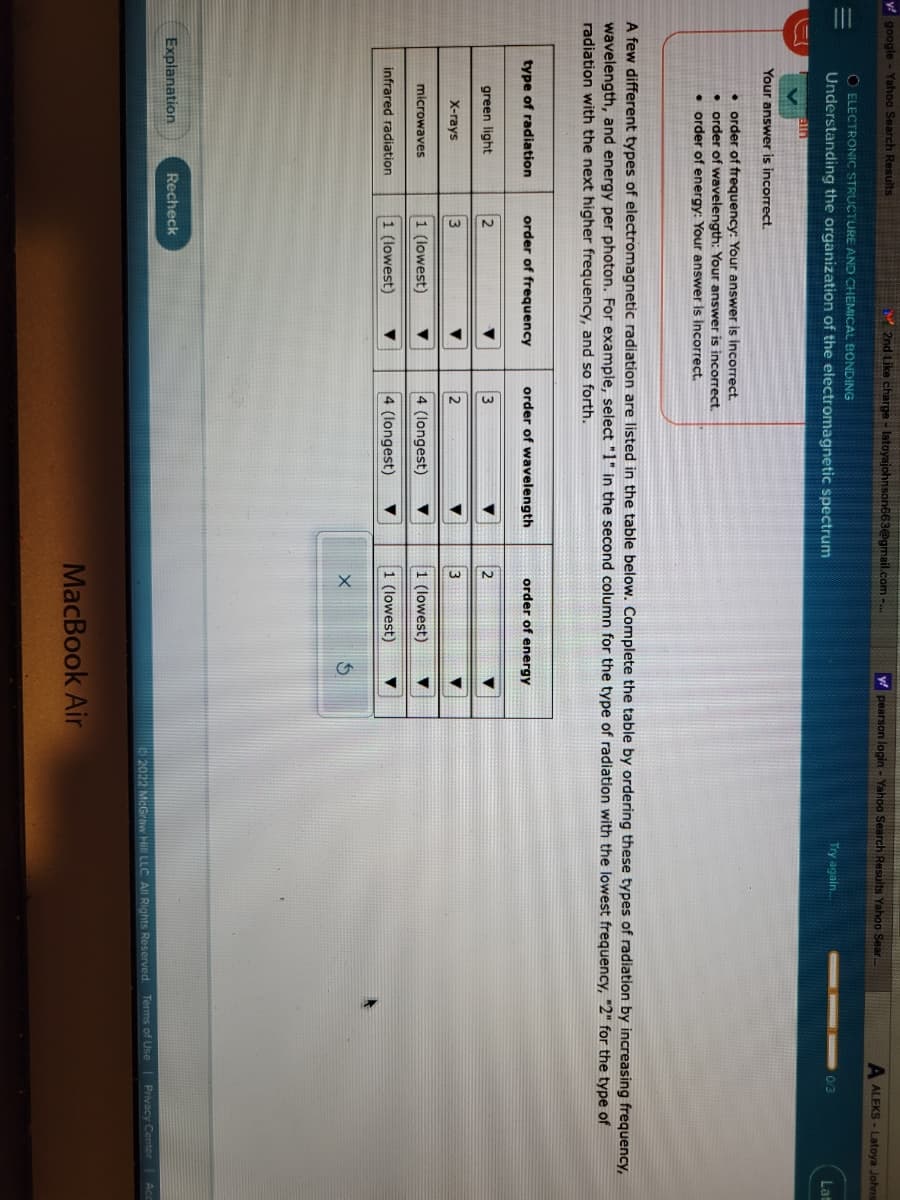
A few different types of electromagnetic radiation are listed in the table below. Complete the table by ordering these types of radiation by incre wavelength, and energy per photon. For example, select "1" in the second column for the type of radiation with the lowest frequency, "2" for t radiation with the next higher frequency, and so forth. type of radiation green light X-rays microwaves infrared radiation order of frequency 2 3 1 (lowest) 1 (lowest) ▼ ▼ order of wavelength 3 2 4 (longest) 4 (longest) ▼ ▼ order of energy 2 3 1 (lowest) 1 (lowest) X $ ▼ ▼
A few different types of electromagnetic radiation are listed in the table below. Complete the table by ordering these types of radiation by incre wavelength, and energy per photon. For example, select "1" in the second column for the type of radiation with the lowest frequency, "2" for t radiation with the next higher frequency, and so forth. type of radiation green light X-rays microwaves infrared radiation order of frequency 2 3 1 (lowest) 1 (lowest) ▼ ▼ order of wavelength 3 2 4 (longest) 4 (longest) ▼ ▼ order of energy 2 3 1 (lowest) 1 (lowest) X $ ▼ ▼
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter6: The Structure Of Atoms
Section: Chapter Questions
Problem 59GQ: If sufficient energy is absorbed by an atom, an electron can be lost by the atom and a positive ion...
Related questions
Question
Transcribed Image Text:y google - Yahoo Search Results
O ELECTRONIC STRUCTURE AND CHEMICAL BONDING
Understanding the organization of the electromagnetic spectrum
Your answer is incorrect.
order of frequency: Your answer is incorrect.
• order of wavelength: Your answer is incorrect.
• order of energy: Your answer is incorrect.
type of radiation
green light
X-rays
microwaves
A few different types of electromagnetic radiation are listed in the table below. Complete the table by ordering these types of radiation by increasing frequency,
wavelength, and energy per photon. For example, select "1" in the second column for the type of radiation with the lowest frequency, "2" for the type of
radiation with the next higher frequency, and so forth.
infrared radiation
2nd Like charge - latoyajohnson663@gmail.com -...
Explanation
order of frequency
2
3
1 (lowest)
1 (lowest)
Recheck
▼
▼
▼
order of wavelength
3
2
4 (longest)
4 (longest)
▼
▼
▼
order of energy
2
3
1 (lowest)
1 (lowest)
y pearson login - Yahoo Search Results Yahoo Sear...
X
$
Try again...
MacBook Air
A ALEKS-Latoya Johns
0/3
Lat
2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Acc
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning