Q: [Review Topics] [References] Be sure to specify states such as (aq) or (s). If a box is not needed…
A:
Q: Show how you might synthesize this compound from an alkyl bromide and a nucleophile in an SN2…
A: In SN2 reaction nucleophile attacks to substrate from back side results into inversion of product
Q: hello i am having difficulty with part a, b, and c A galvanic cell based on the above reaction is…
A: Galvanic cell
Q: Please help what are special rules or laws to predict predominant products for alcohols or et
A: Here's a chart summarizing some special rules or laws to predict predominant products for alcohols…
Q: What is the most likely product resulting from the reaction shown? Н 0= OH НО OH NaBH4 ? од
A: NaBH4 is a good reducing agent. It can reduce aldehydes , ketones , acid chlorides and anhydrides .…
Q: Modify the given copy of the starting material to draw the product. H₂C. Br 1) CH3CI, AICI 2) KMnO4,…
A: The Friedel-Craft alkylation reaction is an electrophilic substitution reaction. Benzene ring on…
Q: draw the structures of the geometric and optical isomers of the compound [Co(gly)3
A: We have been given an inorganic compound [Co(gly)3].We have been asked to draw the structures of…
Q: The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: R E = In…
A:
Q: In a study of the decomposition of ammonia on a tungsten surface at 1100 °C NH3 1/2 N₂ + 3/2 H₂ the…
A:
Q: 13. Please check to see if the following are balanced. If they are not, please balance them. a. K b.…
A: Q 13 balancing reaction will be as follows: a) 2K + Br2 → 2 KBr…
Q: 2. A student performed the experiment as described, using 5.00 mL of an aqueous 3.00% H2O2 solution,…
A: The basic chemical concept like mole concept, mass percentage, ideal gas law etc. are necessary to…
Q: To obtain the solute for 571 g of a 14.1% (w/w) solution of sugar we would weigh out ________ g of…
A:
Q: QUESTION 15 Using the following thermochemical equation, determine the amount of heat produced per…
A: Answer:- This question is answered by using the given balanced chemical equation the stoichiometry…
Q: 4. The following reaction results in the formation of not one, not two, but six(!) different…
A:
Q: QUESTION 12 Use the standard reaction enthalpies given below to determine AH°rxn for the following…
A: The standard enthalpy of any reaction changes with the coefficient of that reaction. If the…
Q: In the mechanism of the following acid catalyzed hydration transformation, there are distinct…
A: Given that, a reaction scheme is shown below We have to give the number of intermediates and the…
Q: The equivalence point in the titration of this weak acid is 50.80 mL. At 25.40 mL, the pH was…
A:
Q: Match each compound with the type of electrolyte it is. Drag and drop options on the right-hand side…
A: The electrolytes given are HNO3, NaCl, sugar, acetic acid, Na2CO3 and CaCO3.
Q: For the reaction below, Kc = 4.60 × 10⁻⁶. Note Kc is sometimes called K. What is the equilibrium…
A: Chemical equilibrium is defined as the stage at which both reactant and product are in equilibrium.…
Q: 234 KJ -150. KJ -117 kJ QUESTION 12 Use the standard reaction enthalpies given below to determine.…
A:
Q: The answer is 6.1 x 10^-2 but I'm not sure how to solve this problem. Please include every step.…
A: For any reaction: aA+bB→cC+dD The rate law will be: rate=kAxBy Here x and y are the orders with…
Q: How many and a bonds are in this molecule? 0 Ö-H H H-C H C=C number of a bonds: C-H H 9 Incorrect…
A: Given molecule :
Q: In biological systems, a FACILITATED transport process is a form of _______ transport, where an…
A: Transport of molecules in which no energy is required for the movement is called passive transport…
Q: Can you give 3 typical job functions of an analytical chemist?
A: An analytical chemist is a professional who specializes in the identification, separation, and…
Q: Suppose 0.753 g of sodium chloride is dissolved in 100. mL of a 57.0 m M aqueous solution of…
A:
Q: Balance the following redox reaction occuring in an acidic solution:
A: We have to balance the given redox reaction
Q: Which of the following substances is/are never in a Bronsted-Lowry acid in an aqueous solution? KOH…
A: A Bronsted-Lowry acid is a substance which donates a proton or H+ ion to the other compound
Q: 5. Provide a step-by-step reaction mechanism for the following transformation. You must show all…
A: Ketals are prepared by reacting a ketone with alcohol in the presence of an acid catalyst. In this…
Q: 1) Consider the following: H₂S (aq) + CO3² (aq) HCO3(aq) + HS (aq) a. Identify the acid, base,…
A: An acid is a species which donates Proton while a base accepts proton. an acid after donating…
Q: 10. Draw a graph of the concentration of NaCl (x-axis) against the calculated density (y-axis) for…
A: A question based on properties of liquids. A set of data for variation of density against %…
Q: 10. If 223.0 mg of AgBr is completely dissolved in 184.0 mL of 0.14 M NH3, what is the equilibrium…
A: Given, The mass of the AgBr = 223.0 mg The concentration of ammonia (NH3) = 0.14 M The volume of the…
Q: Calculate the change in pH when 7.00 mL of 0.100 M HCl(aq) is added to 100.0 mL of a buffer solution…
A: To solve this problem, we need to first determine the initial pH of the buffer solution. The buffer…
Q: t takes 614. kJ/mol to break a carbon-carbon double bond. Calculate the maximum wavelength of light…
A: Answer: Relation between wavelength of light and energy of photon is shown below: E=hCλ Here:…
Q: Gaseous ethane (CH3 CH3) will react with gaseous oxygen (O₂) to produce gaseous carbon dioxide (CO₂)…
A:
Q: Mary is trying to determine if some household substances are acids or bases. She collects the…
A:
Q: Design a reasonable synthesis for the transformation shown Br
A: Three and four-membered cyclic rings usually undergo ring expansion through bond migration due to…
Q: Be sure to answer all parts. From among the 18 constitutional isomers of C8H18, write structural…
A: Definition of isomers~ The molecules having same molecular formula but different structural…
Q: Above: FTIR of L-menthol. 3500 3000 2500 Wavenumber cm-1 2000 1500 1000 500 3246.67 2953.97 2925.63…
A: IR spectra provides the information of functional groups of unknown compound based on the…
Q: Be sure to specify states such as (aq) or (s). If a box is not needed leave it blank. Write the net…
A:
Q: hello i need help with this question Assuming standard conditions, answer the following questions.…
A: “Since you have posted multiple subparts questions, we will provide the solution only to the first…
Q: What would be the major product if diethyl malonate (EtO2CCH2CO2Et) was decarboxylated using H3O+…
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: It takes 151. kJ/mol to break an iodine-iodine single bond. Calculate the maximum wavelength of…
A:
Q: Consider the following reaction. Which side is favored in equilibrium? u ů ^-^ 1 Selected answer…
A: A base abstracts proton. A nucleophile adds to the electrophilic center.
Q: Discovering the 5 types of chemical reactions Reaction Combustion Decomposition Single replacement…
A: We have been given chemical reactions and we have been asked the observations of these reactions. We…
Q: d. NaOH(aq) + Al2(SO4)3(aq) → Na2SO4(aq) + Al(OH)3(s) 7. What is the mole fraction of each of the…
A:
Q: Complete the Ka2 expression for H₂CO3 in an aqueous solution. Ka2 = 4.69 x 10-11 =
A:
Q: A solution of 45 g of sucrose (molar mass 342 g/mol) in 1 kg of water may be approximated as an…
A: Since you have posted a question with multiple subparts, we can only solve first 3 subparts for you…
Q: What is the K₂ reaction of HCN? Ka reaction: The K₂ of HCN is 6.2 x 10-10. What is the K, value for…
A: It is based on the concept of acid and base Here we are required to find the expression of acid…
Step by step
Solved in 2 steps with 1 images

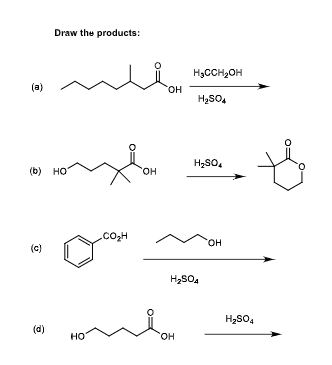
- Melamine, used as a fire retardant and a component of the writing surface of white boards, can be prepared from s-trichlorotriazine through a series of SNAr reactions with ammonia. The first substitution takes place rapidly at room temperature. The second substitution takes place near 100 °C, and the third substitution requires even higher temperature and pressure. Provide an explanation fatr this reactivity.Identify A–E.Draw and name compounds that meet the following descriptions: (a) Three acid chlorides having the formula C6H9ClO (b) Three amides having the formula C7H11NO
- Treatment of compound A (C8H17Br) with NaOCH2CH3 affords two constitutional isomers B and C. Ozonolysis of B affords CH2=O and (CH3CH2CH2)2C=O. Ozonolysis of C affords CH3CH2CH2COCH3 and CH3CH2CHO. What is the structure of A?Draw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. Part (b), then CH3COCl, AlCl3Give two syntheses for (CH3)2CH¬O¬CH2CH3, and explain which synthesis is better
- Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNWhat is the basicity of H2SO4The shrub ma huang (Section 5.4A) contains two biologically activestereoisomers—ephedrine and pseudoephedrine—with two stereogeniccenters as shown in the given structure. Ephedrine is one component ofa once-popular combination drug used by body builders to increaseenergy and alertness, whereas pseudoephedrine is a nasaldecongestant.a.) Draw the structure of naturally occurring (−)-ephedrine, which has the1R,2S configuration.b.) Draw the structure of naturally occurring (+)-pseudoephedrine, whichhas the 1S,2S configuration.c.) How are ephedrine and pseudoephedrine related?d.) Draw all other stereoisomers of (−)-ephedrine and (+) pseudoephedrine, and give the R,S designation for all stereogeniccenters.e.) How is each compound drawn in part (d) related to (−)-ephedrine?
- Treatment of compound A (C8H17Br) with NaOCH2CH3 affords twoconstitutional isomers B and C. Ozonolysis of B affords CH2=O and(CH3CH2CH2)2C=O. Ozonolysis of C affords CH3CH2CH2COCH3 andCH3CH2CHO. What is the structure of A?Rank the compounds in each set in order of increasing acid strength.(a) CH3CH2COOH CH3CHBrCOOH CH3CBr2COOHDraw the products formed when phenol (C6H5OH) is treated with following set of reagents. [1] CH3CH2Cl, AlCl3; [2] Br2, hν



