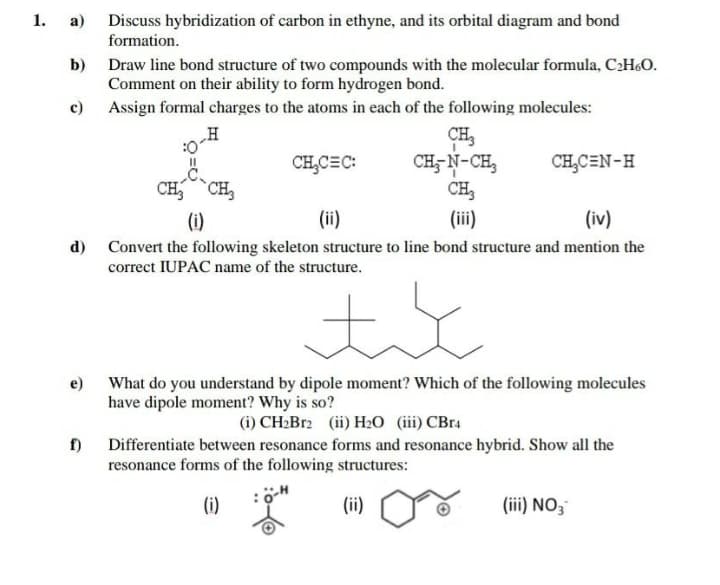
Transcribed Image Text:1. а)
Discuss hybridization of carbon in ethyne, and its orbital diagram and bond
formation.
Draw line bond structure of two compounds with the molecular formula, CH&O.
Comment on their ability to form hydrogen bond.
c) Assign formal charges to the atoms in each of the following molecules:
b)
„H
CH,
CH- N-CH,
CH,
CH,C=C:
CH,CEN-H
CH,
CH,
(i)
(ii)
(ii)
(iv)
d) Convert the following skeleton structure to line bond structure and mention the
correct IUPAC name of the structure.
e) What do you understand by dipole moment? Which of the following molecules
have dipole moment? Why is so?
(i) CH2B12 (ii) H2O (iii) CBr4
f)
Differentiate between resonance forms and resonance hybrid. Show all the
resonance forms of the following structures:
(i)
(ii)
(iii) NO3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 7 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning